Confined-Plume Chemical Deposition
A R T I C L E S
Non-laser-induced thermal treatments were conducted under
continuous gas flow inside a quartz tube using a one-foot Linberg/
Blue tube furnace or inside a bell-jar chamber within a modified
Milestone Drydist microwave reactor (operating at 2.45 GHz).
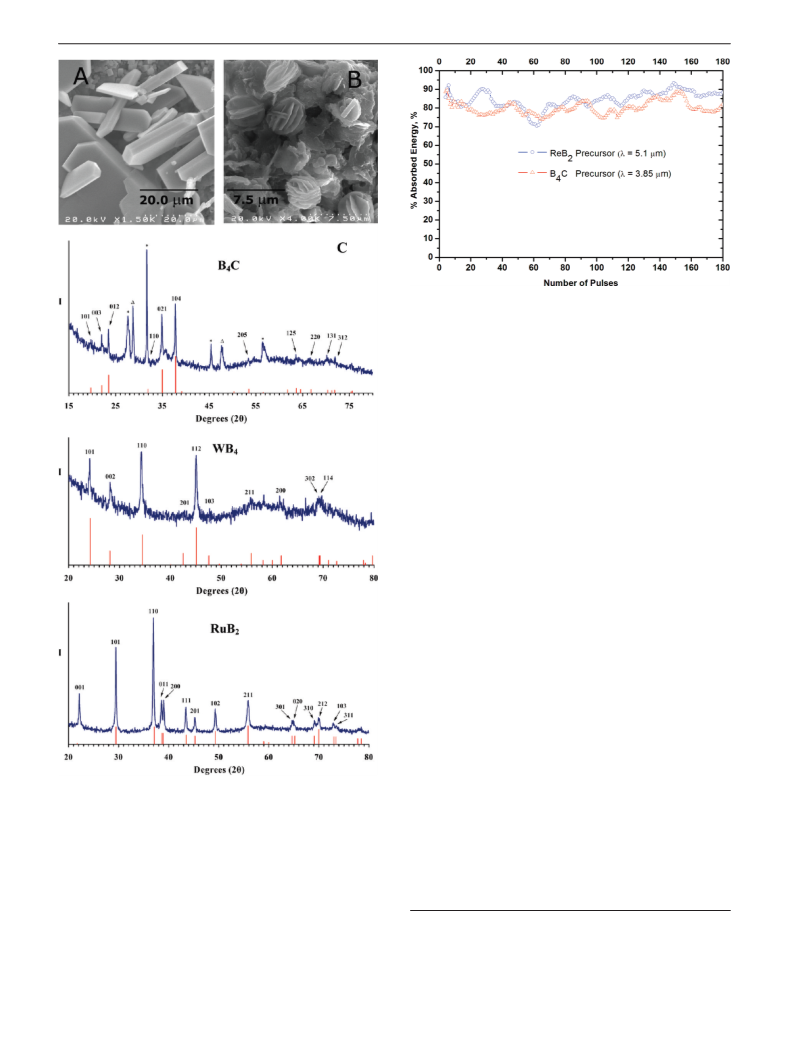
Powder X-ray diffraction scans were obtained on a Scintag X1 θ/θ
automated powder X-ray diffractometer with a Cu target, a Peltier-
cooled solid-state detector, and a zero-background Si(510) sample
support. Powder XRD patterns were analyzed using DICVOL04
to determine space group and approximate unit cell dimensions.
Refined unit cell dimensions were calculated using Checkcell to
give the following composition, space group, and unit cell
parameters [PDF card data] for the microcrystalline materials
prepared by CPCD processing: ReB2, P63/mmc, a ) 2.902(1) Å, c
) 7.487(4) Å [PDF Card #11-0581; P63/mmc, a ) 2.900 Å, c )
7.478 Å]; RuB2, Pmmn, a ) 4.628(7) Å, b ) 2.860(8) Å, c )
4.046(6) Å [PDF Card #15-0213; Pmmn, a ) 4.641 Å, b ) 2.863
Å, c ) 4.044 Å]; WB4, P63/mmc, a ) 5.22(3), c ) 6.326(2) [PDF
low softening temperature of Ultralene film (120 °C) results in
polymer film fusion near the edges of the irradiated area. During
the course of this study, XRD data confirmed that ReB2 particles
formed on Si wafers undergo slow air oxidation to form ReO3.
Air oxidation of metal diboride materials is known and has been
studied in detail for ZrB2 and HfB2.37 Being able to prepare
metal-boride materials with simultaneous surface-passivation
by a shrink-wrapping polymer would be desirable in the
formation of ceramics susceptible to oxidation.
