4-Chloroyphenylboronic acid (4)
4-Chlorophenylboronic acid is prepared using Procedure A. After the addition of 1-bromo-4-chlorobenzene the
reaction mixture is stirred at room temperature for 10 min. before trimethylborate (448.0 µl, 4.0 mmol) is added
at 0 °C. The mixture is then quenched with 0.1 N HCl and extracted with EtOAc. The extract is dried over
1
Na2SO4 and concentrated. The residue is recrystallised from H2O/CH3CN. H-NMR (DMSO-d6, 400 MHz): δ
[ppm] = 7.39 (d, J = 8.6 Hz, 2H), 7.80 (d, J = 8.6 Hz, 2H), 8.14 (bs, 2H); 13C-NMR (DMSO-d6, 100 MHz): δ
[ppm] = 127.4, 135.1, 136.0; 11B-NMR (DMSO-d6, 128 MHz): δ [ppm] = 26.6.
4-Cyanophenylboronic acid (5)
4-Cynaophenylboronic acid is prepared using Procedure B. After the addition of i-PrMgCl·LiCl the reaction
mixture is stirred at 0 °C for 2 h before trimethylborate (1.12 ml, 10.0 mmol) is added at 0 °C. The mixture is
then quenched with 0.1 N HCl and extracted with EtOAc. The extract is dried over Na2SO4 and concentrated.
1
The residue is recrystallised from H2O. H-NMR (DMSO-d6, 400 MHz): δ [ppm] = 7.78 (d, J = 8.1 Hz, 2H),
7.93 (d, J = 8.1 Hz, 2H), 8.35 (bs, 2H); 13C-NMR (DMSO-d6, 100 MHz): δ [ppm] = 112.4, 119.0, 131.0, 134.6;
11B-NMR (DMSO-d6, 128 MHz): δ [ppm] = 26.4.
3-Fluorophenylboronic acid (6)
3-Fluorophenylboronic acid is prepared using Procedure A. After the addition of 3-bromofluorobenzene the
reaction mixture is stirred at room temperature for 15 h before trimethylborate (448.0 µl, 4.0 mmol) is added at 0
°C. The mixture is then quenched with 0.1 N HCl and extracted with EtOAc. The extract is dried over Na2SO4
1
and concentrated. The residue is recrystallised from H2O. H-NMR (DMSO-d6, 400 MHz): δ [ppm] = 7.20 (m,
1H), 7.38 (m, 1H), 7.63 (m, 2H), 8.21 (bs, 2H); 13C-NMR (DMSO-d6, 100 MHz): δ [ppm] = 116.8 (d, J = 21.7
Hz), 120.0 (d, J = 19.2 Hz), 129.6 (d, J = 7.2 Hz), 130.1 (d, J = 2.6 Hz), 162.1 (d, J = 242.0 Hz); 11B-NMR
(DMSO-d6, 128 MHz): δ [ppm] = 28.0; 19F-NMR (DMSO-d6, 376 MHz): δ [ppm] = -114.91;
3-Trifluoromethoxyphenylboronic acid (7)
3-Trifluoromethoxyphenylboronic acid is prepared using Procedure A. After the addition of 3-bromo-(trifluoro-
methoxy)benzene the reaction mixture is stirred at room temperature for 15 h before trimethylborate (448.0 µl,
4.0 mmol) is added at 0 °C. The mixture is then quenched with 0.1 N HCl and extracted with EtOAc. The extract
is dried over Na2SO4 and concentrated. The residue is recrystallised from H2O. 1H-NMR (DMSO-d6, 400 MHz):
δ [ppm] = 7.37 (d, J = 8.1 Hz, 1H), 7.50 (m, 1H), 7.71 (bs, 1H), 7.82 (m, J = 7.1 Hz, 1H), 8.27 (bs, 2H); 13C-
NMR (DMSO-d6, 100 MHz): δ [ppm] = 120.2 (q, J = 255.9 Hz), 122.6, 125.8, 129.5, 133.1, 148.2; 11B-NMR
(DMSO-d6, 128 MHz): δ [ppm] = 28.4; 19F-NMR (DMSO-d6, 376 MHz): δ [ppm] = -57.09.
