M. Nanjo et al.
Inorganica Chimica Acta 528 (2021) 120608
obtained. Relatively good microcrystals of 2 were obtained by recrys-
tallization from dimethyl sulfoxide (DMSO). The crystals included four
equivalents of DMSO and more than ten equivalents of water based on 2
determined by 1H NMR spectroscopy (Fig. S6). Although part of the
water might come from the NMR solvent, it suggests that sodium ger-
manate 2 might also exhibit hygroscopicity. The elemental analysis of 2
was performed by using microcrystals recrystallized from methanol /
DMSO (1:1) mixed solvent. The results indicated that the composition
Table 1
Selected bond distances (Å) and bond angles (degree) of germanate complex 1-
(dme)2.5-(ipa)1.5.
Bond distances
Ge1 – O1
1.8939(19)
1.9008(18)
1.890(2)
Ge1 – O2
Ge1 – O4
Ge1 – O6
Ge2 – O14
Ge2 – O16
Ge2 – O18
Ge1 – Li2
Ge2 – Li4
1.8866(18)
1.8701(18)
1.8760(19)
1.8554(19)
1.896(2)
Ge1 – O3
Ge1 – O5
Ge2 – O13
1.9125(19)
1.9030(19)
1.8606(19)
3.045(5)
Ge2 – O15
should be 2-(CH
3
OH)
3
-(dmso)
4
. However, the amount of the sample was
Ge2 – O17
1.911(2)
Ge1 – Li1
3.265(6)
too small to measure the NMR spectra using the same sample as the
Ge2 – Li3
3.024(5)
3.577(6)
elemental analysis. As similar to 1, the crystals of 3 also contained water,
Bond angles
2 4
which is determined by the elemental analysis (3-(H O) ). The water
O1 – Ge1– O2
O1 – Ge1 – O4
O1 – Ge1 – O6
O2 – Ge1 – O4
O2 – Ge1 – O6
O3 – Ge1 – O5
O4 – Ge1 – O5
O5 – Ge1 – O6
O13 – Ge2 – O15
O13 – Ge2 – O17
O14 – Ge2 – O15
O14 – Ge2 – O17
O15 – Ge2 – O16
O15 – Ge2 – O18
O16 – Ge2 – O18
86.06(8)
98.20(8)
84.15(8)
89.36(8)
96.02(8)
96.12(8)
91.69(8)
86.37(8)
87.43(8)
172.38(9)
172.76(8)
90.03(9)
85.34(8)
82.41(8)
167.26(8)
O1 – Ge1 – O3
O1 – Ge1 – O5
O2 – Ge1 – O3
O2 – Ge1 – O5
O3 – Ge1 – O4
O3 – Ge1 – O6
O4 – Ge1 – O6
O13 – Ge2 – O14
O13 – Ge2 – O16
O13 – Ge2 – O18
O14 – Ge2 – O16
O14 – Ge2 – O18
O15 – Ge2 – O17
O16 – Ge2 – O17
O17 – Ge2 – O18
88.64(8)
169.36(8)
171.94(8)
90.13(8)
85.39(8)
89.46(8)
174.27(8)
85.69(8)
94.90(8)
88.04(8)
93.01(9)
99.58(9)
97.06(9)
91.60(9)
86.46(9)
signal is also observed in the 1H NMR spectrum of the sample recrys-
tallized from isopropyl alcohol (IPA) (Fig. S8). All the germanate com-
plexes are hygroscopic and soluble in water. Especially, sodium
germanate 2 in water could be kept without decomposition for a month.
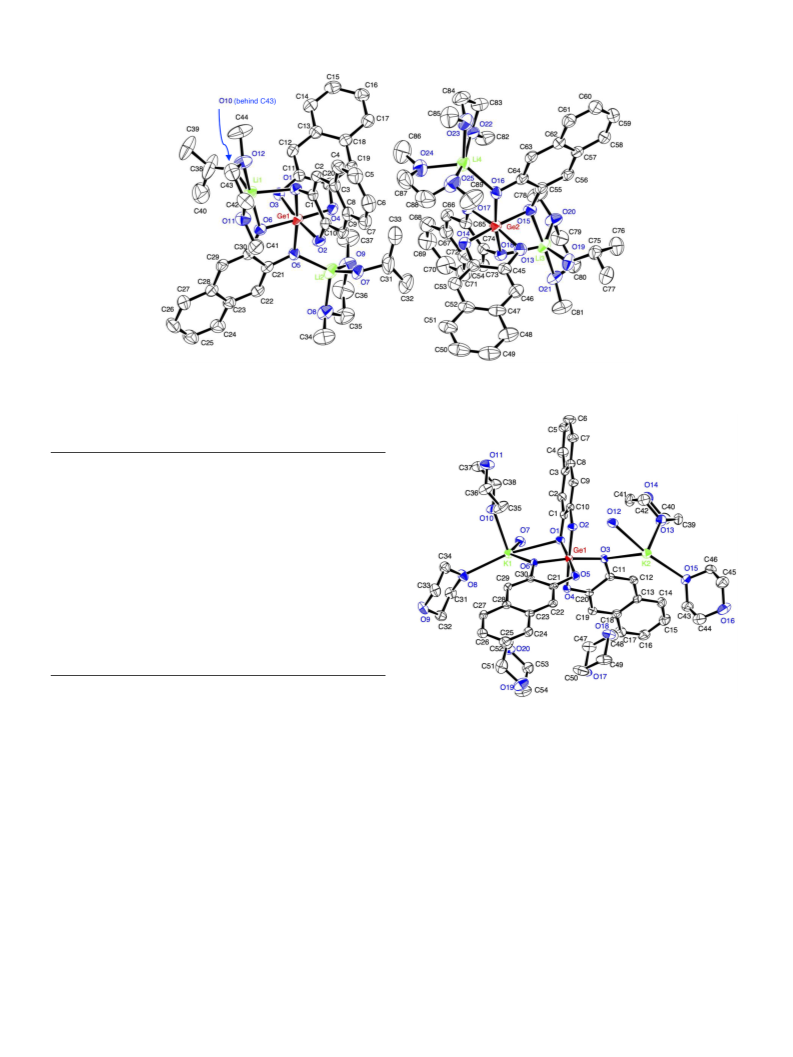
A single crystal of lithium germanate 1 could be obtained by the
recrystallization from a mixed solvent, IPA / 1,2-dimethoxyethane
(
DME). The molecular structure of lithium germanate 1 was estab-
lished by single crystal X-ray diffraction analysis. As the results of the X-
ray diffraction, the obtained crystals contained 2.5 molecules of DME
and 1.5 molecules of IPA for a lithium germanate. Therefore, the lithium
germanate complex in the crystals obtained by recrystallization is
denoted as 1-(dme)2.5-(ipa)1.5. The selected bond distances and angles
are summarized in Table 1. The crystal system is triclinic and the space
group is P-1 (#2). There are two independent molecules in the unit cell,
which structures closely resemble each other (Fig. 1). The molecules
