W. Saenger, J. Messinger, A. Zouni and V. K. Yachandra, Science,
2006, 314, 821.
2 J. L. Cape and J. K. Hurst, J. Am. Chem. Soc., 2008, 130, 827.
3 M. Orlandi, R. Argazzi, A. Sartorel, M. Carraro, G. Scorrano,
M. Bonchio and F. Scandola, Chem. Commun., 2010, 46, 3152;
F. Puntoriero, G. L. Ganga, A. Sartorel, M. Carraro, G. Scorrano,
M. Bonchio and S. Campagna, Chem. Commun., 2010, 46, 4725.
4 Y. V. Geletii, Z. Huang, Y. Hou, D. G. Musaev, T. Lian and
C. L. Hill, J. Am. Chem. Soc., 2009, 131, 7522; Z. Huang, Z. Luo,
Y. V. Geletii, J. W. Vickers, Q. Yin, D. Wu, Y. Hou, Y. Ding,
J. Song, D. G. Musaev, C. L. Hill and T. Lian, J. Am. Chem. Soc.,
2011, 133, 2068.
5 S. Roeser, P. Farras, F. Bozoglian, M. Martınez-Belmonte,
´
J. Benet-Buchholz and A. Llobet, ChemSusChem, 2011, 4, 197.
6 L. Duan, Y. Xu, P. Zhang, M. Wang and L. Sun, Inorg. Chem.,
2010, 49, 209; L. Duan, Y. Xu, M. Gorlov, L. Tong, S. Andersson
and L. Sun, Chem.–Eur. J., 2010, 16, 4659; Y. Xu, L. Duan,
L. Tong, B. Akermark and L. Sun, Chem. Commun., 2010,
46, 6506.
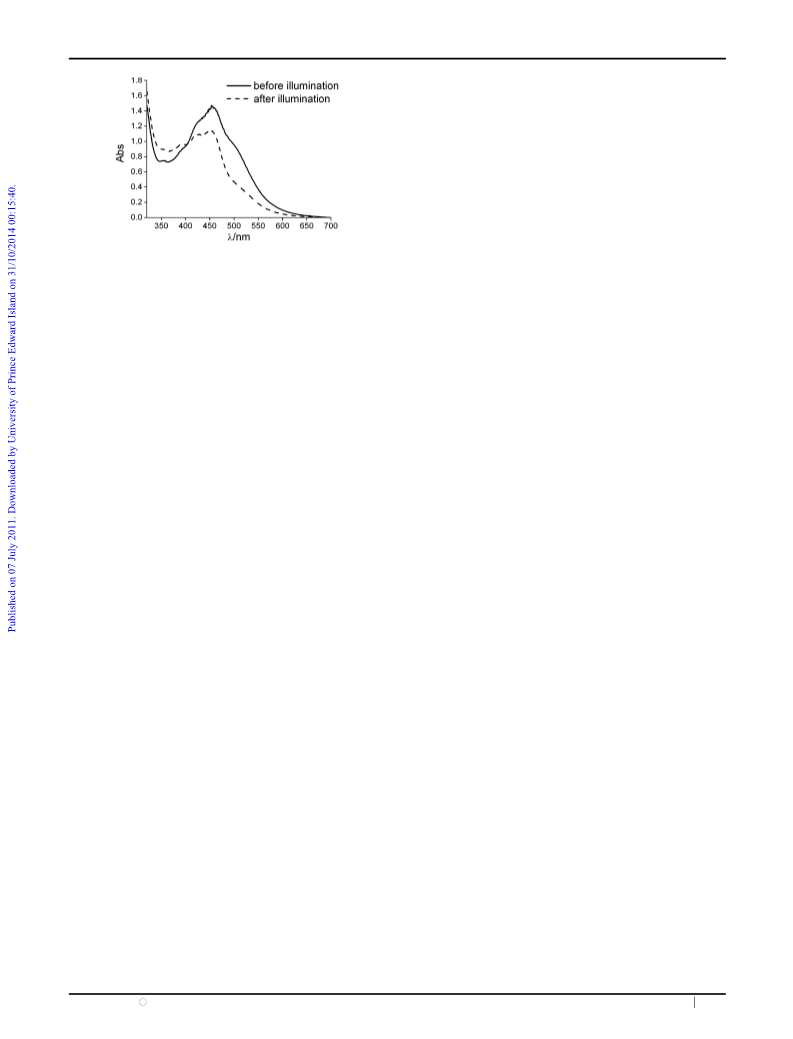
Fig. 1 The adsorption of phosphate buffer solution consisting of
complex (6
10ꢁ5 M), [Ru(bpy)3]2+ (3 10ꢁ5 M) and
1
ꢂ
ꢂ
[Co(NH3)5Cl]2+ (3 ꢂ 10ꢁ4 M) before (solid line) and after (dashed
line) visible light illumination.
