Vol. 28, No. 10 (2016)
Chemical Transformations and Biological Studies of Terpenoids Isolated from Essential Oil of C. scariosus 2155
petri-dish. The experiment was replicated thrice. The petri-
dishes were incubated at 20 ± 2 °C. Petri-dishes were observed
daily for 10 days for germination (radical protrusion of 2 mm).
Primary root length and shoot length were measured at the
end of ten days with a centimeter scale.
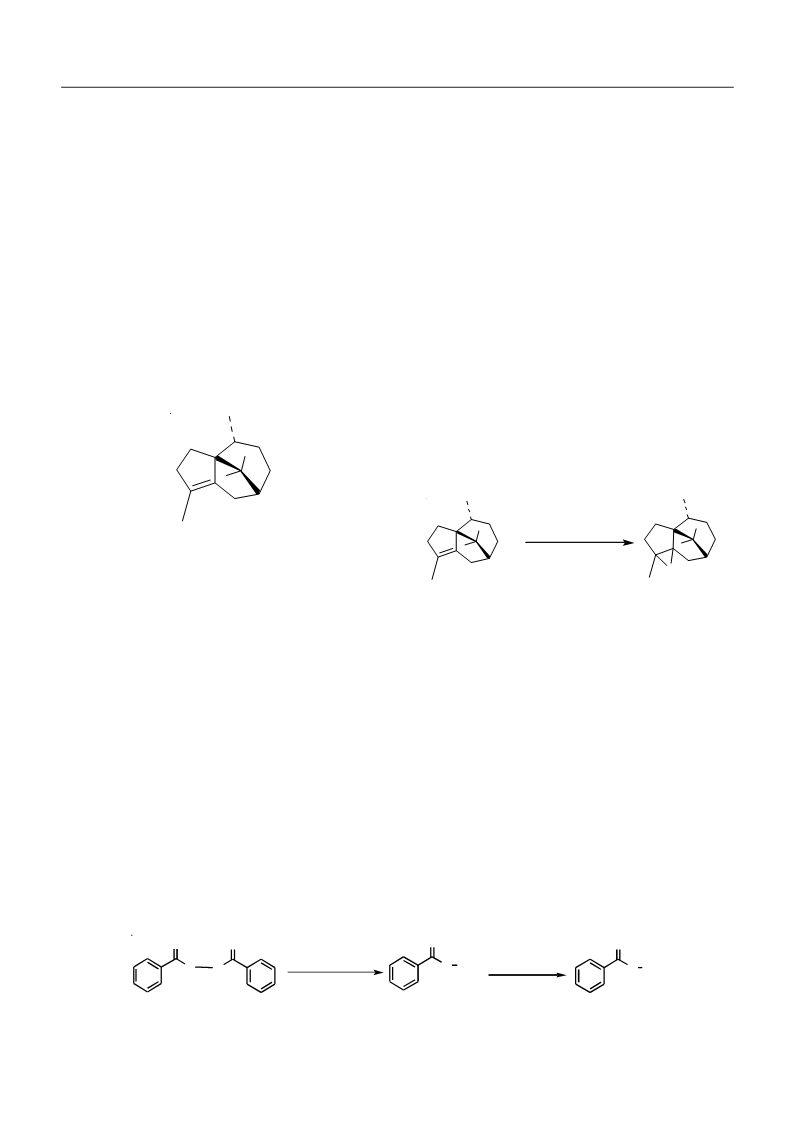
materials in the synthesis of commercially important materials
for enhancing fragrance and flavor. Cyprene epoxide (5) was
obtained from cyprene (1) by two different methods (Scheme-
II). In first method, cyprene (1) was treated with chloroform
solution of perbenzoic acid (4) taken in excess with continuous
stirring. In another method, cyprene (1) was refluxed with
epichlorohydrin for 13 h. In both cases, cyprene epoxide (5)
was obtained as a thick yellow liquid. The purity of the
compound was checked by thin layer chromatography
At the end of 10 days, seedlings were blotted dry and
fresh weight was recorded. For dry weight determination, the
seedlings were oven dried at 60 °C for 3 days in an incubator.
Preparation of stock solution: The stock solution (2000
µg/mL) of oil, its polar and non-polar fractions, cyprene (1)
and cyprene epoxide (5) was prepared by dissolving each
chemical (20 mg) in 1 mL of Tween 20 (polyoxyethylene
sorbitan) and volume was made 10 mL with distilled water.
The stock solution of 2000 µg/mL of each compound thus
prepared on active ingredient basis and was kept in refrigerator
till use. The required dilutions of 25, 20, 10, 5, 2.5, 1 and 0.5
µg/mL were subsequently made from the stock solution by
adding distilled water as and when required.
