SHORT PAPER
Compound 1
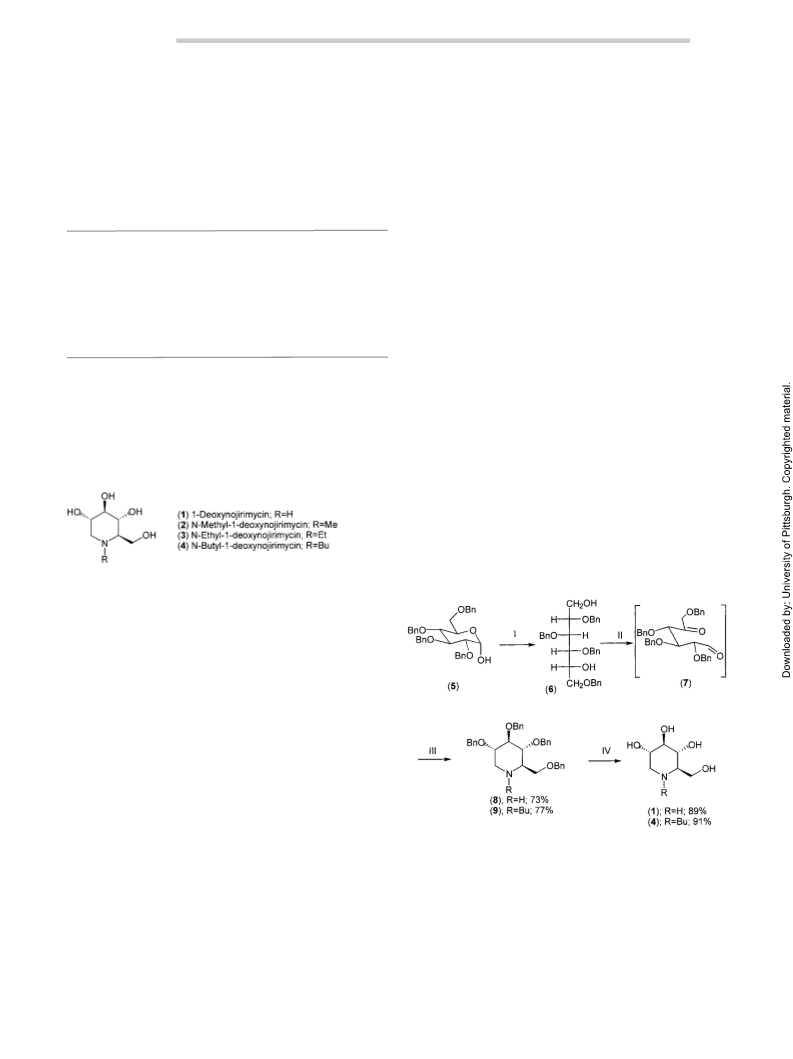
Synthesis of 1-Deoxynojirimycin and N-Butyl-1-deoxynojirimycin
573
(7) Paulsen, H.; Sangster, I.; Heyns, K. Chem. Ber. 1967, 100,
802.
[
α]D25 +42.1 (c = 1, H O); mp 199–201 °C
2
1
H NMR (300 MHz, D O): δ = 2.47 (1 H, dd, J = 10.9, 12.1 Hz),
Behling, J., Farid, P., Medich, J. R., Scaros, M. G., Prunier,
M., Weier, R. M., Khanna, I. Synth. Commun. 1991, 21, 1383.
(8) Yagi, M.; Kauno, K.; Aoyagi, Y.; Murai, H. Nippon Kagaku
Kaishi 1976, 50, 571; Chem. Abstr. 1977, 86, 167851r.
(9) Daigo, K.; Inamori, Y.; Takemoto, T. Chem. Pharm. Bull.
2
2
.55 (1 H, m), 3.13 (1 H, dd, J = 5.1, 12.1 Hz), 3.24 (1 H, t, J = 9.1
Hz), 3.33 (1 H, t, J = 9.1 Hz), 3.50 (1 H, ddd, J = 5.1, 9.1, 10.9 Hz),
3
.64 (1 H, dd, J = 6.2, 11.3 Hz), 3.84 (1 H, dd, J = 2.9, 11.3 Hz).
1
3
C NMR (75 MHz, D O): δ = 51.5, 63.3, 64.2, 73.7, 74.3, 81.2.
2
1986, 34, 2234.
