G Model
IJP 14862 1–9
U.M. Musazzi et al. / International Journal of Pharmaceutics xxx (2015) xxx–xxx
9
637
638
639
640
641
642
643
644
645
646
647
648
712
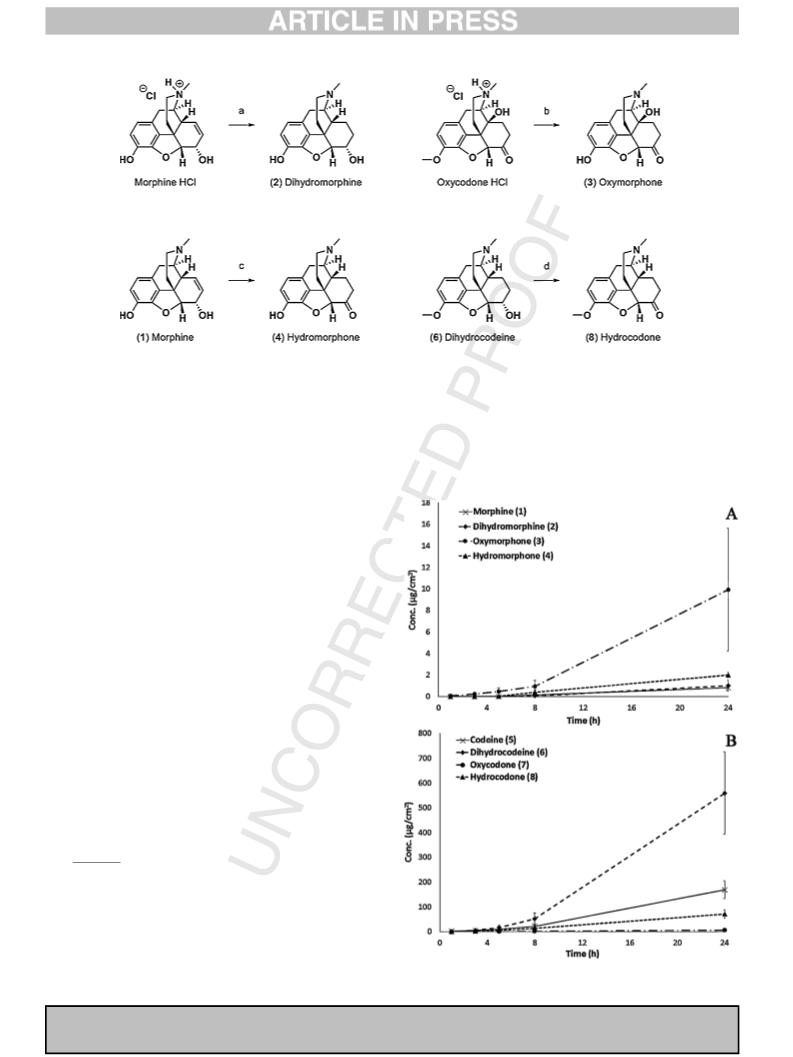
analgesic treatments, the D/Ki ratio confirms the relevance of the 6-
carbonyl group (Table 3; Table 4), a finding in line with the well-
Lattanzi, R., Spetea, M., Schüllner, F., Rief, S.B., Krassnig, R., Negri, L., Schmidhammer,
H., 2005. Synthesis and biological evaluation of 14-alkoxymorphinans.
22.1 Influence of the 14-alkoxy group and the substitution in position 5 in 14-
alkoxymorphinan-6-ones on in vitro and in vivo activities. J. Med. Chem. 48 (9),
3372–3378.
713
714
715
716
717
718
719
720
721
722
723
724
725
726
727
728
729
730
731
732
733
734
735
736
737
738
739
740
741
742
743
744
745
746
747
748
749
750
751
752
753
754
755
756
757
758
759
760
761
762
763
764
765
766
767
768
769
770
771
772
773
774
775
776
777
778
779
780
781
known beneficial effect on the
m-receptor potency of the oxidation
of the 6-hydroxy group especially along with reduction of the 7–
8 double bond (Pasternak and Pan, 2013). Furthermore, among the
compounds having position 6 oxidized, the oxymorphone 3 and
Liu, X., Testa, B., Fahr, A., 2011. Lipophilicity and its relationship with passive drug
permeation. Pharm. Res. 28 (5), 962–977.
Mazak, K., Hosztafi, S., Racz, A., Noszal, B., 2009. Structural and physicochemical
profiling of morphine and related compounds of therapeutic interest. Mini Rev.
