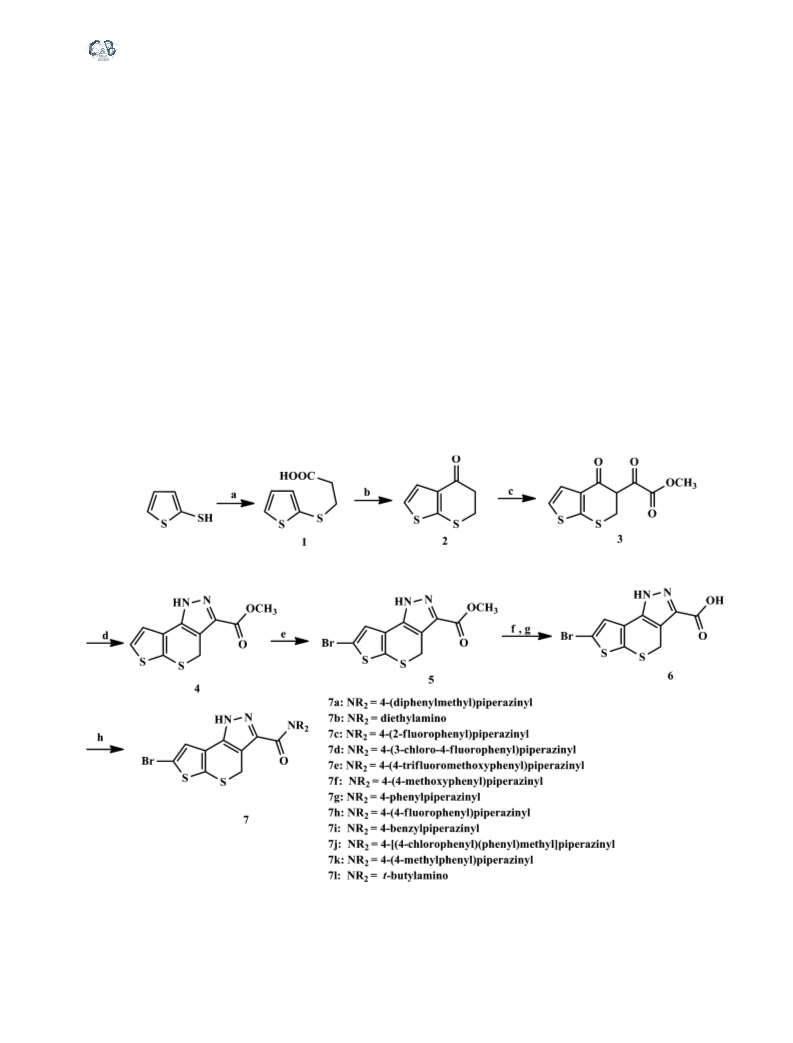
Hu et al.
General synthetic procedure for 5,6-dihydro-4H-
thieno[2,3-b]thiopyran-4-one (2)
of 5, which is a white solid. MP: 182–184 °C; 1H-NMR
(600 MHz, DMSO-d6): d 3.86 (s, 3H), 4.38 (s, 2H), 7.47 (s,
1H).
Under N2 in a three-neck was placed 1 (3.76 g, 0.02 mol),
CH2Cl2 (20 mL) and two drops of DMF. To the solution
was added dropwise oxalyl chloride (2.81 g, 0.022 mol) at
ambient temperature. After 1 h, the solution was cooled to
ꢁ10 °C, and a solution of SnCl4 (2.57 g, 0.01 mol) in
CH2Cl2 (10 mL) was added dropwise. The mixture was
then stirred at 0 °C, and after 0.5 h, H2O (20 mL) was
added. The mixture was separated, and the organic
extracts were washed with saturated Na2CO3-H2O and
brine, dried, filtered, and concentrated to dryness (13).
The residue was recrystallized from petroleum ether, and
the solid was filtered to yield 2.59 g (76.2%) of 2, which is
a white solid. MP: 59–60 °C (reported 65 °C in 14); 1H-
NMR (300 MHz, DMSO-d6): d2.77 (t, 2H), 3.47 (t, 2H),
7.35 (d, 1H, J = 5.4 Hz), 7.38 (d, 1H, J = 5.4 Hz).
