
International Journal of Chemical Kinetics p. 359 - 366 (2004)
Update date:2022-08-11
Topics:
 Khan, Zaheer
Khan, Zaheer
 Raju
Raju
 Akram, Mohd
Akram, Mohd
 Kabir-Ud-Din
Kabir-Ud-Din
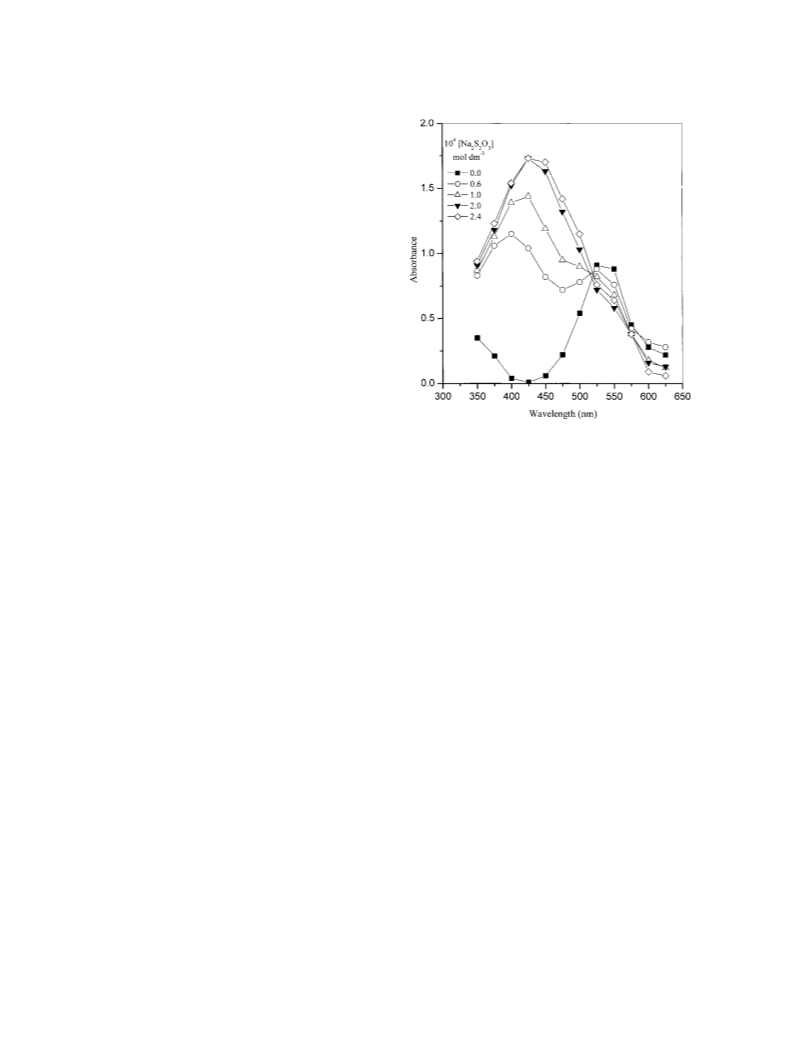
Spectrophotometric method has been used to characterize water-soluble colloidal manganese dioxide obtained by the redox reaction between sodium thiosulphate and potassium permanganate in neutral aqueous medium which shows a single peak in the visible region with λmax = 425 nm. The kinetics of the oxidation of lactic acid by colloidal manganese dioxide (oxidant) has been investigated spectrophotometrically under pseudo-first-order conditions of excess lactic acid. The rate of the noncatalytic reaction pathway was slow which increased with increasing lactic acid concentration. The reaction was first-order with respect to [oxidant] as well as [lactic acid]. In presence of manganase(II) and fluoride ions, the noncatalytic path disappeared completely while the oxidation rate of autocatalytic path increased and decreased, respectively with increasing [Mn(II)] and [F-]. A mechanistic scheme in conformity with the observed kinetics has been proposed with the rate-law: v = -d[MnO2]/dt = κ1κ2[MnO2] [H+][lactic acid]T/ ([H+] Ka)(κ_1 + ≤2)
View More





Hangzhou Dawn Ray Pharmaceutical Co.,Ltd
website:http://www.dawnraypharma.com/
Contact:+86-571-85335020
Address:1018 Guangyin Building,No.42 E.Fengqi Road,Hangzhou,310012 China
Guangxi Nanning Guangtai Agriculture Chemical Co.,Ltd
Contact:+86-771-2311266
Address:Room703,Building12, Software Park Phase II,NO.68,Keyuan Road,Nanning City,Guangxi,China
Chengdu Baishixing Science and Technology Industry Co., Ltd.
website:http://www.cd-bsx.com
Contact:+86-28-88531548
Address:#217,North of Industry Road,Heshan Town,Pujiang County,Chengdu,Sichuan,China.
Henan Techway Chemical Co.,Ltd.
website:http://www.techwaychem.com
Contact:+86-371-66380080
Address:No.27 Shunhe Road,
Shenzhen ZheYi Chemicals Co.,LTD(Shanghai Branch)
Contact:+86-21-54159691
Address:Room1002,Building No.2, Lane 98 Bixiu Road,Minhang District, Shanghai,China 201100
Doi:10.1080/00397919808006830
(1998)Doi:10.1021/ja038672n
(2003)Doi:10.1039/c3ra45787g
(2014)Doi:10.1021/jm401557w
(2013)Doi:10.1021/np5010104
(2015)Doi:10.1039/c4sc03914a
(2015)