Oxidative Cleavage of γ-Hydroxyalkenals
A R T I C L E S
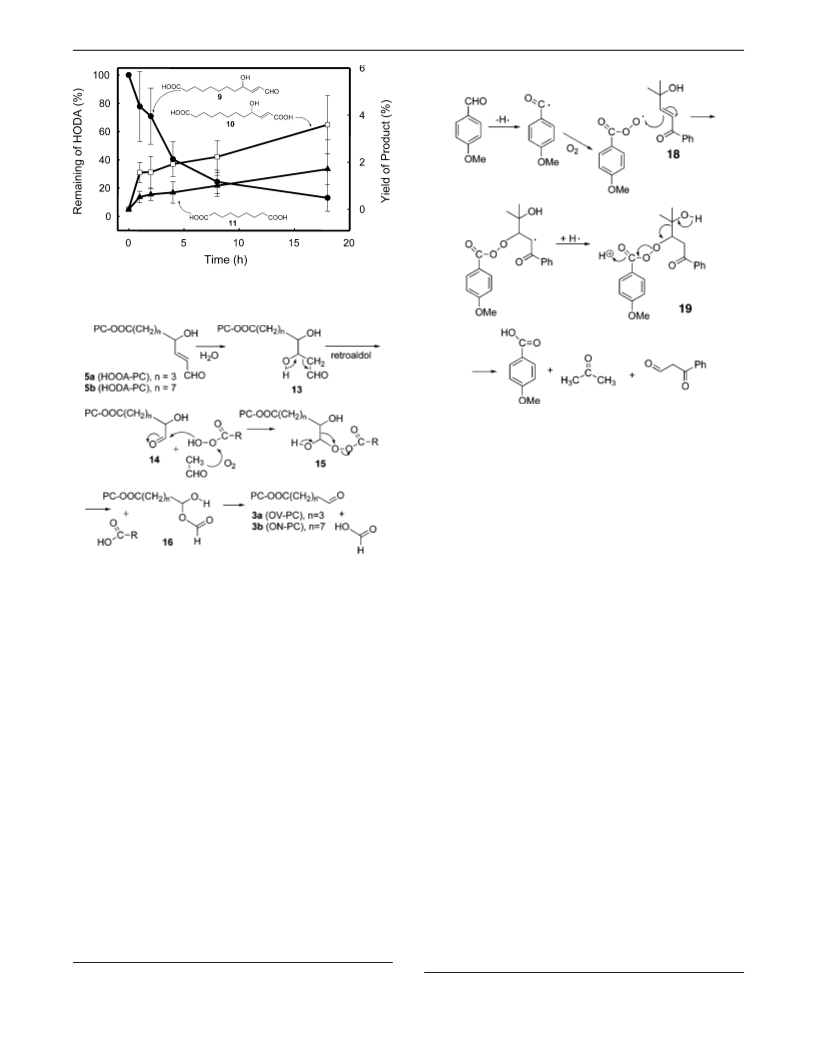
reaction of 18 that is catalyzed by anisic acid presumably
generates dihydrofuran 20. A pair of resonances at 1.34 and
High-performance liquid chromatography (HPLC) was performed with
HPLC grade solvents using a Waters M600A solvent delivery system
and a Waters U6K injector. The eluents were monitored using a SEDEX
1
.53 ppm in the NMR spectrum of the reaction mixture are
5
5 evaporative light scattering detector and an ISCO V4 UV-visable
consistent with the two nonequivalent methyl groups of the
putative dihydrofuran 20.
detector. Analytical RP-HPLC was performed on a Phenomenex LUNA
C18 (2) column (4.6 mm i.d. × 25 cm). Semipreparative RP-HPLC
was performed on a Phenomenex LUNA C18 (2) column (10 mm
i.d. x 25 cm). Flash column chromatography was performed on 230-
4
00 mesh silica gel supplied by E. Merck. All reactions in an inert
atmosphere were in argon or nitrogen. All chemicals were obtained
from Aldrich unless otherwise specified.
The key step in our proposed mechanism for the oxidative
cleavage HODA-PC to give ON-PC is the fragmentation of a
â-hydroxyperester. Precedent for the fragmentation of our
putative intermediate 19 is provided by the closely analogous
fragmentation of â-hydroxyperester 21.2 The same driving
force, simultaneous generation of two carbonyl groups, promotes
the analogous fragmentations of the â-hydroxydialkylperoxide
Liquid Chromatography/Mass Spectrometry. LC/ESI/MS/MS
analysis of autoxidation reaction product mixtures was performed on
a Quattro Ultima (Micromass, Wythenshawe, UK) connected to a
Waters 2790 solvent delivery system with an auto-injector. The source
temperature was maintained at 100 °C, and the desolvation temperature
8
2
at 200 °C. The drying gas (N ) was maintained at ca. 450 L/h, and the
cone flow gas at ca. 50 L/h. The multiplier was set at an absolute value
of 500. MS scans at m/z 20-400 were obtained for standard compounds.