General synthetic procedure for 7-bromo-1,4-
dihydrothieno[30,20:5,6]thiopyrano[4,3-c]pyrazole-
3-carboxylic acid (6)
To a solution of 5 (0.24 g, 1 mmol) in 30 mL water, NaOH
(0.1 g, 2.5 mmol) was added. The mixture was heated at
reflux for 2 h and then cooled to room temperature. Sub-
sequently, the solution was acidified with 1 N HCl until no
precipitation appeared. The mixture was filtered and dried
to give 0.30 g (95%) of 6, which is a white solid. MP:
255–257 °C. 1H-NMR (600 MHz, DMSO-d6): d 4.35 (s,
2H), 7.46 (s, 1H), 13.84 (s, 1H).
General synthetic procedure for 7-bromo-1,4-
dihydrothieno[30,20:5,6]thiopyrano[4,3-c]pyrazole-
3-carboxamides (7)
General synthetic procedure for methyl oxo(4-oxo-
5,6-dihydro-4H-thieno[2,3-b]thiopyran-5-yl)acetate
(3)
A mixture of 6 (0.32 g, 1 mmol), amine (1.1 mmol), 1-(3-
Sodium (0.46 g) was dissolved in 20 mL of absolutely dry
methanol which was removed by distillation. In the
meantime, compound 2 (1.70 g, 0.01 mol) and dimethyl
oxalate (2.36 g, 0.02 mol) were dissolved in 20 mL toluene
and this solution was added to the sodium methoxide. The
mixture was stirred for 24 h at room temperature and then
poured into 100 mL water. The mixture was separated,
and the organic phase was extracted with 50 mL of 10%
NaOH. Then, the water phase was washed with 50 mL of
diethyl ether and acidified with 1 N HCl until no precipitation
appeared. The mixture was filtered and dried to give 1.5 g
(59.0%) of 3, which is a yellow solid. MP: 93–95 °C (11,15);
1H-NMR (600 MHz, DMSO-d6): d 3.76 (s, 3H), 4.15 (s, 2H),
7.35 (d, 1H, J = 4.8 Hz), 7.41 (d, 1H, J = 4.8 Hz).
dimethylaminopropyl)-3-ethylcarbodiimide
hydrochloride
(0.24 g, 1.25 mmol), 1-hydroxybenzotriazole (0.05 g,
0.37 mmol), 1 mL of Et3N and absolutely dry CH2Cl2
(20 mL) was stirred for 24 h at room temperature. The
mixture was filtered, and the filtrate was washed with
20 mL of 1 N HCl for twice, saturated Na2CO3, H2O, and
brine; then it was dried, filtered, and concentrated to dry-
ness. The residue was purified by column chromatogra-
phy on silica with chloroform and methanol [V (chloroform):
V (methanol) = 15:1] as the eluent to give 7 as a white
solid.
Evaluating the enzyme activity of 5-LOX
The enzyme activity of 5-LOX was determined fluores-
cence spectrophotometrically by oxidation of the substrate
H2DCFDA to the highly fluorescent 20, 70-dichlorofluoresce-
in product during 5-LOX’s catalytic reaction (16). The
reactions were initiated by the addition of AA as substrate
and then monitored by excitation at 500 nm and emission
at 520 nm utilizing a multiwall fluorometer (Synergy4;
Biotek, Winooski, VT, USA). Fluorescence signals were
recorded for 5 min with a kinetics mode program. Inhibition
activities were measured as described previously (17). All
inhibition values were tested at the concentration of 91 lM.
Zileuton (reported IC50 = 0.9 lM in 18) was used as posi-
tive control.
General synthetic procedure for methyl 1,4-
dihydrothieno[30,20:5,6]thiopyrano[4,3-c]pyrazole-
3-carboxylate (4)
A mixture of 3 (0.51 g, 2.0 mmol), 80% hydrazine hydrate
(2 mL, 0.032 mol) and 10 mL of HOAc was heated at
reflux for 12 h. Then, the solution was poured into 50 mL
water and filtered to give 0.32 g (63.2%) of 4, which is a
white solid. MP: 94–96 °C (11,15); 1H-NMR (300 MHz,
CDCl3): d3.97 (s, 3H), 4.33 (s, 2H), 7.18 (d, 1H,
J = 5.3 Hz), 7.40 (d, 1H, J = 5.3 Hz).