Med. Chem. 9 (8), 984–995.
hydromorphone
4
could be preferred to oxycodone
7
and
hydrocodone 8, since their Clr values suggested
a
slower
Metzger, H., 1935. Verfahren zur darstellung von dihydromorphinonen [A process
for the preparation of dihydromorphinones]. German patent 607931.
Metzger, H., 1936. Verfahren zur Darstellung von dihydromorphinonen [A process
for the preparation of dihydromorphinones]. German patent 623821.
Minghetti, P., Casiraghi, A., Cilurzo, F., Tosi, L., Montanari, L., Trespidi, L., 2003.
Formulation study and anti-inflammatory efficacy of topical semi-solids
containing a nitro ester of flurbiprofen. Skin Pharmacol. Physiol. 16 (2), 91–99.
Mitragotri, S., Anissimov, Y.G., Bunge, A.L., Frasch, H.F., Guy, R.H., Hadgraft, J.,
Kasting, G., Lane, M., Roberts, M., 2011. Mathematical models of skin
permeability: an overview. Int. J. Pharm. 418, 115–129.
elimination (Table 2). Finally, hydromorphone 4 exhibited a higher
Qret/Jmax value than oxymorphone 3, which should be related to a
reduction of systemic concentration, and, therefore, with lower
potential risks of side effects.
649
References
650
651
652
653
654
655
656
657
658
659
660
Anissimov, Y.G., Jepps, O.G., Dancik, Y., Roberts, M.S., 2013. Mathematical and
pharmacokinetic modelling of epidermal and dermal transport processes. Adv.
Drug Deliv. Rev. 65 (2), 169–190.
Anissimov, Y.G., Roberts, M.S., 1999. Diffusion modeling of percutaneous absorption
kinetics. 1. Effects of flow rate, receptor sampling rate, and viable epidermal
resistance for a constant donor concentration. J. Pharm. Sci. 88 (11), 1201–1209.
Avdeef, A., 1992. pH-Metric log P. Part 1. Difference plots for determining ion-pair
octanol–water partition coefficients of multiprotic substances. Quant. Struct.–
Act. Relat. 11 (4), 510–517.
Avdeef, A., 1993. pH-metric log P. II. Refinement of partition coefficients and
ionization constants of multiprotic substances. J. Pharm. Sci. 82 (2), 183–190.
Bailey, T.S., Gee, P.S., Rezaie, R., 2006. Process for the synthesis of hydromorphone.
US WO2006005112 A1, PCT/AU2005/001002.
Bigliardi, P.L., Tobin, D.J., Gaveriaux-Ruff, C., Bigliardi-Qi, M., 2009. Opioids and the
skin – where do we stand? Exp. Dermatol. 18 (5), 424–430.
Cilurzo, F., Vistoli, G., Selmin, F., Gennari, C.G.M., Musazzi, U.M., Franzé, S., Lo Monte,
M., Minghetti, P., 2014. An insight into the skin penetration enhancement
mechanism of N-methylpyrrolidone. Mol. Pharm. 11 (3), 1014–1021.
Cleek, R., Bunge, A., 1993. A new method for estimating dermal absorption from
chemical exposure. 1. General approach. Pharm. Res. 10 (4), 497–506.
Codd, E.E., Shank, R.P., Schupsky, J.J., Raffa, R.B., 1995. Serotonin and norepinephrine
uptake inhibiting activity of centrally acting analgesics: structural determinants
and role in antinociception. J. Pharm. Exp. Ther. 274 (3), 1263–1270.
Corbe, E., Laugel, C., Yagoubi, N., Baillet, A., 2007. Role of ceramide structure and its
microenvironment on the conformational order of model stratum corneum
lipids mixtures: an approach by FTIR spectroscopy. Chem. Phys. Lipids 146 (2),
67–75.
Monory, K., Greiner, E., Sartania, N., Sallai, L., Pouille, Y., Schmidhammer, H.,
Hanoune, J., Borsodi, A., 1999. Opioid binding profiles of new hydrazone, oxime,
carbazone and semicarbazone derivatives of 14-alkoxymorphinans. Life Sci. 64
(22), 2011–2020.
Pasternak, G.W., Pan, Y.-X., 2013. Mu opioids and their receptors: evolution of a
concept. Pharmacol. Rev. 65 (4), 1257–1317.