Conclusions
Irradiation of single-source or dual-source preceramic precur-
sors confined between hard supports, such as Si wafer or NaCl
plates, by resonant, pulsed FEL IR light tuned to precursor
carbonyl Ct O or B-H stretching band frequencies initiates
reaction plume formation and deposition of microcrystalline
coatings of ReB2, RuB2, WB4, or B4C of high purity. Using
off-resonant FEL IR irradiation precludes reaction plume
formation. Relaxation of spatial precursor confinement leads to
element separation. Microcrystalline coatings of known ceramic
compositions are produced from this confined-plume, chemical
deposition process by rastering the FEL laser beam over a
specimen area. Delivery of resonant FEL IR light as a micro-
second train of picosecond pulses provides temporal confinement
of reaction plume formation, such that crystalline ceramic
coatings can be produced on soft supports without causing
noticeable thermal damage. Microcrystalline coatings of ReB2
can be formed directly from a single-source precursor on IR-
transmissive IR2, Teflon, or Ultralene polymer films with
Ultralene polymer “shrink wrapping” the ReB2 product during
CPCD processing because of its low softening temperature.
Exploration of the scope of CPCD as a synthesis strategy for
the preparation of other known or postulated hard/superhard
materials and more detailed study of the mechanism of ceramic
deposition during CPCD processing is anticipated.
j
Card #19-1373; P63/mmc, a ) 5.200 Å, c ) 6.340 Å]; B4C, R3m,
j
a ) 5.549(7), c ) 12.168(2) [PDF Card #35-0798; R3m, a ) 5.600
Å, c ) 12.08 Å].
SEM images were obtained on a Hitachi S-2400 electron
microscope with an accelerating voltage at either 5 or 20 kV.
Transmission electron microscopic (TEM) bright-field micrographs
were recorded on a 200 kV Phillips CM20T TEM.
Confined-Plume Chemical Decomposition Processing Method.
Infrared light produced by the Vanderbilt FEL is tunable from 2 to
9 µm. For a typical experiment intended to synthesize ReB2, FEL
IR radiation of either 5.1 µm (1960 cm-1; tuned to the most intense
Ct O stretching vibration of precursor 1) or 3.9 µm (2564 cm-1
;
tuned to the most intense B-H stretching vibration of precursor 1)
was delivered to samples as 4.0-µs macropulses (30 Hz) where each
macropulse of 21.5 mJ energy consists of a series of 0.8- to 1-ps
micropulses delivered 350 ps apart. Si wafers (plain or Si(510)
wafers) and NaCl wafers were used as sample supports in a
sandwich arrangement where precursor (0.05 mL) was placed
between two substrates forming a NaCl/precursor 1/Si wafer
assembly. Samples were scanned in a rastering fashion from above
the sample through the top NaCl wafer by the FEL IR beam (linear
scans (0.5 mm/s) separated spatially by 150 µm). The FEL IR beam
was delivered as a spot 350 µm in diameter. FEL experiments were
conducted in the open, ambient atmosphere, and samples were
stored in sealed glass bell jar flushed with nitrogen.
Experimental Section
Reagents and General Methods. Re(CO)5Br was either pur-
chased from Strem Chemicals, Inc. or synthesized from Re2(CO)10
and bromine.42 Complex Re(CO)4(B3H8), 1, was prepared as
described in the literature.43 Note: Re(CO)4(B3H8) is moderately
air sensitive but can be stored indefinitely as a solid under nitrogen
or vacuum in sealed ampules below 0 °C. Re2(CO)10, Ru3(CO)12,
W(CO)6, B10C2H12 (o-carborane), and B10H14 (decaborane) were
purchased from Strem Chemicals, Inc. [Me4N][B3H8] was purchased
from Dr. Lee J. Todd, Indiana University. All other reagents were
purchased from Sigma-Aldrich. Thin-film 3525 Ultralene was
purchased from Spex CertiPrep, softened at 120 °C, and pressed
into wafers ca. 0.5-mm thick. IR2 polymer was purchased from
Fresnel Technologies, Inc. Teflon was purchased from Dupont, and
Kevlar was purchased from Fiber Glast Development Corporation.
Doped silicon wafers (phosphorus, 1-10 ohms ·cm) type N(100)
used in microwave experiments were purchased from Silicon, Inc.
and were cut to the desired sizes as needed. Undoped, polished
silicon fragments with a (510) orientation were purchased from Mr.
Dana Smith at The Gem Dugout, Pennsylvania, and cut to the
desired sizes as needed. Polished undoped Si(510) wafers were used
to prepared samples for easy XRD characterization. Polished and
unpolished circular NaCl single-crystal plates 13 mm × 2 mm and
13 mm × 1 mm in size were purchased from International Crystal
Laboratories. A commercial FTIR pellet holder (13-mm diameter)
was used as the sample holder for FEL laser experiments.