7 B. Meunier, S. P. Visser and S. Shaik, Chem. Rev., 2004, 104, 3947;
M. Costas, M. P. Mehn, M. P. Jensen and L. Que, Jr., Chem. Rev.,
2004, 104, 939; M. M. Abu-Omar, A. Loaiza and N. Hontzeas,
Chem. Rev., 2005, 105, 2227.
8 J. T. Groves and Y. Watanabe, J. Am. Chem. Soc., 1988, 110,
8443–8452; B. Meunier, Chem. Rev., 1992, 92, 1411; L. Que, Jr.,
Acc. Chem. Res., 2007, 40, 493; W. Nam, Acc. Chem. Res., 2007,
40, 522.
80% yield (Table 2, entry 7). The remarkable catalytic abilities
of 1 and 2 could be expanded to other sulfides. For instance,
up to 91% yield (455 TON) and 100% selectivity in the
oxidation of p-methoxyphenylmethyl sulfide to the corres-
ponding sulfoxide was achieved (Table 2, entry 8).
The photooxidation process was investigated by means of
9 H. Inoue, M. Sumitani, A. Sekita and M. Hida, J. Chem. Soc.,
Chem. Commun., 1987, 1681; S. Takagi, M. Suzuki, T. Shiragami
and H. Inoue, J. Am. Chem. Soc., 1997, 119, 8712; T. Shiragami,
K. Kubomura, D. Ishibashi and H. Inoue, J. Am. Chem. Soc.,
1996, 118, 6311; S. Funyu, T. Isobe, S. Takagi, D. A. Tryk and
H. Inoue, J. Am. Chem. Soc., 2003, 125, 5734; H. Inone,
T. Shimada, Y. Kou, Y. Nabetani, D. Masui, S. Takagi and
H. Tachibana, ChemSusChem, 2011, 4, 173; H. Inoue, S. Funyu,
Y. Shimada and S. Takagi, Pure Appl. Chem., 2005, 77, 1019;
S. Funyu, M. Kinai, D. Masui, S. Takagi, T. Shimada,
H. Tachibana and H. Inoue, Photochem. Photobiol. Sci., 2010,
9, 931.
10 J. A. Treadway, J. A. Moss and T. J. Meyer, Inorg. Chem., 1999,
38, 4386.
11 W. Chen, F. N. Rein and R. C. Rocha, Angew. Chem., Int. Ed.,
2009, 48, 9672; W. Chen, F. N. Rein, B. L. Scott and R. C. Rocha,
Chem.–Eur. J., 2011, 17, 5595.
12 S. Fukuzumi, T. Kishi, H. Kotani, Y.-M. Lee and W. Nam, Nat.
Chem., 2010, 3, 38.
13 D. Kalita, B. Radaram, B. Brooks, P. P. Kannam and X. Zhao,
ChemCatChem, 2011, 3, 571.
14 H. Kotani, T. Suenobu, Y.-M. Lee, W. Nam and S. Fukuzumi,
J. Am. Chem. Soc., 2011, 133, 3249.
15 Y. Hirai, T. Kojima, Y. Mizutani, Y. Shiota, K. Yoshizawa and
S. Fukuzumi, Angew. Chem., Int. Ed., 2008, 47, 5772.
16 Y.-M. Lee, S. N. Dhuri, S. C. Sawant, J. Cho, M. Kubo, T. Ogura,
S. Fukuzumi and W. Nam, Angew. Chem., Int. Ed., 2009, 48, 1803.
17 J. P. Collin and J. P. Sauvage, Inorg. Chem., 1986, 25, 135.
18 C. L. Bailey and R. S. Drago, J. Chem. Soc., Chem. Commun.,
1987, 179; A. S. Goladstein and R. S. Drago, J. Chem. Soc., Chem.
