252
SHARMA AND EBADI
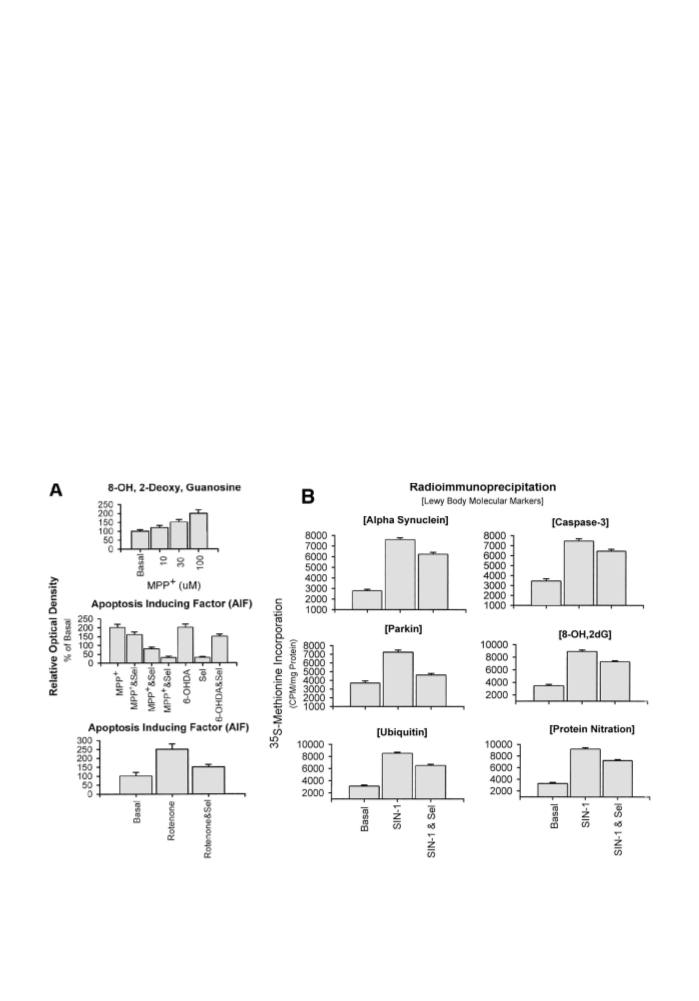
tion, caspase-3 activation, and mitochondrial 8-hydroxy-2-
deoxyguanosine (8-OH-2dG) synthesis in human DA (SK-N-
SH) cell line, and in MT-overexpressed striatal DA neurons.
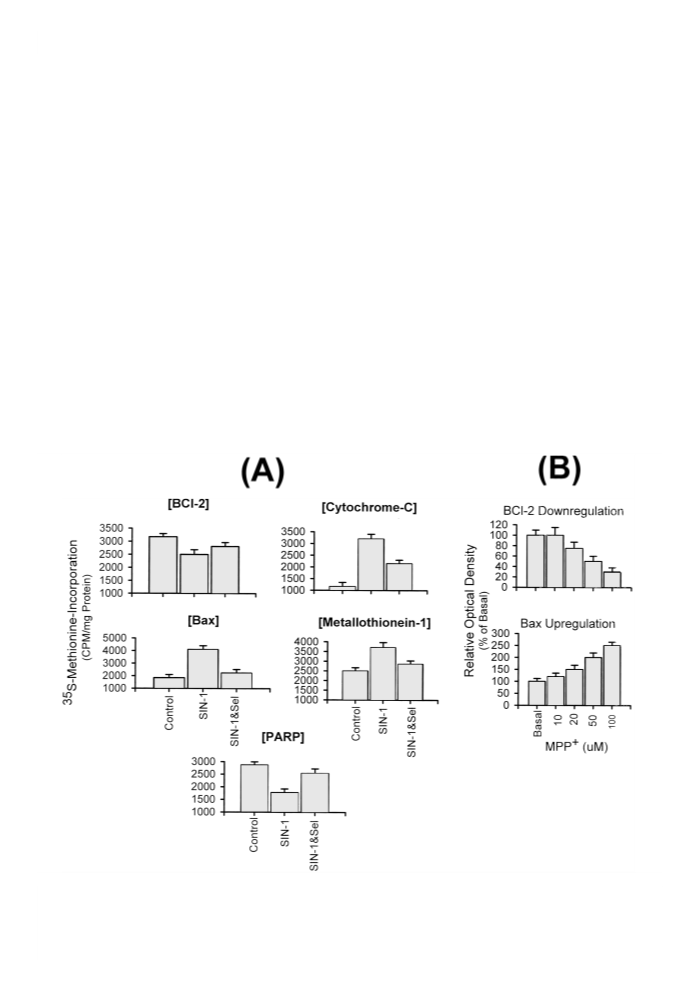
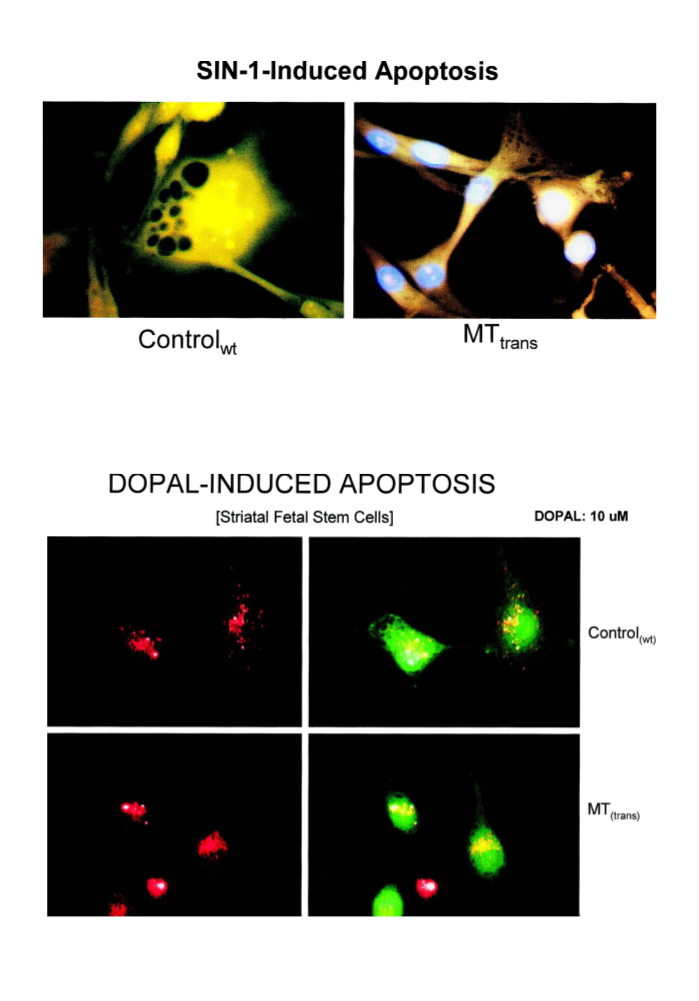
These studies have shown that selegiline affords neuroprotec-
tion by enhancingMT gene expression and by down-regulating
a-Syn expression, a Lewy body molecular marker. MT pre-
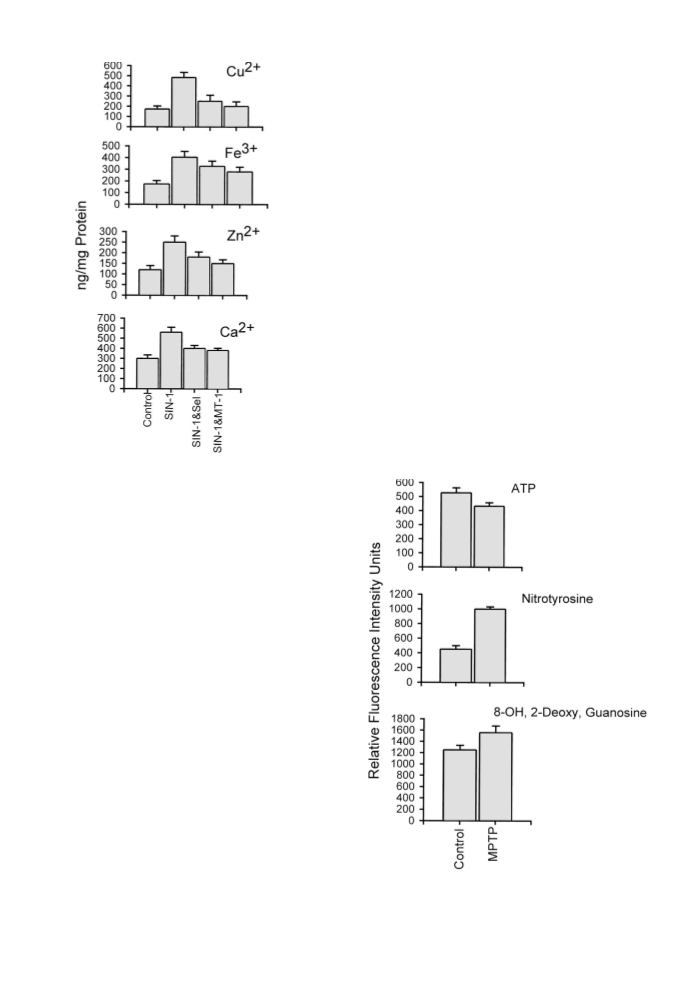
treatment also attenuated SIN-1-induced intramitochondrial
accumulation of metal ions (Cu2+, Fe3+, and Ca2+), known to
be involved in the etiopathogeneis of PD, suggesting a possi-
ble neuroprotective potential for MT gene overexpression in
attenuating neurotoxin-induced parkinsonism.
INTRODUCTION
LTHOUGH THE EXACT ETIOPATHOGENESIS OF PARKINSON’S
DISEASE (PD) remains unknown, it has been hypothe-
A
sized that the neuronal demise of nigrostriatal dopaminergic
neurons (DA neurons) could occur due to the production of