Pedretti, A., Villa, L., Vistoli, G., 2002a. Modeling of binding modes and inhibition
mechanism of some natural ligands of farnesyl transferase using molecular
docking. J. Med. Chem. 45 (7), 1460–1465.
Pedretti, A., Villa, L., Vistoli, G., 2002b. VEGA: a versatile program to convert, handle
and visualize molecular structure on windows-based PCs. J. Mol. Graph. Model.
21 (1), 47–49.
Przybyl, A.K., Flippen-Anderson, J.L., Jacobson, A.E., Rice, K.C., 2003. Practical and
high-yield syntheses of dihydromorphine from tetrahydrothebaine and
efficient syntheses of (8S)-8-bromomorphide. J. Org. Chem. 68 (5), 2010–2013.
Roy, S.D., Flynn, G.L., 1988. Solubility and related physicochemical properties of
narcotic analgesics. Pharm. Res. 5 (9), 580–586.
Roy, S.D., Flynn, G.L.,1989. Transdermal delivery of narcotic analgesics: comparative
permeabilities of narcotic analgesics through human cadaver skin. Pharm. Res. 6
(10), 825–832.
Santini, D., Lanzetta, G., Dell'Aquila, E., Vincenzi, B., Venditti, O., Russano, M.,
Papapietro, N., Denaro, V., Tonini, G., Ripamonti, C., 2013. ‘Old’ and ‘new’ drugs
for the treatment of cancer pain. Exp. Opin. Pharmacother. 14 (4), 425–433.
Sawynok, J., 2014. Topical analgesics for neuropathic pain: preclinical exploration,
clinical validation, future development. Eur. J. Pain 18 (4), 465–481.
Schmidt, H., Vormfelde, S.V., Klinder, K., Gundert-Remy, U., Gleiter, C.H., Skopp, G.,
Aderjan, R., Fuhr, U., 2002. Affinities of dihydrocodeine and its metabolites to
opioid receptors. Pharmacol. Toxicol. 91 (2), 57–63.
661
662
663
664
665
666
667
668
669
670
671
672
673
674
675
676
677
678
679
680
681
682
683
684
685
686
687
688
689
690
691
692
693
694
695
696
697
698
Cross, S.E., Roberts, M.S., 2008. Use of in vitro human skin membranes to model and
predict the effect of changing blood flow on the flux and retention of topically
applied solutes. J. Pharm. Sci. 97 (8), 3442–3450.
Smith, P.R., Frohwein, A.K., Hays, P.A., Lurie, I.S., 2005. Identification and
quantitation of hydromorphone hydrochloride in Palladone (extended time-
release) capsules. Microgram J. 3 (1-2), 39–45.
Evans, H.C., Easthope, S.E., 2003. Transdermal buprenorphine. Drugs 63 (19),
1999–2010.
Ständer, S., Gunzer, M., Metze, D., Luger, T., Steinhoff, M., 2002. Localization of
m-
Farley, P., 2011. Should topical opioid analgesics be regarded as effective and safe
when applied to chronic cutaneous lesions? J. Pharm. Pharmacol. 63 (6),
747–756.
Franzè, S., Gennari, C.G.M., Minghetti, P., Cilurzo, F., 2015. Influence of chemical and
structural features of low molecular weight heparins (LMWHs) on skin
penetration. Int. J. Pharm. 481 (1–2), 79–83.
Fujioka, N., Nguyen, J., Chen, C., Li, Y., Pasrija, T., Niehans, G., Johnson, K.N., Gupta, V.,
Kratzke, R.A., Gupta, K., 2011. Morphine-induced epidermal growth factor
pathway activation in non-small cell lung cancer. Anesth. Analg. 113 (6),
1353–1364.
Gindelberger, D.E., 2011. Heterogeneous ruthenium metal catalyst for the
production of hydrocodone, hydromorphone or a derivative thereof. WIPO
patent 2011035276A2.
Graham, T., Grocott, P., Probst, S., Wanklyn, S., Dawson, J., Gethin, G., 2013. How are
topical opioids used to manage painful cutaneous lesions in palliative care? A
critical review. PAIN 154 (10), 1920–1928.
Gupta, M.A., Gupta, A.K., Vujcic, B., Piccinin, M., 2014. Use of opioid analgesics in skin
disorders: results from a nationally representative US sample. J. Dermatolog.
Treat. 1–6.