HRMS (CI) calcd for C H NO : 164.0923. Found 164.0916.
6
13
4
(10) Schimidt, D. D.; Fronsmer, W.; Miller, L.; Trushit, E.
Naturwissenschaften 1979, 66, 584.
(11) Hughes, A. B.; Rudge, A. J. Nat. Prod. Rep. 1994, 135.
(12) Saeki, H.; Ohki, E. Chem. Pharm. Bull. 1968, 16, 2477.
Compound 4
[
α]D25 –15.9 (c = 0.77, H O); mp 125–126 °C
2
1
H NMR (300 MHz, D O): δ = 0.91 (3 H, t, J = 7.3 Hz), 1.29 (2 H,
2
(
(
(
(
13) Hofmeister, H.; Lemmes, R. Carbohydr. Res. 1979, 68, 391.
14) Paulsen, H.; Tadt, K. Adv. Carbohydr. Chem. 1968, 23, 115.
15) Bernotas, R. C.; Ganem, B. Tetrahedron Lett. 1985, 26, 1123.
16) Fleet, G. W. J., Fellows, L. E.; Smith, P. W. Tetrahedron
sextet, J = 7.3 Hz), 1.47 (2 H, br m), 2.25 (1 H, m), 2.28 (1 H, dd, J
10.9, 11.6 Hz), 2.61 (1 H, m), 275 (1 H, m), 3.03 (1 H, dd, J = 4.8,
1.4 Hz), 3.26 (1 H, t, J = 9.2 Hz), 3.38 (1 H, t, J = 9.5 Hz), 3.55 (1
H, dt, J = 5.0, 10.7 Hz), 3.83 (1 H, dd, J = 2.5, 12.5 Hz), 3.92 (1 H,
dd, J = 2.5, 12.5 Hz).
=
1
1
987, 43, 979.
17) Iida, H.; Yamazaki, N.; Kibayashi, C. J. Org. Chem. 1987, 52,
337.
(
1
3
C NMR (75 MHz, D O): δ = 16.1, 23.0, 27.5, 54.7, 58.1, 60.4,
2
3
6
8.0, 71.7, 73.0, 81.2.
Kim, N. S.; Choi, J. R.; Cha, J. K. J. Org. Chem. 1993, 58,
7096.
HRMS (CI) calcd for C H NO : 220.1549. Found 220.1545.
1
0
21
4
Furneaux, R. H.; Gainsford, G. J.; Lynch, G. P.; Yorke, S. C.
Tetrahedron 1993, 49, 9605.
Kilonda, A.; Compernolle, F.; Hoornaert, G. J. J. Org. Chem.
Acknowledgement
1
995, 60, 5820.
Financial Support: FUJB, CNPq, CAPES and CEPEG-UFRJ; Ana-
lytical Support: Instituto de Química (IQ), Departamento de Quími-
ca Orgânica (DQO) - UFRJ, Farmanguinhos, Fundação Osvaldo
Cruz, (FIOCRUZ) Ministério da Saúde and Núcleo de Pesquisas de
Produtos Naturais (NPPN) - UFRJ, Department of Chemistry, Wa-
terloo University, Canada. We also thank Professor Jari Nóbrega
Cardoso (LADETEC, IQ-UFRJ) and Victor Snieckus, Department
of Chemistry, Waterloo University, Canada for important com-
ments and suggestions.
Barili, P. L.; Berti, G.; Castelani, G.; D’Andrea, F. Gazz.
Chim. Ital. 1992, 122, 135.
(
18) Kinast, G.; Schedel, M. Angew. Chem., Int. Ed. Engl. 1981,
20, 805.
(
19) Osten, C. H.; Wong, C. H.; Sinskey, A. J.; Barbas, C. F.;
Pederson, R. L.; Wang, Y. F. J. Am. Chem. Soc. 1989, 111,
3924.
(
20) Schultz, P. G. Angew. Chem., Int. Ed. Engl. 1989, 28, 1283.
Kajimoto, T.; Liu, K. K. C.; Pederson, R. L.; Zhong, Z.;
Ichikawa, Y.; Porco Jr. J. A.; Wong, C. H. J. Am. Chem. Soc.
1
991, 113, 6187.
21) Schultz, P. G.; Lemer, R. A.; Benkovic, S. J. Chem. Eng. News
990, 26.
References
(
(
(1) Anzeveno, P. B.; Creemer, L. J.; Daniel, J. K.; King, C. H. R.;
1
Liu, P. S. J. Org. Chem. 1989, 54, 2539.
