A novel ‘build-bottle-around-ship’ method to encapsulate metalloporphyrins
in zeolite-Y. An efficient biomimetic catalyst
Bi-Zeng Zhan and Xiao-Yuan Li*
Department of Chemistry, The Hong Kong University of Science and Technology, Clear Water Bay, Kowloon, Hong Kong,
PR China
Electrostatic interaction is introduced between the host and
guest in the synthesis of faujasite-Y confined metallote-
MTMAnP4+ cations in the zeolitic supercages in such a readily
controllable manner is the electrostatic interaction between the
anionic aluminosilicate species and the cationic peripheral
substituents on the porphyrin macrocycle. The excellent
aqueous solubility of the cationic MP may contribute to the
enhanced loading concentration in the zeolite, but this factor
alone does not guarantee successful encapsulation. This asser-
tion is supported by the following experimental facts: (a) in
addition to MTMAnP4+, we have also successfully encapsu-
lated other metalloporphyrins with cationic peripheral substi-
tuents into zeolitic cages such as metallo–tetrakis(N-methyl-
trakis(N,N,N-trimethylanilinium)
porphyrin
cations
(MTMAnP4+); the synthesized composites display high
catalytic activity in the oxidation of cyclohexene.
One of the most attractive properties of zeolites are their well
organized nanopores and nanochannels which serve readily as
supporting hosts for various molecules. Encapsulation of
catalytically active transition metal complexes inside the
nanopores of zeolites, often referred to as ‘ship-in-a-bottle’
systems,1 has been believed to be one of the most promising
strategies in the development of viable industrial catalysts.1–6
Extensive effort has been devoted to the synthesis and catalytic
properties of zeolite entrapped metallocomplexes with such
ligands as bipyridine, salen, polyamines and phthalocya-
nines.1–6 In most of these syntheses, a so-called ‘assemble-ship-
inside-bottle’ approach has usually been adopted in which the
desired catalytic metallocomplex is synthesized inside the
nanopores/channels of the zeolite in the presence of an excess of
ligand or its synthetic precursors. As a consequence, what was
usually obtained is a mixture of the desired complex with
unreacted free ligand as well as side-products. For example, the
synthesis of metalloporphyrin (MP) inside zeolites often leads
to a significant portion of undesired polymerization product and
the unmetallated free base porphyrin ligand.7–9
We report here a novel, efficient and quantitative method to
synthesize high purity MPs incorporated in faujasite-Y at a
controllable loading concentration. The novel aspect of this
method is that an electrostatic interaction was introduced
between the host (anionic aluminosilicate species) and the guest
(cationic peripheral substituents on MPs) in a ‘build-bottle-
around-ship’ approach, namely to synthesize the nanocages of
zeolite around the high purity cationic MPs. The selected guest
molecules have excellent solubility in aluminosilicate gel. In a
typical synthesis, 240 mg of MTMAnPCl5 was added into an
aluminosilicate gel, freshly prepared by mixing silicate and
aluminate solutions containing 4.6 g of silica, 6.2 g of NaOH,
3.2 g of NaAlO2 and 80 ml of H2O. The gel was then
crystallized at 95 ± 2 °C under static and autogeneous
conditions in a stainless steel bomb (250 ml) for 48 h. After
cooling to room temp., a solid product was recovered by
filtration. The complexes adsorbed on the exterior surfaces were
removed by a thorough extraction with distilled water, metha-
nol, pyridine (2%)–methanol, and methanol again, respectively.
The removed complexes can be fully recovered for the next
round of synthesis. The crystals were then dried at 60 °C for
24 h. Thermogravimetry (TG) of the composite indicates that
1.51 mass% is attributable to the confined guest, corresponding
to ca. one MP complex for every forty supercages or a
supercage occupancy of 2.5%.
4-pyridyl)porphyrins (M
=
transition metal ion); (b)
metalloporphyrins with anionic peripheral substituents, such as
metallo–tetra(4-sulfonatophenyl)porphyrin (MTPPS42), could
not be entrapped into the supercages of zeolite by this method;
and (c) metalloporphyrins with neutral peripheral substituents,
such as metallo–tetraphenylporphyrin (MTPP), could only be
incorporated into the zeolite in trace amounts independent of the
initial concentration of the porphyrin in the gel. On the other
hand, it is believed that the first step in the construction of the
zeolitic framework is the formation of anionic aluminosilicate
species.