including Ge1 or Ge2 are abbreviated as mol. A and mol. B, respectively.
The germanium atom is coordinated by six oxygen atoms in a slightly
distorted octahedral configuration. The Ge-O bond lengths are within
the range from 1.870 Å (Ge1-O4) to 1.901 Å (Ge1-O3) in mol. A, and
from 1.855 Å (Ge2-O14) to 1.913 pm (Ge2-O13) in mol. B. Considering
◦
1
71.96 , which deviates from linear form due to the influence of the
bulky naphthalene rings.
There are two kind of lithium atoms in the crystal unit cell. Li1, Li2,
and Li3 are coordinated by one IPA, one DME, and two oxygen atoms of
a germanate complex, respectively. Only Li4 is solvated by two DME and
is coordinated by one oxygen atom (O16) of a germanate complex. Li1,
Li3, and Li4 atoms are coordinated by five oxygen atoms to form dis-
torted trigonal bipyramid, and Li2 atom is coordinated by four oxygen
atoms to form tetrahedral configuration. The interatomic distances be-
tween germanium and lithium are>3.024 Å, suggesting that there is no
covalent or electrostatic interaction between them.
3
that the typical Ge(sp )-O bond length is 1.73–1.75 Å [10], the Ge-O
bond lengths in 1-(dme)2.5-(ipa)1.5 are clearly elongated for the hyper-
coordinate compound. These values are slightly longer than those found
in tris(oxalato)germanium complexes (1.8641–1.8898 Å) [7] or octa-
hedral bis(3,5-di-tert-butylcatecholato)germanium tetramethylethyle-
nediamine adduct (1.8457–1.856 Å) [6] probably due to the weaker
donor nature of naphthalene-2,3-dialkoxide ligands. The Ge-O bond
lengths in 1⋅2.5DME⋅1.5IPA are apparently longer than those found in
bis(3,5-di-tert-butylcatecholato)germanium complexes (1.839–1.841 Å
Growing a single crystal of potassium germanate 3 was also suc-
cessful by the recrystallization from 1,4-dioxane with small amount of
water. The crystal structure of 3 was determined by X-ray diffraction
analysis, and selected bond lengths and angles are summarized in
Table 2. As the results of the X-ray diffraction, the obtained crystals
contained four molecules of 1,4-dioxane and two molecules of water for
a potassium germanate. Therefore, the potassium germanate complex in
[
5] or 1.847–1.849 Å [6]) or bis(3,6-di-tert-butylcatecholato)germa-
nium complexes (1.819–1.8256 Å) [11], where it should be noted that
two catecholate ligands in these complexes are bonded to germanium
center in coplanar fashion. When O2 and O3 are in the apical position
4 8 2 4 2 2
the single crystal is denoted as 3-(C H O ) -(H O) . The crystal struc-
ture contained four clathrate dioxane molecules in the unit cell. The
germanium center is coordinated by six oxygen atoms of 2,3-naphthale-
nedialkoxide ligands to form an octahedral arrangement, as shown in
Fig. 2. The average length of Ge-O bonds is 1.874 Å, which is slightly
against the germanium center, the averaged vertical angle O-Ge-O is
◦
9
0.14 , suggesting that four oxygen atoms O1, O4, O5, and O6 are
located on a same square plane. However, the angle O2-Ge1-O3 is
Scheme 1. Syntheses of germanate complexes (1 – 3).
2

 Goto, Mitsuki
Goto, Mitsuki
 Nakashima, Yuta
Nakashima, Yuta
 Nanjo, Masato
Nanjo, Masato