126
Z.P. Pai et al. / Journal of Molecular Catalysis A: Chemical 332 (2010) 122–127
Table 2
Catalytic oxidation of organic substrates to mono- and dicarbonic acids.
No
Substrate (sub)
T,◦C
[Sub]/[Cat]
[Ox]/[Sub]
ꢃ, h
Acid
Yield, %
1
2
3
4
5
Cyclohexene
Cyclooctene
Octan-1-ol
Benzyl alcohol
Benzyl alcohol
92
90
93
90
90
200
200
500
500
500
4
3
2
2.5
3
3
4
3
Hexane-1,6-dioic (Adipic)
Octane-1,8-dioic (Cork)
n-Octanoic (Caprylic)
Benzoic
96–98
85–87
60–65
82–85
98–99
3
2.5a
Benzoic
a
H2O2 solution was dosed by three equal portions in 1 h each.
3.3. Testing of catalytic complex II in oxidation of organic
substrates
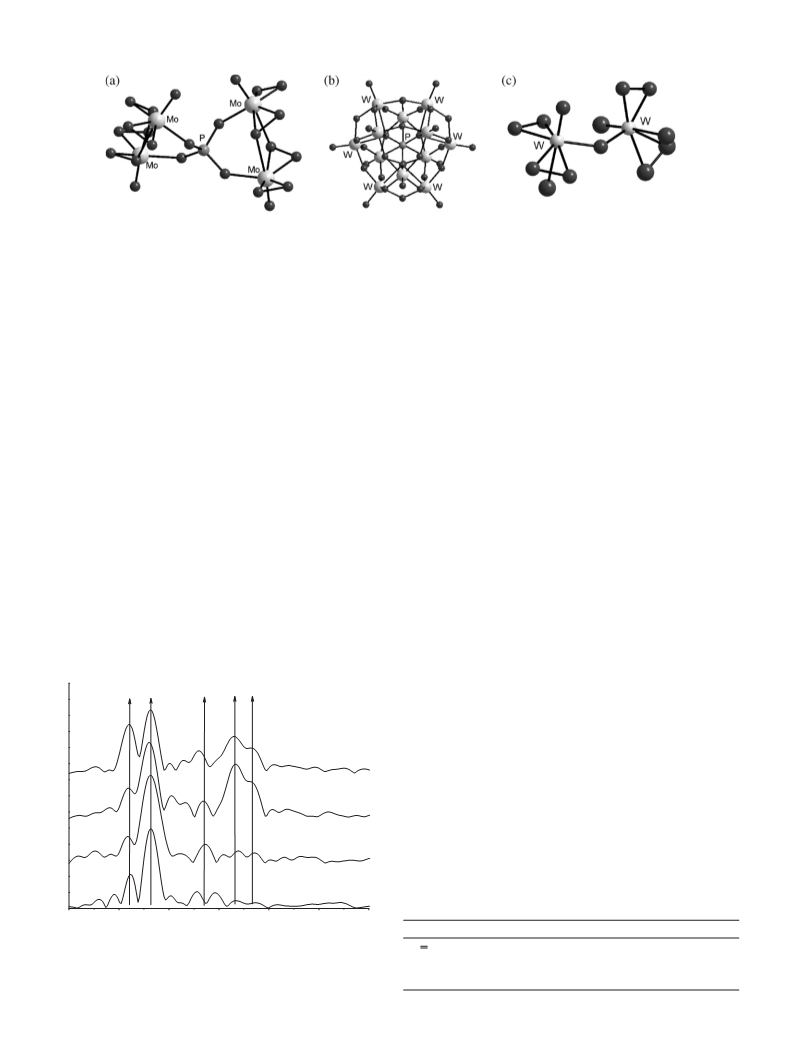
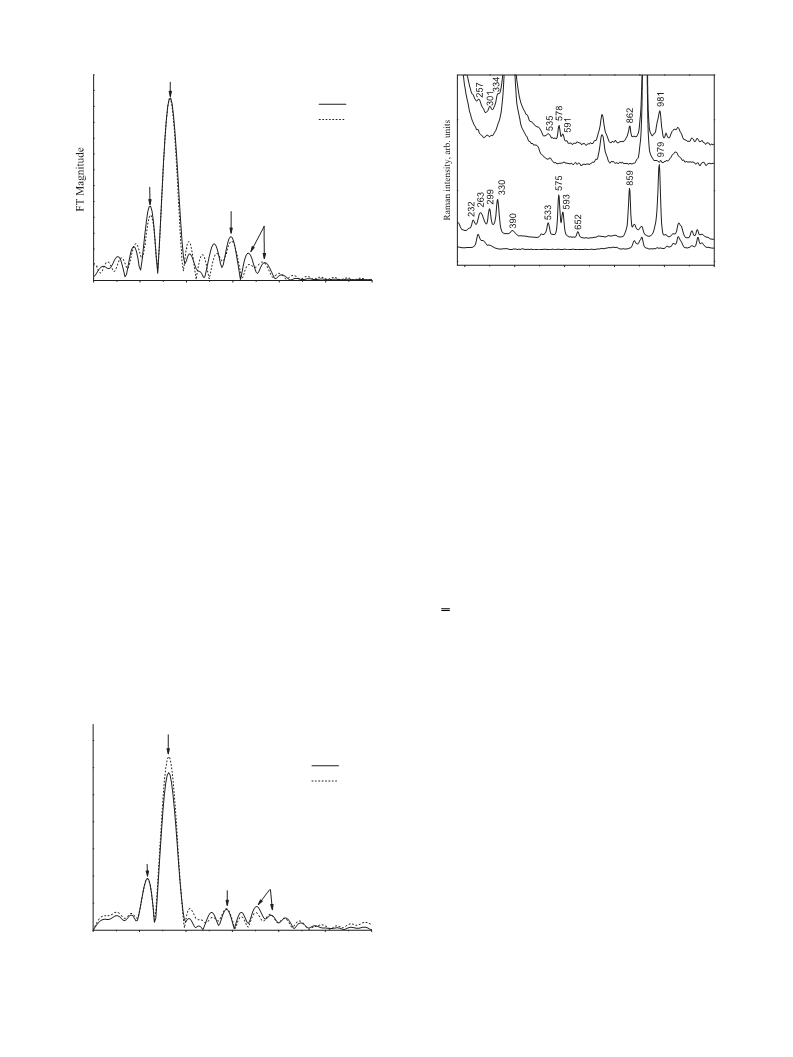
the local structure near to structure of Venturello’s complex [6a]
which is retained, when complex is dissolved in acetonitrile. This
complex has more rigid local structure as (n-Bu4N)2[HPW2O14
]
Complex II was tested for its catalytic activity in the oxidative
cleavage of C C bond in cyclic alkenes yielding the corresponding
dicarbonic acids, as well as in oxidation of alcohols yielding mono-
as oxidant (Ox). Reaction was performed in the two-phase system
(organic phase–aqueous phase) with no organic solvents. Testing
results are given in Table 2.
According to Table 2 at oxidation of cyclic alkenes in the pres-
ence of complex II the high yields of mono- and dicarbonic acids
may be attained at temperatures 90–95 ◦C. During cyclohexene
yield attains 98% (Table 2, example 1), which is essentially higher
than that given in [7b] (70% in 24 h), when catalyst was pre-
pared in situ. Similar result was obtained at cyclooctene oxidation
(Table 2, example 2), while in the presence of in situ prepared cat-
alyst cork acid yield never exceeded 24% [7b]. This fact indirectly
confirms the idea that, when catalyst is prepared in situ there is
not only active complex II but also a less active tungsten com-
plex, containing a binuclear anion [W2O3(O2)4(H2O)2]2−, which we
