50
X. Li et al. / Applied Catalysis A: General 488 (2014) 48–57
2.2. Preparation of Pt/Al2O3@SBA-15 catalysts
cooled down to room temperature, the samples were exposed to
pyridine vapor for 20 min. After degassing at 573 K for 1 h, the spec-
trum was recorded to characterize the acidity of the samples. 29Si
and 27Al MAS NMR spectra were recorded at 400 MHz on a Var-
ian VNMRS400WB spectrometer equipped with a magic angle spin
probe at room temperature. The leached amount of Pt atoms into
solution after reaction were detected with a Thermo Elemental
IRIS Intrepid II XSP inductively coupled plasma-atomic emission
spectroscopy (ICP-AES).
The 5 wt.% Pt/xAS catalysts were prepared mainly according to
Ref. [23]. The xAS composites were impregnated with H2PtCl6 solu-
tion dissolved in a 1:1 (v/v) mixture of water and ethanol. According
to desirable 5 wt.% Pt loading, the required volume of H2PtCl6 solu-
tion with a concentration of 14.8 mg Pt/ml was calculated firstly
and then about 3–4 mL of water and ethanol mixed solvent (1:1,
v/v) was added to make the support immerse well in the solution.
After stirring for 6 h, the mixture was evaporated to remove the
excess solvent, followed by drying at 393 K overnight. The cata-
lyst precursor was then directly reduced in an aqueous solution
of sodium formate by refluxing at 363 K for 2 h. To make the cat-
alyst precursor immerse well in the solution, about 20 mL water
was added. Finally, the mixture was washed by plenty of water to
remove chlorine ions and dried at 353 K overnight.
2.4. Catalytic tests
In a typical reaction, 0.1 g 5 wt.% Pt/xAS catalyst was pre-
treated under a hydrogen flow at 673 K for 2 h before use. The
catalyst was then mixed with 20 mL acetic acid, 10 mg cinchoni-
dine and 1 mL EOPB. The mixture was subsequently transferred
to a 100 mL autoclave. The hydrogenation reaction began with
stirring (1000 rpm) at room temperature after 4.0 MPa of hydro-
gen was introduced into the autoclave. The reaction was stopped
after 15 min and the products were analyzed by GC-FID (GC-2014,
Shimadzu Co.) equipped with a chiral capillary column (HP19091G-
B213, 30 m × 0.32 mm × 0.25 m, Agilent Co.). The optical yield was
expressed as the enantiomeric excess (% ee) of (R)-(+)-ethyl lactate:
ee (%) = ([R] − [S])/([R] + [S]) × 100.
2.3. Characterization of xAS composites and Pt/xAS catalysts
The X-ray diffraction (XRD) patterns of samples were collected
on a Bruker D8 Advance instrument using Cu-K␣ radiation. The
nitrogen adsorption–desorption isotherms were measured at 77 K
on a Quantachrome Autosorb-3B system, after the samples were
evacuated for 10 h at 473 K. The BET specific surface area was cal-
culated using adsorption data in the relative pressure range from
0.05 to 0.30. The pore size distributions were calculated from the
analysis of the adsorption branch of the isotherm using the BJH
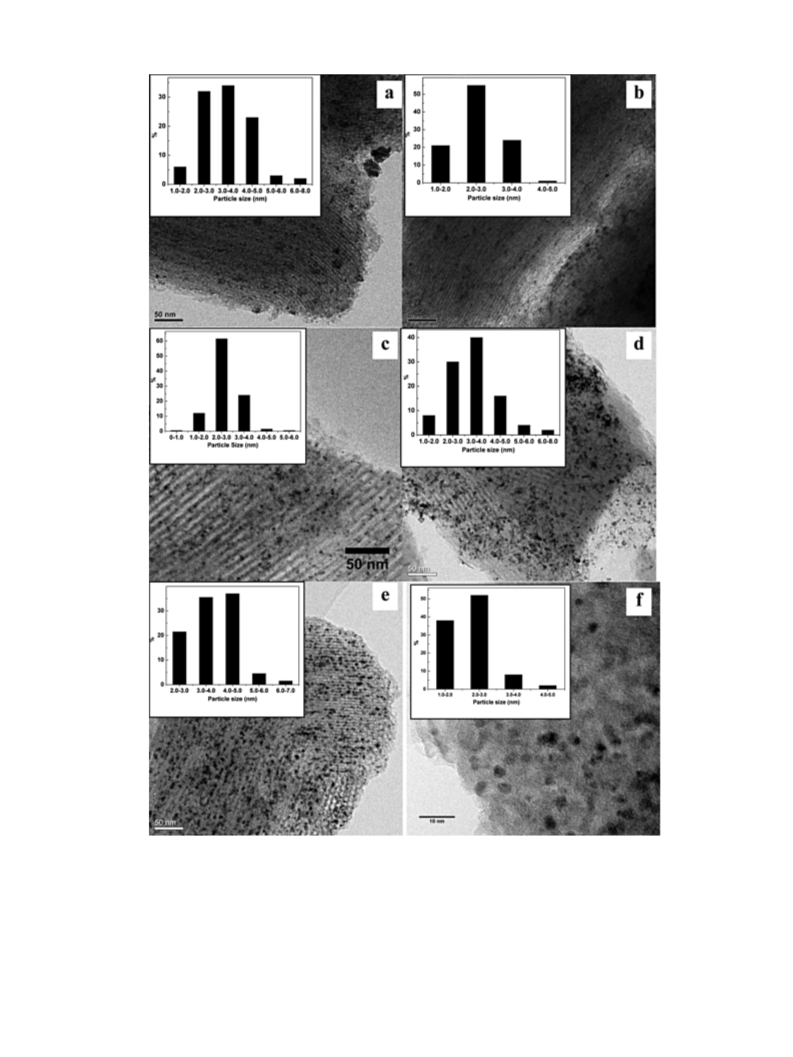
algorithm. The transmission electron microscopy (TEM) images
were taken on an FEI Tecnai G2-TF30 microscope at an acceler-
ation voltage of 300 kV. The scanning electron microscopy (SEM)
images were taken on a Hitachi S4800 electromicroscope with an
acceleration voltage of 20 kV.
