1530
C.-H. Li et al. / Electrochimica Acta 56 (2011) 1529–1534
2. Experimental
2.1. Chemicals
the electrolysis, the electrolyte was cooled to room temperature,
accompanied by the precipitation of a large amount of adipic acid.
After filtered, the crude adipic acid was obtained and recrystal-
lized twice with ethanol–petroleum ether, then dried at 60 ◦C in
a vacuum oven. In addition, mother liquor can be recycled.
N,N-Dimethylformamide (DMF) (Alfa Aesar, AR) was kept over
anhydrous MgSO4 for several days. Tetrabutyl ammonium bromide
(n-Bu4NBr) (Alfa Aesar, AR) was dried at 60 ◦C under vacuum for
10 h. Other dienes (AR) were all purchased from Alfa Aesar and were
used without further purification. 1,3-butadiene was purchased
from Guangzhou Petrochemicals Co. Ltd.
2.5. Characterizations of products
(Z)-3-hexene-1,6-dioic acid: IR (neat): ꢀ: 3480 (OH), 1700
(C O) cm−1. 1H NMR (DMSO-d6, 400 MHz): ı 12.2 (s, 2H), 5.62–5.64
(m, 2H), 3.02 (d, J = 4.4 Hz, 4H). 13C NMR (DMSO-d6, 100 MHz): ı
172.9, 125.1, 33.2. MS (EI): m/z 144 (M+).
(E)-3-hexene-1,6-dioic acid: IR (neat): ꢀ: 3450 (OH), 1700
(C O) cm−1. 1H NMR (DMSO-d6, 400 MHz): ı 12.2 (s, 2H), 5.57–5.59
(m, 2H), 2.99 (d, J = 13.4 Hz, 4H). 13C NMR (DMSO-d6, 100 MHz): ı
173.2, 126.5, 37.9. MS (EI): m/z 144 (M+).
(E)-3-methyl-3-hexene-1,6-dioic acid: IR (neat): ꢀ: 3426 (OH),
1690 (C O) cm−1. 1H NMR (DMSO-d6, 400 MHz): ı 12.2 (s, 2H), 5.39
(t, J = 8 Hz, 1H), 2.99 (d, J = 8 Hz, 2H), 2.95 (s, 2H), 1.63 (s, 3H). 13C
NMR (DMSO-d6, 100 MHz): ı 173.0, 172.7, 131.8, 120.8, 44.5, 33.5,
16.6. MS (EI): m/z 158 (M+).
(E)-3,4-dimethyl-3-hexene-1,6-dioic acid: IR (neat): ꢀ: 3424 (OH),
1695 (C O) cm−1. 1H NMR (DMSO-d6, 400 MHz): ı 12.1 (s, 2H), 2.29
(s, 4H), 1.68 (s, 6H). 13C NMR (DMSO-d6, 100 MHz): ı 172.7, 125.4,
41.2, 19.3. MS (EI): m/z 172 (M+).
2.2. Instrumentation
The electrolytic cell is the same as that reported previously [41].
FTIR spectra were measured by a TENSOR27 spectrometer. 1H NMR
and 13C NMR were determined on a Bruker DRX-400 spectrome-
ter with DMSO-d6 as the solvent in the presence of SiMe4 as an
internal standard. Mass spectral analyses were done on a Shimadzu
QP5050A spectrometer.
Prior to the cyclic voltammetry (CV) experiment, the electrodes
were cleaned by diluted hydrochloric acid and carefully washed
with distilled water, and finally dried. The working electrode and
counter-electrode (Al) were built with the same size (2 mm × 2 cm,
Alfa Aesar, 99.999%). CV experiments were carried out at an Auto
LAB (PGSTAT 30) electrochemical workstation, with Ni or Ag as a
working electrode and a saturated calomel electrode (SCE) as a ref-
erence electrode. The solution was saturated with CO2 (0.1 MPa)
during the CV experiments.
(E)-2,5-diphenyl-3-hexene-1,6-dioic acid: IR (neat): ꢀ: 3409 (OH),
1696 (C O) cm−1 1H NMR (DMSO-d6, 400 MHz): 12.1 (s, 2H),
.
7.21–7.33 (m, 10H), 5.93 (dd, J1 = 2.4 Hz, J2 = 5.2 Hz, 2H), 4.33 (dd,
J1 = 2.4 Hz, J2 = 5.2 Hz, 2H).13C NMR (DMSO-d6, 100 MHz): ı 173.7,
139.5, 130.9, 129.0, 128.2, 127.4, 54.2. MS (EI): m/z 296 (M+).
2-cyclohexene-1,4-dicarboxylic acid: IR (neat): ꢀ: 3430 (OH),
2.3. The electrocarboxylation of 1,3-butadiene with carbon
dioxide
Before the electrosynthesis, theanodeand cathodewerecleaned
with detergent and diluted hydrochloric acid, followed by wash-
ing with distilled water and then dried. The electrosynthesis was
carried out in the high-pressure stainless-steel undivided cell fit-
ted with a nickel sheet cathode (wet surface area 6 cm2) and an
aluminum sheet anode (wet surface area 6 cm2). In a typical exper-
imental procedure, dried DMF (N,N-dimethylformamide) solvent
(35 mL) and n-Bu4NBr (2.5 mmol) were added to the cell, respec-
tively. After the electrochemical cell was sealed, 1,3-butadiene (gas)
was charged to reach the saturated state. The accurate quantity of
1,3-butadiene was determined according to its amount dissolved in
DMF solvent after superfluous 1,3-butadiene was discharged from
the cell. Then, CO2 was charged into the cell to the desired pressure.