found in the mother liquor (sample 4). We may also suggest that
complex containing anion [W2O3(O2)4(H2O)2]2− is easier to deac-
tivate than complex II, which contains the stabilizing phosphate
group.
have the excess of oxidant over stoichiometry. Example 3 in Table 2
shows that when oxidant to substrate ratio is close to reaction sto-
ichiometry, product yield is low. Oxidant excess (see examples 1,
2, 4; Table 2) allows essentially higher product yields. We have
already discussed this fact in our paper [12], and came to conclu-
sion that it is possible to decrease the oxidant consumption using
method suggested in [21]. Hydrogen peroxide addition portion by
portion appears to be quite effective. In this case we can minimize
the peroxide decomposition, and thus ratio [Ox]/[Sub] is close to
stoichiometry, which is confirmed by example 5 in Table 2, showing
a 98–99% yield of benzoic acid.
[9a].
Synthesized catalytic complex [Bun4N]3{PO4[WO(O2)2]4} was
tested in the oxidation of cyclic alkenes and alcohols by hydro-
gen peroxide in two-phase systems (organic phase–aqueous phase)
with no additional organic solvents. Apparently individual catalytic
complex [Bun4N]3{PO4[WO(O2)2]4} provides the higher yields of
mono- and dicarbonic acids than in situ synthesized catalyst.
The next parts of this series of publications will be dedicated
to changes of the local structure of catalysts depending on solvent
polarity and in stoichiometric reactions.
Acknowledgements
The work was supported by Russian Foundation for Basic
Research, Project 09-03-00395, and Department of Chemistry and
Sciences on New Materials RAS, projects 5.7.3.
References
[1] M.M. Godneva, D.L. Motov, R.F. Okhrimenko, V.Ya. Kuznetsov, Zh. Neorg. Khim.
39 (1994) 740–742.
[2] J. Gao, Y. Chen, B. Han, Z. Feng, C. Li, N. Zhou, S. Gao, Z. Xi, J. Mol. Catal. A: Chem.
210 (2004) 197–204.
[3] (a) V.V. Kanazhevskii, V.P. Shmachkova, N.S. Kotsarenko, V.N. Kolomiichuk, D.I.