endogenous neurotoxins, such as tetrahydroisoquinolines, or
by exposure to various environmental neurotoxins, such as
rotenone (15). These neurotoxins produce a significant down-
regulation of mitochondrial complex-1 (ubiquinone NADH-
oxidoreductase), as observed in the majority of PD patients.
Furthermore, significantly reduced glutathione in the sub-
stantia nigra (SN) enhances the risk of free radical [mainly
hydroxyl (?OH) and nitric oxide (?NO)] overproduction, lead-
ing to neuronal damage in PD.
MATERIALS AND METHODS
NO plays a critical role in mediating neurotoxicity associ-
ated with various neurological disorders, such as stroke, PD,
HIV dementia (1, 5, 9, 16, 20), and multiple sclerosis (50). In
the SN of PD patients, a significant increase in the density of
glial cells expressing tumor necrosis factor-a, interleukin-1b,
and interferon-g has been observed. Although CD23 was not
detectable in the SN of control subjects, it was found in both
astroglial and microglial cells of parkinsonian patients, indi-
cating the existence of cytokine/CD23-dependent activation
pathways of inducible NO synthase (iNOS) and of proinflam-
matory mediators in glial cells and their involvement in the
pathophysiology of PD (21). In addition to NO, accumulation
of iron in the SN has been implicated in the death of DA neu-
Chemicals
Cell culture materials, including powdered Dulbecco’s
modified Eagle medium (DMEM), fetal bovine serum, strepto-
penicillin, Ham’s F-12 medium, trypsin, and specific primers
for reverse transcription–polymerase chain reaction (RT-PCR)
were purchased from GibcoBRL Life Technologies (Rock-
ville, MD, U.S.A.). First-strand cDNA kit was purchased
from Stratagene (La Jolla, CA, U.S.A.). Mouse anti-MT anti-
body was purchased from Zymed Laboratories (San Fran-
cisco CA, U.S.A.). Anti-a-Syn antibody, apoptosis-inducing
factor (AIF), and anti-nitrotyrosine polyclonal antibodies were
purchased from Chemicon International Inc. (Temecula, CA,
U.S.A.). Caspase-3 assay kit was purchased from Pharmin-
gen, Becton–Dickinson (Palo Alto, CA, U.S.A.). [35S]Methio-
nine was purchased from NEN Dupont (Boston, MA, U.S.A.).
