Paper
RSC Advances
faster than that for Fe2O3/Al2O3 reduction, also indicated that
the methane more easily diffusion through the product layer of
Fe2O3/Al2O3/TiO2 oxygen carriers. The activation energy for the
product layer diffusion reaction (Eapl) estimated for CLP by
employment of Fe2O3/Al2O3 and Fe2O3/Al2O3/TiO2 as oxygen
carrier are about 476 and 447 kJ molꢀ1 for oxygen carrier
reduction with methane, respectively. Approximately, methane
is completely combusted for experiments conducted in the
moving bed reactor with Fe2O3/Al2O3 and Fe2O3/Al2O3/TiO2
operated at Fe2O3/CH4 molar ratio reached about 5.4 and 4.4,
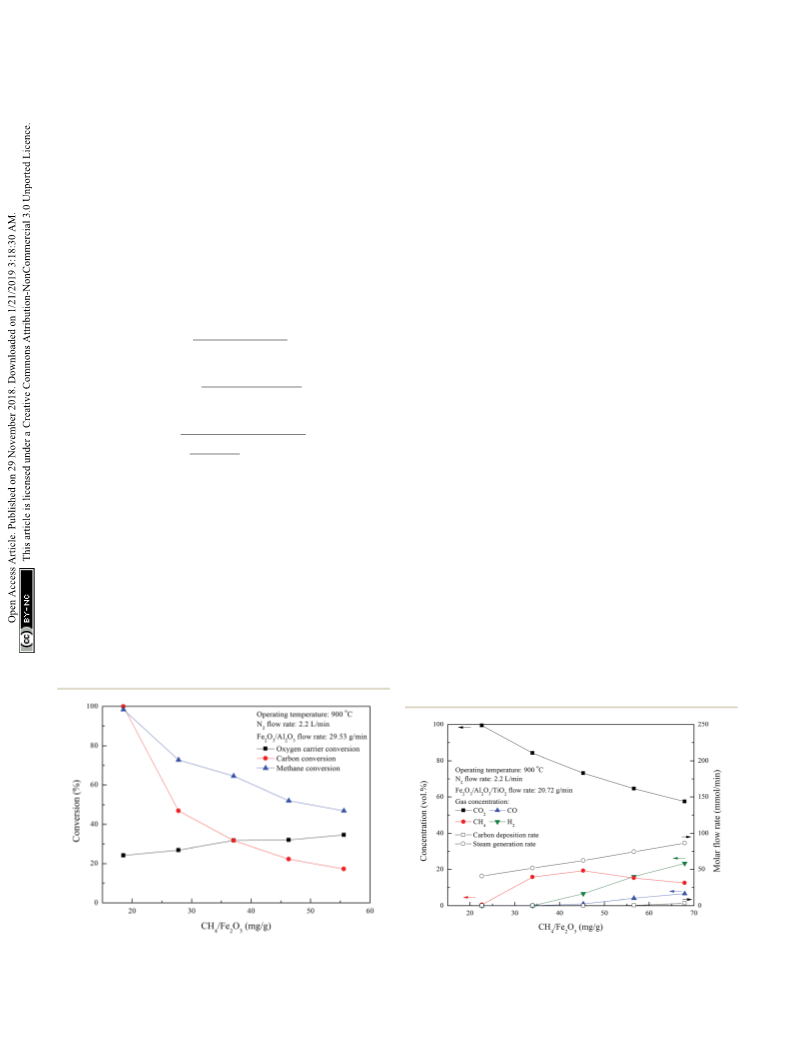
respectively; in addition, the oxygen carrier conversions are
respectively reached about 24.2% and 29.7%. Carbon deposi-
tion during methane combustion is noticeably avoided by using
Fe2O3/Al2O3/TiO2 as oxygen carrier. According to the XRD
characterization, Al2FeO4 is the major crystalline phase gener-
ated during CLC of methane with Fe2O3/Al2O3 oxygen carrier.
The formations of Fe3C and Fe were observed for experiment
conducted with CH4/Fe2O3 ratio of 67.9 mg gꢀ1 and Fe2O3/Al2O3
ow rate of 29.53 g minꢀ1, which would serve as catalyst for
methane decomposition. For Fe2O3/Al2O3/TiO2 oxygen carriers
sampled aer reduction, Fe, Fe3O4, Al2FeO4 and FeTiO3 were
identied by the XRD pattern, demonstrating Al2FeO4 and
3 U.S. Energy Information Administration (EIA), Annual Energy
Outlook 2016 with Projections to 2040, 2016.
4 L. S. Fan, Chemical Looping Systems for Fossil Energy
Conversions, John Wiley & Sons, Inc., New York, 2010.
´
5 J. Adanez, A. Abad, F. Garcia-Labiano, P. Gayan and L. F. de
Diego, Progress in Chemical-looping Combustion and
Reforming Technologies, Prog. Energy Combust. Sci., 2012,
38, 215–282.