Commun., 1991, 21; A. S. Goladstein, R. H. Beer and R. S. Drago,
J. Am. Chem. Soc., 1994, 116, 2424.
UV-Vis spectroscopy. As shown in Fig. 1, the spectrum of
[Ru(bpy)3]2+ and complex
1 mixture exhibits intensive
absorption bands at 420, 450 and 500 nm. Comparing with
their own absorptions, the first two bands at 420 and 450 nm
are ascribed to the MLCT transitions of [Ru(bpy)3]2+ and the
shoulder at 500 nm belongs to the MLCT transition of 1.
Upon light irradiation, the absorption at 500 nm gradually
disappears and the MLCT transition bands of [Ru(bpy)3]2+
are maintained. A similar phenomenon was also observed in
the photooxidation of 2 (Fig. S2, ESIw). It is possible that high
valent metal-oxo species such as [RuIV(dmp)2(H2O)(O)]2+
(Fig. S3, ESIw) and [RuIV(tpy)(bpy)(O)]2+ generated by
photoinduced electron transfer (Fig. S4, ESIw) are involved
in the current system.18 However, kinetic studies are required
to elucidate the mechanistic details.
In conclusion, we have demonstrated the high chemical and
photochemical catalytic properties of two polypyridyl ruthenium
complexes in the oxidation of hydrocarbons using water as an
oxidant. Specifically, this study represents a clean and efficient
method to highly selective sulfoxidation, making it attractive in
the practical relevance to green chemistry. Further investigations
are currently underway to perform enantioselective photo-
oxidation of sulfide with chiral catalysts.
We thank the National Basic Research Program of China
(973 program) (2009CB220009), the National Natural Science
Foundation of China (20923006), the Fundamental Research
Funds for the Central Universities (DUT11LK29), the State
Key Laboratory of Fine Chemicals, the Swedish Energy
Agency and K&A Wallenberg Foundation for financial
support of this work.
19 V. Kogan, M. M. Quintal and R. Neumann, Org. Lett., 2005,
7, 5039.
20 M. C. Carreno, Chem. Rev., 1995, 95, 1717; I. Fernandez and
´
N. Khiar, Chem. Rev., 2003, 103, 3651; H. Pellissier, Tetrahedron,
2006, 62, 5559; J. Legros, J. R. Dehli and C. Bolm, Adv. Synth.
Catal., 2005, 347, 19.
21 E. Baciocchi, T. Del Giacco, M. I. Ferrero, C. Rol and
G. V. Sebastiani, J. Org. Chem., 1997, 62, 4015; E. L. Clennan
and D. Aebisher, J. Org. Chem., 2002, 67, 1036; D. A. Panayotov,
D. K. Paul and J. T. Yates, Jr., J. Phys. Chem. B, 2003, 107, 10571;
S. Fujita, H. Sato, N. Kakegawa and A. Yamagishi, J. Phys. Chem. B,
2006, 110, 2533; J.-M. Zen, S.-L. Liou, A. S. Kumar and M.-S. Hsia,
Angew. Chem., Int. Ed., 2003, 42, 577.
Notes and references
1 K. N. Ferreira, T. M. Iverson, K. Maghlaoui, J. Barber and
S. Iwata, Science, 2004, 303, 1831; B. Loll, J. Kern, W. Saenger,
A. Zouni and J. Biesiadka, Nature, 2005, 438, 1040; J. Yano,
J. K. Sauer, M. J. Latimer, Y. Pushkar, J. Biesiadka, B. Loll,
c
This journal is The Royal Society of Chemistry 2011
Chem. Commun., 2011, 47, 8949–8951 8951

 Li, Fei
Li, Fei
 Yu, Miao
Yu, Miao
 Jiang, Yi
Jiang, Yi
 Huang, Fang
Huang, Fang
 Li, Yanqing
Li, Yanqing
 Zhang, Biaobiao
Zhang, Biaobiao
 Sun, Licheng
Sun, Licheng