ATP-LiteTM kit was purchased from Packard (Meriden, CT,
U.S.A.). Vector (pEGFP-N1) was purchased from BD Bio-
science Clontech (Palo Alto, CA, U.S.A.). Liposome-based
Effectine Transfection reagent with DNA enhancer was pur-
chased from Qiagen Inc. (Stanford, CA, U.S.A.). Protein assay
dye was purchased from Bio-Rad Laboratories (Hercules,
CA, U.S.A.). ECL chemiluminescence kit and nitrocellulose
membranes were purchased fromAmersham Bioscience Corp.
(Piscataway, NJ, U.S.A.). Mitochondrial membrane potential
(DC) fluorochrome, 5,596,69-tetrachloro-1,19,3,39-tetraethyl-
benzimidazolocarbocyanide iodide (JC-1), nucleocytoplas-
mic fluorochromes, 49,6-diamidino-2-phenylindole dihydro-
chloride (DAPI), ethidium bromide, acridine orange, and
fluorescein isothiocyanate (FITC)-conjugated antimouse IgG
were purchased from Molecular Probes (Eugene, OR, U.S.A.).
All other chemicals were of reagent grade quality and were
purchased from Sigma Chemical Company (St. Louis, MO,
U.S.A.).
2
rons in PD (55, 56). Peroxynitrite (ONOO ) ions, generated
in the mitochondria by Ca2+-dependent NO synthase (NOS)
activation during oxidative- and nitrative stress, readily react
with lipids, aromatic amino acids, or metalloproteins, inhibit-
ing mitochondrialrespiratory complexes, and hence are thought
to be involved in the etiopathogenesis of many diseases, in-
cluding PD (52).
Although NO has been shown to possess both apoptogenic
and apoptostatic properties, its overproduction during oxida-
tive and nitrative stresses could induce deleterious conse-
quences on mitochondrial complex-1 activity (6, 17). We
have discovered recently that metallothionein (MT) gene
overexpression in MT transgenic (MTtrans) mouse brain inhib-
ited the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine(MPTP)-
induced nitration of a-synuclein (a-Syn), and preserved mi-
tochondrial coenzyme Q10 levels, affording neuroprotection
against nitrative and oxidative stress of aging brain (48, 49).
In addition, MT isoforms are able to suppress 6-hydroxy-
dopamine (6-OHDA)-induced ?OH radical generation (48).
We have also reported that selegiline, a monoamine oxidase B
inhibitor, provides neuroprotection via MT gene overexpres-
sion (14).
Experimental animals
As the involvement of oxidative and nitrative stresses is
now advocated in the etiopathogenesis of PD (1, 5, 9, 16, 20),
a detailed study was needed to explore the exact molecular
mechanismof NO-mediated neurodegenerationand MT-induced
neuroprotection in PD. 3-Morpholinosydnonimine (SIN-1), a
vasorelaxant, a soluble guanylyl cyclase stimulator, and a po-
Experimental animals were housed in temperature- and
humidity-controlled rooms with a 12-h day and 12-h night
cycle and were provided with commercially prepared chow
ad libitum.
and water
The animals were acclimated to labora-
tory conditions for at least 4 days prior to experimentation.
Care was also taken to avoid any distress to animals during
the period of experiment. Breeder pairs of control wild-type
(controlwt) C57BJ6, MT double knockout (MTdko), and MTtrans
mice weighing 25–30 g were purchased from Jackson’s Labo-
ratories (Minneapolis, MN, U.S.A.). The animals were main-
2
tent ONOO generator, produced not only oxidative, but also
nitrative stresses in DA neurons, portending to play an impor-
tant role in understanding the exact etiopathogenesis of PD
(6). Therefore, we have investigated the extent of neuropro-
tection afforded by MT against SIN-1-induced lipid peroxida-

 Dupont
Dupont
 Dulou
Dulou