General synthetic procedure for methyl 7-bromo-
1,4-dihydrothieno[30,20:5,6]thiopyrano[4,3-c]
pyrazole-3-carboxylate (5)
To a solution of 4 (0.24 g, 1 mmol) in 10 mL glacial acetic
acid, Br2 (0.32 g, 2 mmol) was added at ice bath within
30 min. The mixture was stirred at room temperature for
12 h. Subsequently, the solution was poured into 50 mL
water, and the solid was filtered to yield 26.4 mg (80.0%)
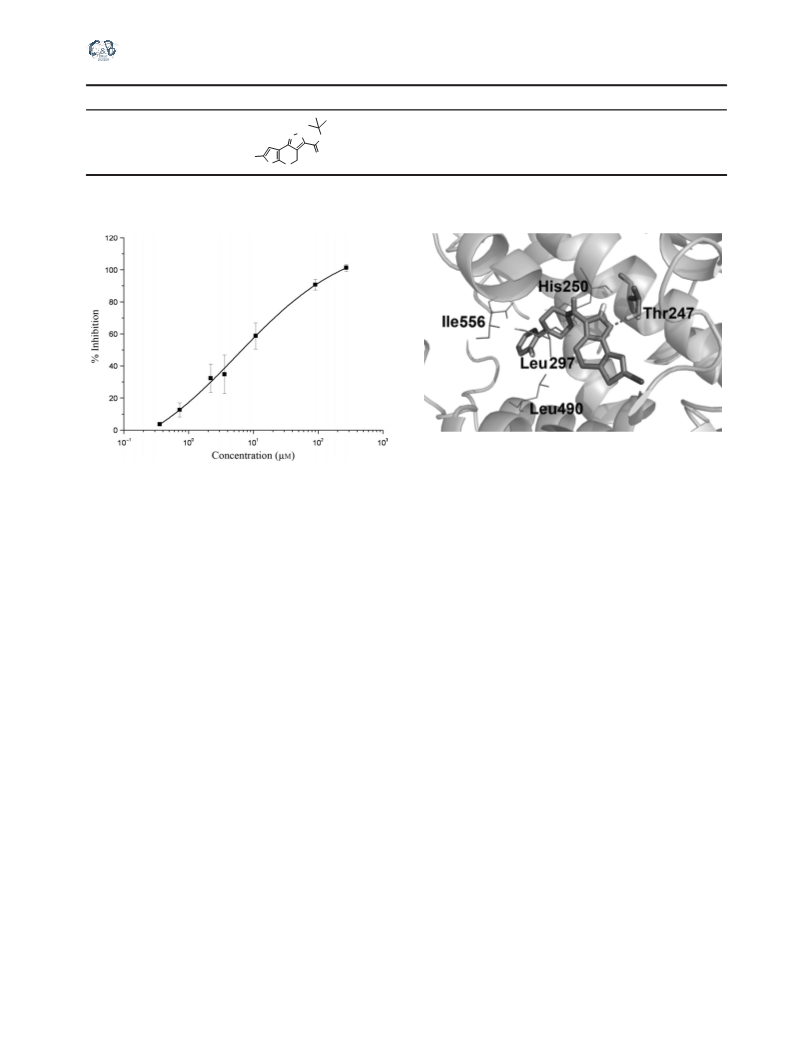
Inspecting the selectivity of compound 7c
The rates of 5-LOX, 12-LOX, and 15-LOX were deter-
mined (19) by following the formation of product at
235 nm (e = 25 000 M/cm). The enzyme was added into
quartz 96-well microtiter plates in sodium phosphate buffer
(100 mM, pH 7.4) and incubated with a compound (dis-
solved in DMSO) at room temperature for 1 min. The reac-
2
Chem Biol Drug Des 2014

 Hu, Jianshu
Hu, Jianshu
 Zhu, Wei
Zhu, Wei
 Meng, Hu
Meng, Hu
 Liu, Ying
Liu, Ying
 Wang, Xin
Wang, Xin
 Hu, Chun
Hu, Chun