Kochubei, J. Struct. Chem. 47 (2006) 860–868;
(b) O.V. Klimov, A.V. Pashigreva, D.I. Kochubei, G.A. Bukhtiyarova, A.S. Noskov,
Dokl. Phys. Chem. 424 (2009) 497–501.
[4] G.B. Payne, P.H. Williams, J. Org. Chem. 24 (1959) 54–55.
[5] J.-M. Bregeault, M. Vennat, L. Salles, J.-Y. Piquemal, Y. Mahha, E. Briot, P.C.
Bakala, A. Atlamsani, R. Thouvenot, J. Mol. Catal. A: Chem. 250 (2006) 177–189.
[6] (a) C. Venturello, E. Alneri, M. Ricci, J. Org. Chem. 48 (1983) 3831–3833;
(b) C. Venturello, R. D’Aloisio, J.C.J. Bart, M. Ricci, J. Mol. Catal. A: Chem. 32
(1985) 107–110;
(c) C. Venturello, M. Ricci, J. Org. Chem. 51 (1986) 1599–1602.
[7] (a) Y. Matoba, H. Inoue, J. Akagi, T. Okabayashi, Y. Ishii, M. Ogawa, Synth. Com-
mun. 14 (1984) 865–873;
(b) Y. Ishii, K. Yamawaki, T. Ura, H. Yamada, T. Yoshida, M. Ogawa, J. Org. Chem.
53 (1988) 3587–3593;
(c) S. Sakaguchi, Y. Nishiyama, Y. Ishii, J. Org. Chem. 61 (1996) 5307–5311.
[8] R. Noyori, M. Aoki, K. Sato, Chem. Commun. (Cambridge, UK) 16 (2003)
1977–1988.
[9] (a) L. Salles, C. Aubry, R. Thouvenot, F. Robert, C. Doremieux-Morin, G. Chottard,
H. Ledon, Y. Jeannin, J.-M. Bregeault, Inorg. Chem. 33 (1994) 871–878;
(b) Y. Mahha, L. Salles, J.-Y. Piquemal, E. Briot, A. Atlamsani, J.-M. Bregeault, J.
Catal. 249 (2007) 338–348;
(c) L. Salles, R. Thouvenot, J.-M. Bregeault, Dalton Trans. (2004) 904–907.
[10] L. Salles, C. Aubry, R. Touvenot, F. Robert, G. Chottard, R. Thouvenot, H. Ledon,
J.M. Bregeault, New J. Chem. 17 (1993) 367–375.
4. Conclusion
Our research in catalysis with metal peroxocomplexes using
EXAFS and Raman spectroscopy revealed the structural features
of polyoxo- and peroxopolyoxocomplexes of tungsten, forming as
phosphorus tungsten HPA (H3PW12O40) in aqueous solution inter-
acts with the 30%-aqueous solution of H2O2. This interaction is
shown to reduce the nuclearity of precursor, which initially has
structure W12. The symmetry of oxygen environment of tungsten
is found to change, distances W–W remaining the same. After com-
plex [Bun4N]3{PO4[WO(O2)2]4} is isolated, binuclear anion stays in
[11] J.F. Keggin, Proc. R. Soc. A 144 (1934) 75–100.
[12] F.W.B. Einstein, B.R. Penfold, Acta Cryst. 17 (1964) 1127–1133.
[13] Z.P. Pai, A.G. Tolstikov, P.V. Berdnikova, G.N. Kustova, T.B. Khlebnikova, N.V. Seli-
vanova, A.B. Shangina, V.G. Kostrovskii, Russ. Chem. Bull. 54 (2005) 1847–1854.
[14] (a) Z.P. Pai, T.B. Khlebnikova, Y.V. Mattsat, V.N. Parmon, React. Kinet. Catal. Lett.
98 (2009) 1–8;
(b) Z.P. Khlebnikova, L.A. Pai, Y.V. Fedoseeva, T.B. Mattsat, React. Kinet. Catal.
Lett. 98 (2009) 9–17.
[15] D.I. Kochubey, EXAFS Spectroscopy of Catalysts, Nauka, Novosibirsk, 1992.
[16] K.V. Klementev, Nucl. Instrum. Methods Phys. Res., Sect. A 448 (2000) 299–
301.
the aqueous phase [W2O3(O2)4(H2O)2]2−
.
EXAFS combined with Raman spectroscopy used to study local
structure of the isolated complex in its solid state shows that it most
likely has four-nuclear structure [Bun4N]3{PO4[WO(O2)2]4}, with
[17] J.J. Rehr, A.L. Ankudinov, Radiat. Phys. Chem. 70 (2004) 453–463.

 Pai, Zinaida P.
Pai, Zinaida P.
 Kochubey, Dimitry I.
Kochubey, Dimitry I.
 Berdnikova, Polina V.
Berdnikova, Polina V.
 Kanazhevskiy, Vladislav V.
Kanazhevskiy, Vladislav V.
 Prikhod'Ko, Irina Yu.
Prikhod'Ko, Irina Yu.
 Chesalov, Yuriy A.
Chesalov, Yuriy A.