(benzene:ethyl acetate, 19:1) having R = 0.65 and the structure
f
was confirmed by spectroscopic analysis.
12
1
8
2
3
9
1
7
3
1
4
6
1
0
4
5
1
1O
RESULTS AND DISCUSSION
Structure of cyprene epoxide (5)
C. scariosus oil was obtained from laboratory stock and
was reddish brown liquid. The physical properties of the oil
were examined such as refractive index (1.39), specific gravity
1
The H NMR (CDCl
singlets at δ = 1.19 ppm (C11-CH
.90 ppm (C14-CH ). It also showed a doublet at 1.00 ppm
J = 6.84 Hz) corresponding to C12-CH group. The chemical
3
) spectrum of compound 5 showed
3
), 0.74 ppm (C13-CH ) and
3
(
1.508), viscosity (2.061 poise) and pH (3.3).
0
(
3
The essential oil was insoluble in water, sparingly soluble
in hexane and completely soluble in acetone and ethanol. Thin
layer chromatography of the C. scariosus oil showed seven
3
shifts observed in case of cyprene epoxide (5) were downfield
as compared to those in cyprene (1) because of deshielding
occurred due to formation of epoxide ring. Further, the forma-
tion of epoxide was determined with the help of IR spectrum.
The IR analysis of cyprene expoxide (5) showed the presence
of bands at 1277 cm corresponding to C-O stretching and
89 and 803 corresponding to C-O bending of epoxy group.
The intense bands corresponding to gem dimethyl group were
found at 1378 and 1316 cm .
coloured spots having R
f
values of 0.95, 0.90, 0.83, 0.70, 0.63,
0
.55 and 0.45. Gas chromatography-mass spectrometry (GC-
MS) data of isolated C. scariosus oil showed the presence of
cyprene (1) (18.57 %) as the major compound. Other minor
compounds present were cyclopropazulen-7-ol, caryophyllene
oxide, isolongifolen-5-one, longiverbenone, zierone etc. C.
scariosus oil was subjected to column chromatography to
fractionate it into non-polar (hexane) and polar fractions
-1
8
-1
Evaluation of Cyperus scariosus oil and its components
for plant growth regulator activity: After the successful
derivatization of cyprene (1) to cyprene epoxide (5), C. scariosus
oil, its non-polar and polar fractions, cyprene (1) and cyprene
epoxide (5) were screened for their plant growth regulation
studies. For initial screening of the oil, its fractions, isolated
compound and its derivative, we preferred to use those seeds
which can give results within 7-10 days e.g. wheat, mung beans
etc. Therefore, two different varieties of wheat (Triticum
aestivum), HD 2967 and PBW 621 were selected. The effect
of oil, its non-polar and polar fractions, cyprene (1) and cyprene
epoxide (5) were studied on root length, shoot length, fresh
weight, dry weight and seedling vigour index.
Observations: The different parameters such as fresh
weight, dry weight, root length and shoot length were observed
in control and are listed in Table-1.
The following observations were recorded in case of wheat
seedlings treated with the oil, its fractions, isolated compound
and its derivative:
(
dichloromethane). The TLC of non-polar fraction showed one
major pinkish-brown spot having R = 0.95, along with other
minor spots. However, the dichloromethane fraction showed
six spots having R values 0.90, 0.83, 0.70, 0.63, 0.55 and 0.45.
f
f
Isolation of cyprene (1) from Cyperus scariosus oil:
Column chromatography of non-polar (hexane) fraction of C.
scariosus afforded major spot cyprene (1). The purity of the
compound was checked byTLC and the structure was confirmed
by spectral analysis.
1
The H NMR (CDCl
3
) spectrum of compound 1 showed
) which indicated the presence
of tetrasubstituted double bond, 0.77 ppm (C13-CH ) and 0.95
ppm (C14-CH ). A doublet at 0.80 ppm (J = 6.4 Hz) was
observed for the -CH group at C12 position. The compound
singlets at δ = 1.62 ppm (C11-CH
3
3
3
3
failed to show any signal corresponding to the olefinic protons,
thus, it confirmed that the double bond was tetrasubstituted.
The IR analysis of compound 1 showed intense bands at
-1
1
382, 1372 cm corresponding to C13 and C14 gem dimethyl
-1
group.A band was observed at 3078 and 1644 cm correspon-
ding to C-H stretching and C=C stretching of tetrasubstituted
double bond, respectively.
Germination percentage: Germination percentage of the
seeds germinated was calculated using the formula:
Epoxidation of cyprene: The oxidation of terpenes is an
important industrial application as epoxides are used as starting
Number of seeds germinated
Germination (%) =
×100
Total number of seeds kept

 Sharma
Sharma
 Utreja
Utreja
 Bedi
Bedi