´
´
´
6 J. Adanez, L. F. de Diego, F. Garcıa-Labiano, P. Gayan,
A. Abad and J. M. Palacios, Selection of Oxygen Carriers for
Chemical-looping Combustion, Energy Fuels, 2004, 18, 371–
377.
7 M. Johansson, T. Mattisson and A. Lyngfelt, Investigation of
Fe2O3 with MgAl2O4 for Chemical-looping Combustion, Ind.
Eng. Chem. Res., 2004, 43, 6978–6987.
8 Q. Zafar, T. Mattisson and B. Gevert, Redox Investigation of
Some Oxides of Transition-state Metals Ni, Cu, Fe, and
Supported on SiO2 and MgAl2O4, Energy Fuels, 2006, 20,
33–44.
9 B. M. Corbella and J. M. Palacios, Titania-supported Iron
Oxide as Oxygen Carrier for Chemical-looping Combustion
of Methane, Fuel, 2007, 86, 113–122.
FeTiO3 generated during reduction might become support 10 F. Li, S. Luo, Z. Sun, X. Bao and L. S. Fan, Role of Metal Oxide
materials as well as oxygen carriers in practical operation by
moving bed reactor for chemical looping process. Output pro-
cessing capacity estimated for CLC by employment of Fe2O3/
Support in Redox Reactions of Iron Oxide for Chemical
Looping Applications: Experiments and Density Functional
Theory Calculations, Energy Environ. Sci., 2011, 4, 3661–3667.
Al2O3 and Fe2O3/Al2O3/TiO2 as oxygen carrier are increased for 11 L. Qin, Z. Cheng, J. A. Fan, D. Kopechek, D. Xu,
experiments conducted with higher CH4/Fe2O3 ratio, and
reached 350 W and 578 W as methane conversion reached
around 64.6% and 72.7%, respectively. About 269 W and 270 W
are released for complete methane combustion with Fe2O3/
N. Deshpande and L. S. Fan, Nanostructure Formation
Mechanism and Ion Diffusion in Iron-titanium Composite
Materials with Chemical Looping Redox Reactions, J.
Mater. Chem. A, 2015, 3, 11302–11312.
Al2O3 and Fe2O3/Al2O3/TiO2 oxygen carriers in the ADMBR, 12 Y. Zhao and F. Shadman, Kinetics and Mechanism of
indicating that roughly 98% of Qin released for methane
combustion using Fe2O3/Al2O3 and Fe2O3/Al2O3/TiO2 oxygen
carriers in the ADMBR.
Ilmenite Reduction with Carbon Monoxide, AIChE J, 1990,
36, 1433–1438.
13 P. C. Chiu, Y. Ku, Y. L. Wu, H. C. Wu, Y. L. Kuo and
Y. H. Tseng, Characterization and Evaluation of Prepared
Fe2O3/Al2O3 Oxygen Carriers for Chemical Looping
Process, Aerosol Air Qual. Res., 2014, 14, 981–990.
14 M. C. Lo, Composite Fe-Ti Based Oxygen Carrier for Chemical
Looping Combustion and Hydrogen Generation, Master
Thesis in Department of Chemical Engineering, National
Taiwan University of Science and Technology, Taipei City,
Taiwan, 2014.
Conflicts of interest
There are no conicts to declare.
Acknowledgements
This research was supported by Grant MOST 106-3113-E-007-
002-from the National Science and Technology Program-
Energy, Taiwan, and by Grant MOST 105-2622-E-011-019-CC2
and MOST 103-2221-E-011-002-MY3 from the Ministry of
Science and Technology, Taiwan. The authors appreciated
China Steel Corp. for providing hematite powders for the
preparation of oxygen carriers.
¨
15 P. Markstrom and A. Lyngfelt, Designing and Operating
Cold-ow Model of 100 kW Chemical-looping
a
a
Combustor, Powder Technol., 2012, 222, 182–192.
16 Y. De Vos, M. Jacobs, P. Van Der Voort, I. Van Driessche,
F. Snijkers and A. Verberckmoes, Optimization of Spray
Dried Attrition-resistant Iron Based Oxygen Carriers for
Chemical Looping Reforming, Chem. Eng. J., 2017, 309,
824–839.
17 R. W. Breault, C. S. Yarrington and J. M. Weber, The Effect of
Thermal Treatment of Hematite Ore for Chemical Looping
Combustion of Methane, J. Energy Resour. Technol., 2016,
138, 0422021–0422028.
References
1 World Energy Council (WEC), World Energy Resources –
Unconventional Gas, a Global Phenomenon, 2016.
2 U.S. Energy Information Administration (EIA), Annual Energy 18 F. Li, L. Zeng, L. G. Velazquez-Vargas, Z. Yoscovits and
Outlook 2014 with Projections to 2040, 2014.
L. S. Fan, Syngas Chemical Looping Gasication Process:
This journal is © The Royal Society of Chemistry 2018
RSC Adv., 2018, 8, 39902–39912 | 39911

 Wu, Hsuan-Chih
Wu, Hsuan-Chih
 Ku, Young
Ku, Young