The electrosynthesis was carried out under continuously stirring at
room temperature until electricity 4 F mol−1 of starting substrates
had been passed through the cell. After the electrolyte was distilled
at reduced pressure, the residue was acidified with hydrochloric
acid (2 mol L−1), then continuously stirred for 6 h, and extracted
with diethyl ether (3 × 25 mL). The ether phase was washed twice
with distilled water and dried with anhydrous MgSO4. After evap-
oration of ether, the obtained yellow crude product was dried in a
vacuum oven at 45 ◦C for 5 h, and recrystallized twice with ethyl
acetate–petroleum ether, and dried at 60 ◦C in a vacuum oven.
1700 (C O) cm−1 1H NMR (DMSO-d6, 400 MHz): ı 12.5 (s, 2H),
.
5.76–5.82 (m, 2H), 3.04–3.42 (m, 2H), 1.89–1.95 (m, 2H), 1.62–1.70
(m, 2H). 13C NMR (DMSO-d6, 100 MHz): ı 174.8, 126.9, 40.2, 23.8.
MS (EI): m/z 170 (M+).
3. Results and discussion
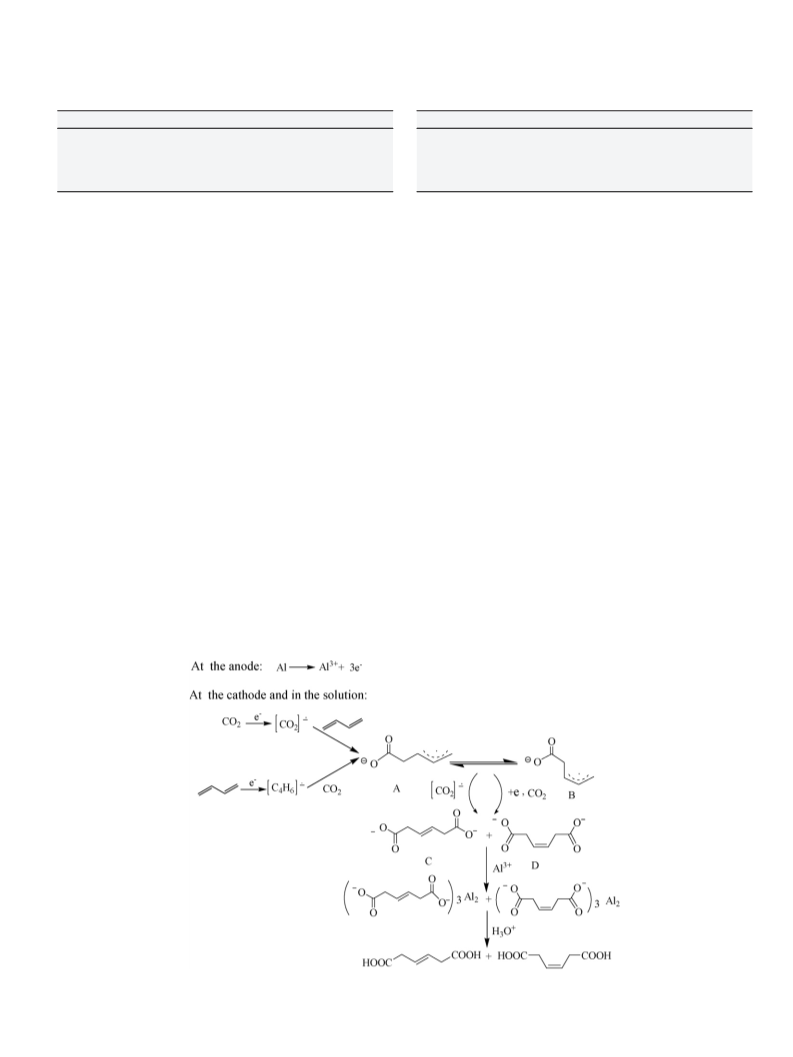
3.1. Electrosynthesis of 3-ene-1,6-dioic acids from dienes and CO2
Bu4NBr-DMF as electrolyte, the electricity 4 F mol−1 of starting
for 1,3-butadiene, the configuration of the product corresponding
to linear dienes is only trans form (Table 1, entries 2–4). Cyclohexa-
diene could be also converted into the corresponding dicarboxylic
acids in good yield (70%, Table 1, entry 5). In addition, very small
amount of polymers were found in our present experimental con-
ditions.
3.2. Influence of cathode materials
2.4. The electroreduction of 3-hexene-1,6-dioic acid into adipic
acid
1,3-Butadiene was selected as
a model molecule to sys-
temically investigate the influence of some key factors on the
electrosynthesis. Some electrochemical parameters such as sup-
porting electrolyte, temperature, substrate concentrations and
current density have been discussed in details previously [41,42],
which could provide some references for the present work. Here the
effect of different electrode materials was first investigated on the
electrocarboxylation of 1,3-butadiene with CO2. As known, elec-
trode materials are very important for electrochemical reactions.
At room temperature, the as-prepared 3-hexene-1,6-dioic acid
(20 mmol) and H2SO4 aqueous solution (1 mol L−1, 40 mL) were
added to the undivided cell (100 mL) fitted with Ni cathode (wet
surface area 4 cm2) and Pt anode (wet surface area 4 cm2). In a
typical experiment, the electroreduction was performed at 60 ◦C
under continuously stirring until electricity 3 F mol−1 of start-
ing substrates had been passed through the cell. At the end of

 Li, Chuan-Hua
Li, Chuan-Hua
 Yuan, Gao-Qing
Yuan, Gao-Qing
 Ji, Xiao-Chen
Ji, Xiao-Chen
 Wang, Xiu-Jun
Wang, Xiu-Jun
 Ye, Jian-Shan
Ye, Jian-Shan
 Jiang, Huan-Feng
Jiang, Huan-Feng