After each run, the catalyst was recovered by centrifugation and
washed with fresh solvent for several times. Then, fresh reactant,
solvent and cinchonidine were charged to the autoclave together
with the recovered catalyst to conduct the next run reaction.
3. Results and discussion
CO chemisorption of samples was measured at 308 K on a
Quantachrome CHEMBET-3000 pulse chemisorption analyzer after
the samples were pretreated in a 5 vol.% H2/95 vol.% Ar flow at 673 K
for 2 h. The degree of dispersion and the mean particle size (cubic
model) were estimated from the measured CO uptake, assuming a
cross-sectional area for a surface platinum atom of 8.0 × 10−20 m2
and a stoichiometric factor of one, using nominal platinum concen-
trations.
The surface electronic state of platinum particles was exam-
ined by diffuse reflectance infrared Fourier-transform spectroscopy
(DRIFTS) using CO as probe molecules. The analysis was conducted
with a Nicolet NEXUS 670 spectrometer. The catalyst sample was
pretreated in a 5 vol.% H2/95 vol.% Ar stream at 673 K for 2 h, and
then was purged with He for 30 min. When the sample was cooled
down to 308 K, CO was introduced into the IR cell until the adsorp-
tion was saturated. Then, the sample was flushed with a He flow
before the spectrum was recorded.
The surface electronic state of Pt was also evaluated using X-ray
photoelectron spectroscopy (XPS) measurements with a Thermo
Fisher Scientific ESCALAB 250Xi spectrometer with Al K␣ radia-
tion (1486.6 eV) as incident beam with a monochromator. All the
samples were pretreated in flowing hydrogen at 673 K for 1 h in a
reactor attachment of the XPS spectrometer. All the spectra were
obtained at room temperature, and the binding energies of ele-
ments were referenced to the adventitious C1s peak at 284.8 eV.
The spectra shown in the figures have been corrected by subtrac-
tion of a Shirley background. Spectral fitting and peak integration
was done using the XPSPEAK software.
The acidic properties of the xAS composites were characterized
by IR spectroscopy, using pyridine as probe molecules. The infrared
spectra of xAS and Pt/xAS samples were also recorded with a Nicolet
NEXUS 670 Fourier transform infrared spectrometer. Before the IR
investigation, the samples were pressed into self-supported wafers
and activated under vacuum at 773 K for 1 h. When the cell was
3.1. Structural characterization of xAS composites and Pt/xAS
catalysts
The small angle XRD patterns of xAS composites and the resul-
tant Pt/xAS catalysts are shown in Fig. S1 in the Supplementary data.
The xAS composites and the resultant Pt/xAS catalysts all displayed
the hexagonal space group p6mm structure [18]. Compared with
SBA-15 host, the diffraction angles for the Al2O3@SBA-15 compos-
ites shifted slightly to higher region, probably due to the shrinkage
during the re-calcination process [24]. This demonstrates that
inclusion of alumina inside SBA-15 mesopores and even loading of
Pt nanoparticles did not destroy the structure of SBA-15 host. This
also implies that the xAS composites possess a strong hydrothermal
stability as the Pt/xAS catalysts suffered from impregnation with
the acidic H2PtCl6 solution and were refluxed at a high tempera-
ture in an aqueous solution of sodium formate in order to reduce
the Pt precursors.
The mesoporous structure of xAS composites and the resul-
tant Pt/xAS catalysts was further confirmed by the nitrogen
adsorption–desorption isotherms. As displayed in Figs. S2 and S3
in the Supplementary data, the xAS composites and the Pt/xAS
catalysts exhibited typical type IV isotherms and showed clear H1-
type hysteresis loops in the range of P/P0 = 0.5–0.8. Nevertheless,
the mesopores of SBA-15 host were partly plugged by introduced
alumina in some cases, especially for those with higher loading
of alumina; as a result, the pore volumes were correspondingly
decreased. Table 2 lists the relevant physicochemical properties
of all the xAS composites. The BET specific surface area and the
pore volume for all the xAS composites, regardless of the different
preparation method, decreased with increasing alumina loading
compared with the SBA-15 host, because the more alumina was
included, the more surface area and the more pore volume of the
SBA-15 host was occupied. For the xAS composites, the BET specific

 Li, Xiaohong
Li, Xiaohong
 Pan, Huiyan
Pan, Huiyan
 Wang, Haihong
Wang, Haihong
 Wang, Yi Meng
Wang, Yi Meng
 Wu, Peng
Wu, Peng