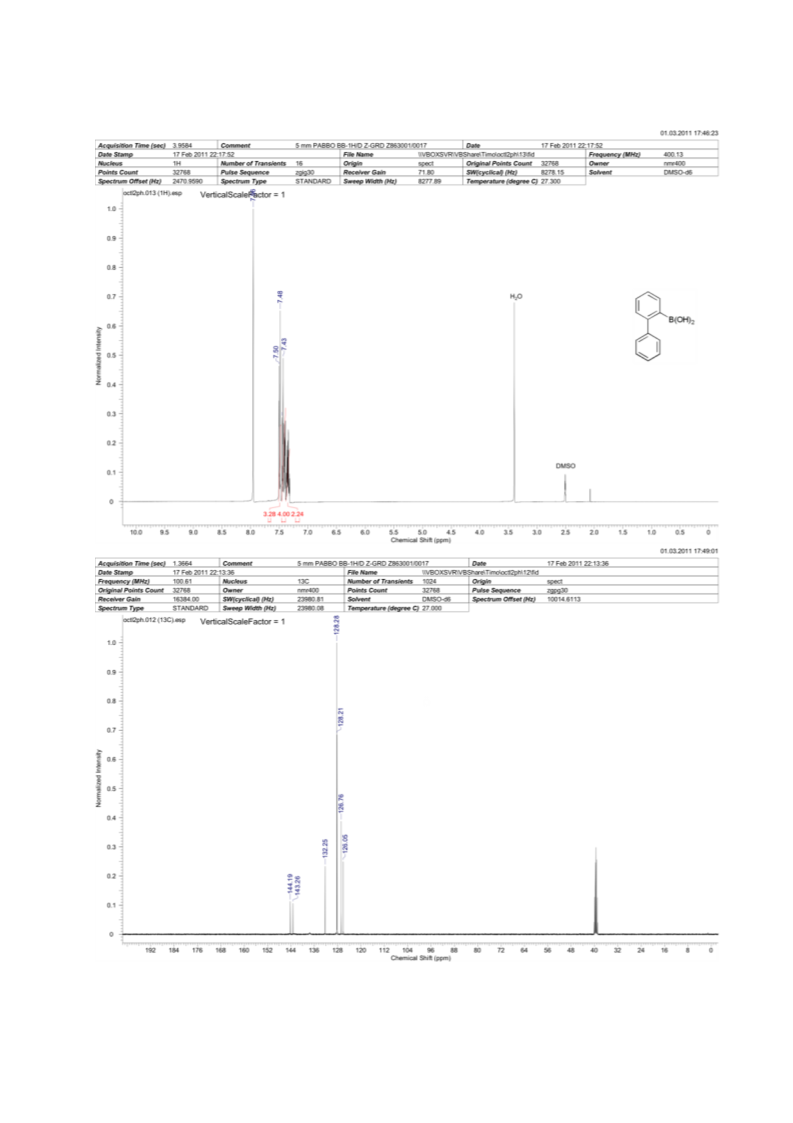
2-Biphenylboronic acid (8)
2-Biphenylboronic acid is prepared using Procedure A. After the addition of 2-bromobiphenyl the reaction
mixture is stirred at room temperature for 1 h before trimethylborate (448.0 µl, 4.0 mmol) is added at 0 °C. The
mixture is then quenched with 0.1 N HCl and extracted with EtOAc. The extract is dried over Na2SO4 and
1
concentrated. The residue is recrystallised from H2O/CH3CN. H-NMR (DMSO-d6, 400 MHz): δ [ppm] = 7.35
(m, 2H), 7.44 (m, 4H), 7.49 (m, 3H), 7.96 (s, 2H); 13C-NMR (DMSO-d6, 100 MHz): δ [ppm] = 126.1, 126.8,
128.2, 128.3, 128.3, 132.3, 143.3, 144.2; 11B-NMR (DMSO-d6, 128 MHz): δ [ppm] = 29.9.
1-Naphthaleneboronic acid (9)
1-Naphthaleneboronic acid is prepared using Procedure A. After the addition of 1-bromonaphthalene the
reaction mixture is stirred at room temperature for 15 h before trimethylborate (448.0 µl, 4.0 mmol) is added at 0
°C. The mixture is then quenched with 0.1 N HCl and extracted with EtOAc. The extract is dried over Na2SO4
1
and concentrated. The residue is recrystallised from H2O. H-NMR (DMSO-d6, 400 MHz): δ [ppm] = 7.48 (m,
3H), 7.75 (d, J = 6.6 Hz, 1H), 7.88 (ψt, J = 8.3 Hz, 2H), 8.39 (d, J = 8.1 Hz, 1H), 8.43 (bs, 2H); 13C-NMR
(DMSO-d6, 100 MHz): δ [ppm] = 125.6, 125.9, 126.1, 128.7, 129.0, 129.7, 132.6, 133.3, 136.0; 11B-NMR
(DMSO-d6, 128 MHz): δ [ppm] = 28.6.
SI-3

 Leermann, Timo
Leermann, Timo
 Leroux, Frederic R.
Leroux, Frederic R.
 Colobert, Francoise
Colobert, Francoise