X-Ray powder diffraction (XRD) patterns of the unloaded
zeolite and zeolite entrapped FeTMAnP4+ and MnTMAnP4+ are
in excellent agreement with the calculated simulation of the
XRD pattern for faujasite zeolite.10 The excellent signal to noise
ratio in the XRD indicates that our synthesized faujasite crystals
are of high quality. X-Ray induced fluorescence (XIF) analysis
indicates that all the samples have a Si/Al ratio of ca. 1.6. This
suggests that the framework around the guest molecule
MTMAnP4+ is faujasite-Y (denoted MTMAnP@NaY)
(M = FeIII, MnIII etc.). The encapsulation of the cationic MP
inside the zeolite nanopores is also supported by TG, UV–VIS
DRS, surface area measurements, and resonance microRaman
spectroscopy (mRR, vide infra). This observation suggests that
the occluded MTMAnP4+ cations fit well into the supercages of
faujasite-Y. In view of the 13 Å diameter of supercage of
faujasite, ca. 18 Å MTMAnP4+ cation [across two opposite
trimethylanilinium (TMAn) substituents assuming a planar
macrocycle] must distort itself somewhat in order to fit into a
supercage. The most plausible distortion is that each of the four
peripheral TMAn groups swings toward one of the four channel
windows (ca. 7 Å in diameter) tetrahedrally located in a
supercage, leading to an overall ruffling of the macrocycle, a
distortion commonly observed in the X-ray structures of similar
MPs.11
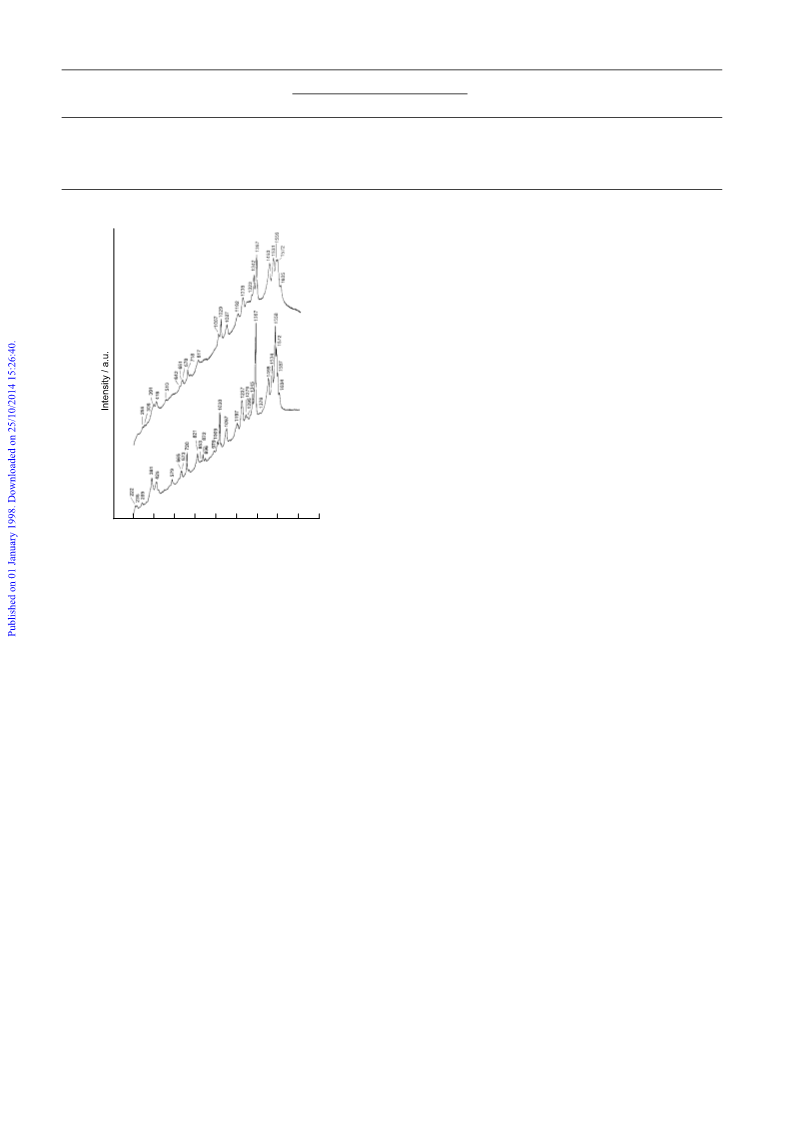
Resonance Raman techniques are very powerful in the study
of hemeproteins and zeolite confined molecules.12,13 Here, we
applied mRR to study the synthesized zeolite–porphyrin com-
posites. Laser excitation at 632.8 nm, in close resonance with
the Q0 absorption band of MTMAnPCl5, was used. Fig. 1 shows
the mRR spectra of MnTMAnPCl5 and its faujasite-Y confined
composite MnTMAnP@NaY. The normal modes of
MnTMAnPCl5 and other similar compounds have been thor-
oughly studied and assigned.13 Fig. 1 clearly shows that the
basic RR features of MnTMAnPCl5 are all retained in the
spectrum of MnTMAnP@NaY, indicating that neither decom-
The loading concentration of MTMAnP4+ is adjustable by
controlling its concentration in the aluminosilicate gel before
crystallization. The largest loading of ca. 5 mass% of the guest
molecule can be achieved if 800 mg of MTMAnPCl5 is added
to ca. 94 g of aluminosilicate gel. We speculate that the main
driving force for the very successful nanoinclusion of
Chem. Commun., 1998
349

 Zhan, Bi-Zeng
Zhan, Bi-Zeng
 Li, Xiao-Yuan
Li, Xiao-Yuan