Argon was used as collision gas at a pressure of 5 psi for MS/MS
analysis. For MS/MS analysis, the collision energy was optimized for
each compound. For multiple reaction monitoring (MRM) experiments,
the optimum collision energy (giving the strongest signal) was
determined for each m/z ion pair. For acidic compounds, the mass
spectrometer was operated in the negative ion mode. For PFB
derivatives, it was operated in the positive ion mode. Online chro-
matographic separation was achieved using a 150 × 2.0 mm i.d. Prodigy
ODS-2, 5 µ column (Phenomenex, UK), with a binary solvent (water
and methanol) gradient. The solvents were supplemented with 0.2%
formic acid, whenever the mass spectrometer was operated in positive
mode. For acidic compounds, the gradient started with 100% water
and rose to 100% methanol linearly in 15 min, and elution was
continued for 5 min with 100% methanol. Then the gradient was
reversed to 100% water in 0.5 min and then held for 9.5 min at 100%
water. For dipentaflurobenzyl ester compounds, the gradient started
with 85% methanol, rose to 88% methanol linearly in 12 min, and
then rose to 100% methanol linearly over 3 min, which was held for
29
30
2
2
and the â-hydroxyhydroperoxide 23. The oxidative
cleavage of ribulose bisphosphate is an especially pertinent
example that occurs in vivo during photorespiration. It involves
fragmentation of the â-hydroxyhydroperoxide 24.31
1
2 min. Then the gradient was reversed to 85% methanol in 4 min and
held for 10 min. The solvents were delivered at 200 µL/min.
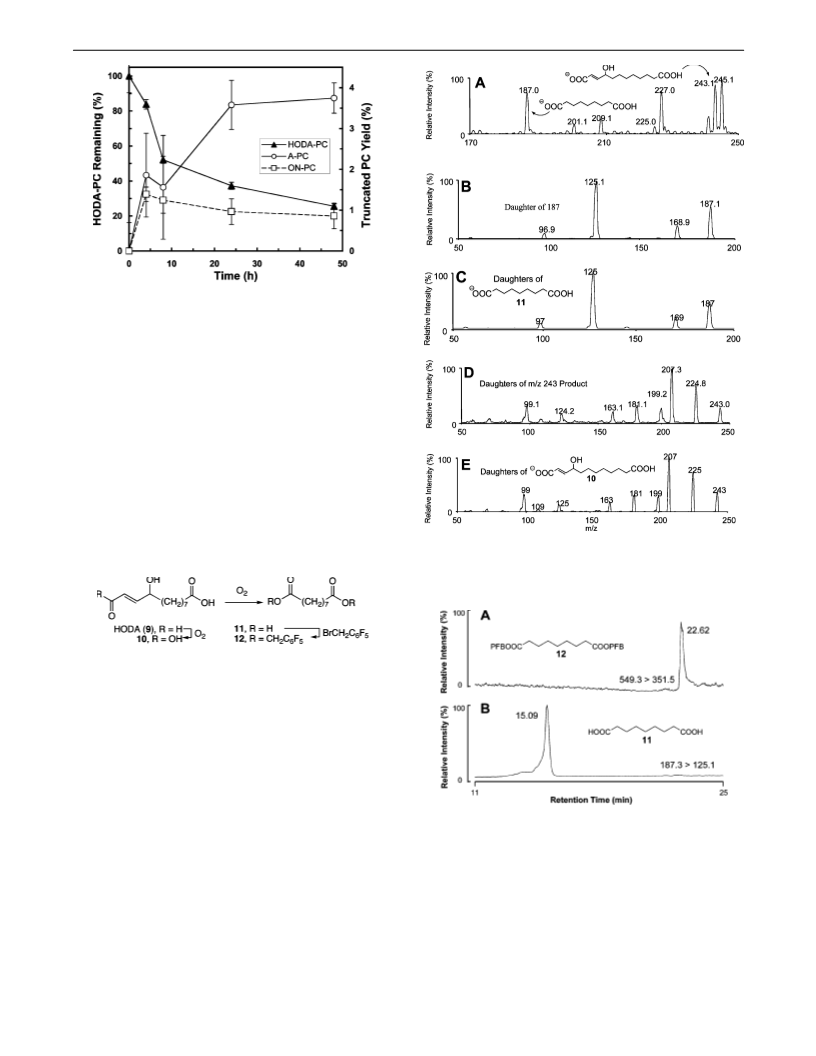
-Hydroxydodec-2-enedioic acid (10) was synthesized by selective
oxidation of HODA (9). A mixture of NaClO (11 mg, 0.12 mmol),
NaH PO (8.3 mg, 0.06 mmol), 2-methyl-2-butene, and t-BuOH/H O
4
Experimental Procedures
2
1
General Methods. All proton magnetic resonance ( H NMR) spectra
2
4
2
were recorded on Varian Gemini spectrometers operating at 200 or
(5/1 (v/v), 0.5 mL) was added to a flask containing HODA (9 mg,
0.04 mmol). The resulting mixture was stirred for 2 h. The product
was extracted with ethyl acetate. Flash chromatography (hexane/ethyl
3
00 MHz. Proton chemical shifts are reported in parts per million on
the δ scale relative to tetramethylsilane (δ 0.00) or CDCl (δ 7.27) or
(δ 7.157). H NMR spectral data are tabulated
3
1
1
CD
3
OD (δ 3.30) or C
D
6 6
acetate (30/70)) produced 10 (7 mg, 71%). H NMR (CD OD, 200
3
in terms of multiplicity of proton absorption (s, singlet; d, doublet; dd,
doublet of doublets; t, triplet; m, multiplet; br broad), coupling constants