22) Reitz, A. B.; Baxter, E. W. Tetrahedron Lett. 1990, 31, 6777.
Reitz, A. B.; Baxter, E. W. Biomed. Chem. Lett. 1992, 2, 1419.
Reitz, A. B.; Baxter, E. W. J. Org. Chem. 1994, 59, 3175.
Tsuda, Y.; Okuno, Y.; Kanemitsu, K. Heterocycles 1988, 27,
Robinson, K. M.; Begovic, M. E.; Rhinehart, B. L.; Heineke,
E. W.; Ducep, J. B.; Kastner, P. R.; Marshall F. N.; Danzin, C.
Diabetes 1991, 40, 825.
(2) Spearman, M. A.; Jameson, J. C., Wright, J. A. Exp. Cell Res.
63.
1987, 168, 116.
(
(
(
23) Barilli, P. L.; Berti, G.; Catelani, G.; D’Andrea, F.; De Rensis
F.; Puccioni, L. Tetrahedron 1997, 53, 3407.
24) Cadotte, J. E.; Smith, F.; Spriestersbach, D. J. Am. Chem. Soc.
Tsukamoto, K. T.; Uno, A.; Shrimada, S.; Imokaw, G. Clin.
Res. 1989, 37A, 722.
(
3) Fleet, G. W. J.; Karpas, A.; Dwek, R. A.; Fellows, L. E.;
Tyms, A. S; Petursson, S.; Namgoong, S. K.; Ramsden, N. G.;
Smith, P. W.; Son, J. C.; Wilson, F.; Witty, D. R.; Jacob, G.
S.; Rademacher, T. W. FEBS Lett. 1988, 237, 128.
Khanna, I. K.; Mueller, R. A.; Weier, R. M.; Stealey, M. A.
U.S. Patent 5216168, 1993; Chem. Abstr. 1993, 119, 25037.
Karpas, A.; Fleet, G. W.; Dwek, R. A.; Petursson, S.;
Namgoong, S. K.; Ramsden, N. G.; Jacob, G. S.; Rademacher,
T. W. Proc. Natl. Acad. Sci. U.S.A. 1988, 85, 9229.
Montefiori, D. C.; Robinson, W. E.; Mitchel, W. M. Proc.
Natl. Acad. Sci. U.S.A. 1988, 85, 9248.
1952, 74, 1501.
25) Perrine, T. D.; Claudemans, C. P. J.; Ness, R. K.; Kyle, J.;
Fletcher Jr, H. G. J. Org. Chem. 1967, 32, 664.
26) Fukase, H.; Horii, S. J. Org. Chem. 1992, 57, 3642.
27) Chiara, J. L.; Valle, N. Tetrahedron: Asymmetry 1995, 6,
(
(
1895.
(
28) HPLC and GC analysis from crude mixtures and purified
samples detected only the formation of D-glucitol products 8
and 9; the presence of the corresponding L-iditol isomers were
not observed. These results can be explained by hydride attack
on the bottom face of intermediate B to give the favorable
conformer A. The formation of conformer C has not been
observed due the torsional strain and steric interactions.
(
4) Platt, F. M.; Neises, G. R.; Dwek, R. A.; Butters, T. D. J. Biol.
Chem. 1994, 269, 8362.
Block, X.; Lu X.; Platt, F. M.; Foster, G. R.; Gerlich, W. H.;
Blumberg, B. S.; Dwek, R. A. Proc. Natl. Acad. Sci. U.S.A.
1994, 91, 2235.
(
5) Mann, J.; Davidson R. S.; Hobbs, J. B.; Banthorpe, D. V.;
Harborne, J. B. Natural Products; Longman Scientific &
Technical: England, 1994; p 442.
(
6) Inouye, S.; Tsuruoka, T.; Ito, T.; Niida, T. Tetrahedron 1968,
(
29) Arris, J.; Baddiley, J.; Buchanan, J. G.; Thain, E. M. J. Chem.
Soc. 1956, 4968.
24, 2125.
Synthesis 1999, No. 4, 571–573 ISSN 0039-7881 © Thieme Stuttgart · New York

 Matos, Carlos R. R.
Matos, Carlos R. R.
 Lopes, Rosangela S. C.
Lopes, Rosangela S. C.
 Lopes, Claudio C.
Lopes, Claudio C.