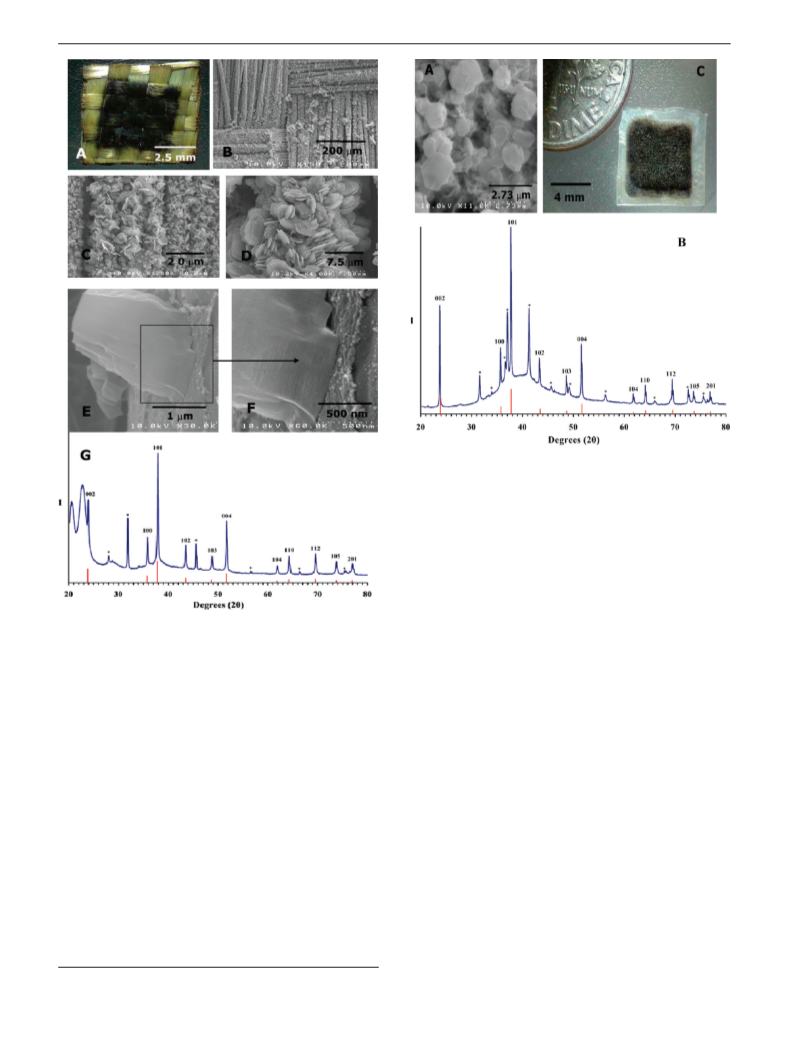
Single-Source Precursor Synthesis of ReB2. Re(CO)4(B3H8)
was either deposited (0.05 mL) onto a polished 13 mm × 1 mm
NaCl wafer or impregnated on woven Kevlar fabric. The precursor-
loaded NaCl or NaCl/Kevlar half assembly was physically pressed
together with a Si(510) or clean NaCl wafer forming a completed
sandwich assembly and was loaded into a sample holder. FEL
irradiation (λ ) 5.1 µm, E ) 21.5 mJ) was rastered over the
sandwich assembly, and then the treated sandwich assembly was
characterized by SEM and XRD. ReB2 film thickness, measured
by SEM, for the coated Kevlar fabric specimen is 5-7 µm.
Single-Source Precursor Synthesis of B4C. O-Carborane (0.052
g, 0.091 mmol) was dissolved in benzene (1 mL), and the resulting
solution was deposited (0.05 mL) onto a polished 13 mm × 1 mm
NaCl wafer and allowed to dry. The precursor-loaded NaCl wafer
and a Si(510) wafer were physically pressed together in a sandwich
assembly and were loaded into a sample holder. FEL irradiation
(λ ) 3.85 µm, E ) 33.0 mJ) was rastered over the sandwich
assembly, and then the treated sandwich assembly was characterized
by SEM and XRD. B4C film thickness, estimated by SEM, for this
specimen ranges from 4 to 25 µm.
Dual-Source Precursor Synthesis of WB4. W(CO)6 (0.032 g,
0.091 mmol) and decaborane (0.077 g, 0.630 mmol) were codis-
solved in tetrahydrofuran (1 mL), and the resulting solution was
deposited (0.05 mL) onto a polished 13 mm × 1 mm NaCl wafer
and allowed to dry. The precursor-loaded NaCl wafer and a Si(510)
wafer were physically pressed together in a sandwich assembly and
loaded into a sample holder. FEL irradiation (λ ) 5.1 µm, E )
(42) Schmidt, S. P.; Trogler, W. C.; Basolo, F.; Urbancic, M. A.; Shapley,
J. R. Inorg. Synth. 1990, 28, 160.
(43) Gaines, D. F.; Hildebrandt, S. J. Inorg. Chem. 1978, 17, 794.
9
J. AM. CHEM. SOC. VOL. 131, NO. 33, 2009 11749

 Ivanov, Borislav L.
Ivanov, Borislav L.
 Wellons, Matthew S.
Wellons, Matthew S.
 Lukehart, Charles M.
Lukehart, Charles M.