MHz) δ 6.79 (dd, J ) 15.6 Hz, J ) 5.3 Hz, 1H), 5.97 (dd, J )
1
2
1
15.6 Hz, J ) 1.5 Hz, 1H), 6.28 (dd, J ) 15.6 Hz, J ) 7.8 Hz, 1H),
2
1
2
1
3
(
Hz), number of protons. Carbon magnetic resonance ( C NMR) spectra
were recorded on a Varian Gemini spectrometer operating at 50 MHz.
These spectra are reported in ppm on the δ scale relative to CDCl (δ
7.23) or CD OD (δ 49.0). Proton and carbon NMR samples were
analyzed as solutions in CDCl or CD OD or C
4.1-4.2 (m, 1H), 2.26 (t, J ) 7.2 Hz, 2 H), 1.4-1.7 (4H), 1.2-1.4
(8H).
Dipentaflurobenzyl azeleate (12) was prepared by stirring a mixture
of azeleic acid (11, 100 µg) in dry acetonitrile (100 µL), containing
3
7
3
3
3
6 6
D .
1
0 wt % pentafluorobenzyl bromide and 20 wt % N,N-diisopropyleth-
Chromatography was performed with ACS grade solvents (ethyl
acetate, hexane, chloroform, and methanol). Thin-layer chromatography
ylamine, for 2 h. After the solvent was evaporated with nitrogen, the
residue was dissolved in 1 mL of water, and the products were extracted
with 1 mL of ethyl acetate. ESI-MS/MS analysis in the positive ion
(TLC) was performed on glass plates precoated with silica gel (Kieselgel
6
0 F254, E. Merck, Darmstadt, West Germany). R values are quoted
f
+
mode produced [MH] at 549 and characteristic fragmentations: at
for plates of thickness 0.25 mm. TLC plates were visualized by viewing
the developed plates under short-wavelength UV light or with iodine.
m/z 351.5, m/z 181.
Autoxidation of HODA-PC. Glass tubes (1 mL) containing a dry
film of HODA-PC (100 ng) were heated in air at 37 °C. After
incubation, the vials were stored at -78 °C until analysis. Before
analysis, 1,2-ditridecanoyl-sn-glycero-3-phosphatidylcholine (DT-PC,
3
2
Aldehydes were visualized with Purpald reagent, peroxides with
3
3,34
and phospholipids with molybdenum spray.35
thiocyanate reagent,
(
(
(
(
28) Witkop, B.; Patrick, J. B. J. Am. Chem. Soc. 1951, 73, 2196-2200.
29) Nishinaga, A.; Rindo, K.; Matsuura, T. Synthesis 1986, 1038-1041.
30) Kwon, B. M.; Foote, C. S. J. Am. Chem. Soc. 1988, 110, 6582-6583.
31) Voet, D.; Voet, J. G.; Pratt, C. W. Fundamentals of Biochemistry 1999,
(33) Abraham, M. H.; Davies, A. G.; Llewellyn, D. R.; Thain, E. M. Anal. Chim.
Acta 1957, 17, 499-503.
(34) Summerbell, R. K.; Hyde, D. K. A. J. Org. Chem. 1960, 25, 1809-1811.
(35) Dittmer, J. C.; Lester, R. L. J. Lipids Res. 1964, 5, 126-127.
5
56.
(32) Hopps, H. B. Aldrichimica Acta 2000, 33, 28-30.
J. AM. CHEM. SOC.
9
VOL. 126, NO. 37, 2004 11527

 Balamraju, Yuvaraju N.
Balamraju, Yuvaraju N.
 Sun, Mingjiang
Sun, Mingjiang
 Salomon, Robert G.
Salomon, Robert G.