International Narcotics Control Board (INCB), 2011. Report of the International
Narcotics Control Board on the Availability of Internationally Controlled Drugs:
Ensuring Adequate Access for Medical and Scientific Purposes 2010. INCB, New
York, United Nations.
opioid receptor 1A on sensory nerve fibers in human skin. Regul. Peptides 110
(1), 75–83.
Tavakol, H., Esfandyari, M., Taheri, S., Heydari, A., 2011. Investigation of structure,
vibrational and NMR spectra of oxycodone and naltrexone: a combined
experimental and theoretical study. Spectrochim. Acta A 79 (3), 574–582.
Trost, B.M., Tang, W., 2002. Enantioselective synthesis of (ꢃ)-codeine and
(ꢃ)-morphine. J. Am. Chem. Soc. 124 (49), 14542–14543.
Varadi, A., Gergely, A., Beni, S., Jankovics, P., Noszal, B., Hosztafi, S., 2011. Sulfate
esters of morphine derivatives: synthesis and characterization. Eur. J. Pharm.
Sci. 42 (1–2), 65–72.
Varghese, V., Hudlicky, T., 2013. Total synthesis of dihydrocodeine and hydrocodone
via a double claisen rearrangement and C-10/C-11 closure strategy. Synlett 24
(3), 369–374.
Weiss, U., Weiner, N., 1949. The isomerization of morphine to O-
desmethylthebainone. J. Org. Chem. 14 (2), 194–203.
Welling, A., 2007. A randomised controlled trial to test the analgesic efficacy of
topical morphine on minor superficial and partial thickness burns in accident
and emergency departments. Emerg. Med. J. 24 (6), 408–412.
Wentland, M.P., Lou, R., Lu, Q., Bu, Y., Denhardt, C., Jin, J., Ganorkar, R., VanAlstine, M.
A., Guo, C., Cohen, D.J., Bidlack, J.M., 2009. Syntheses of novel high affinity
ligands for opioid receptors. Bioorg. Med. Chem. Lett. 19 (8), 2289–2294.
Wertz, P.W., van den Bergh, B., 1998. The physical, chemical and functional
properties of lipids in the skin and other biological barriers. Chem. Phys. Lipids
91 (2), 85–96.
Zaslansky, R., Ben-Nun, O., Ben-Shitrit, S., Ullmann, Y., Kopf, A., Stein, C., 2014. A
randomized, controlled, clinical pilot study assessing the analgesic effect of
morphine applied topically onto split-thickness skin wounds. J. Pharm.
Pharmacol. 66 (11), 1559–1566.
699
700
701
702
703
704
705
706
707
708
709
710
711
782
783
784
785
786
787
788
789
790
791
792
793
Koizumi, H., Yokoshima, S., Fukuyama, T., 2010. Total synthesis of (ꢃ)-morphine.
Chem. – Asian J. 5 (10), 2192–2198.
Kok, G.B., Scammells, P.J., 2012. Improved synthesis of 14-hydroxy opioid
pharmaceuticals and intermediates. RSC Adv. 2 (30), 11318–11325.
Korb, O., Stützle, T., Exner, T.E., 2009. Empirical scoring functions for advanced
protein–ligand docking with plants. J. Chem. Inf. Model. 49 (1), 84–96.
Kvernenes, O.H., Nygard, A.M., Heggelund, A., Halvorsen, H., 2007. Process useful in
the preparation of morphinan antagonists. WIPO patent 2007137785A2.
Lane, M.E., 2013. The transdermal delivery of fentanyl. Eur. J. Pharm. Biopharm. 84
(3), 449–455.
Zeppetella, G., Paul, J., Ribeiro, M.D.C., 2003. Analgesic efficacy of morphine applied
topically to painful ulcers. J. Pain Symptom Manag. 25 (6), 555–558.
Zur, E., 2014. Topical treatment of neuropathic pain using compounded
medications. Clin. J. Pain 30 (1), 73–91.
Please cite this article in press as: Musazzi, U.M., et al., On the selection of an opioid for local skin analgesia: Structure-skin permeability

 Musazzi, Umberto M.
Musazzi, Umberto M.
 Matera, Carlo
Matera, Carlo
 Dallanoce, Clelia
Dallanoce, Clelia
 Vacondio, Federica
Vacondio, Federica
 De Amici, Marco
De Amici, Marco
 Vistoli, Giulio
Vistoli, Giulio
 Cilurzo, Francesco
Cilurzo, Francesco
 Minghetti, Paola
Minghetti, Paola