
Carbohydrate Research p. 2750 - 2756 (2007)
Update date:2022-08-31
Topics:
 Roncal, Tomas
Roncal, Tomas
 Oviedo, Alberto
Oviedo, Alberto
 de Armentia, Iratxe Lopez
de Armentia, Iratxe Lopez
 Fernandez, Laura
Fernandez, Laura
 Villaran, M. Carmen
Villaran, M. Carmen
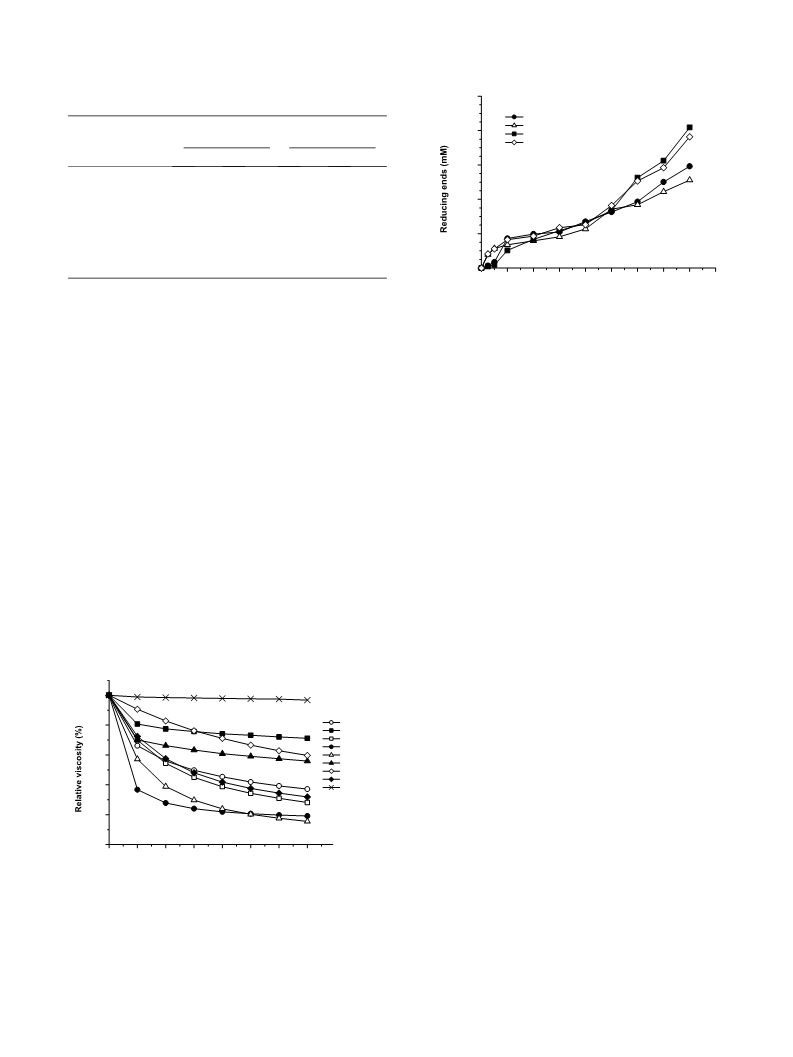
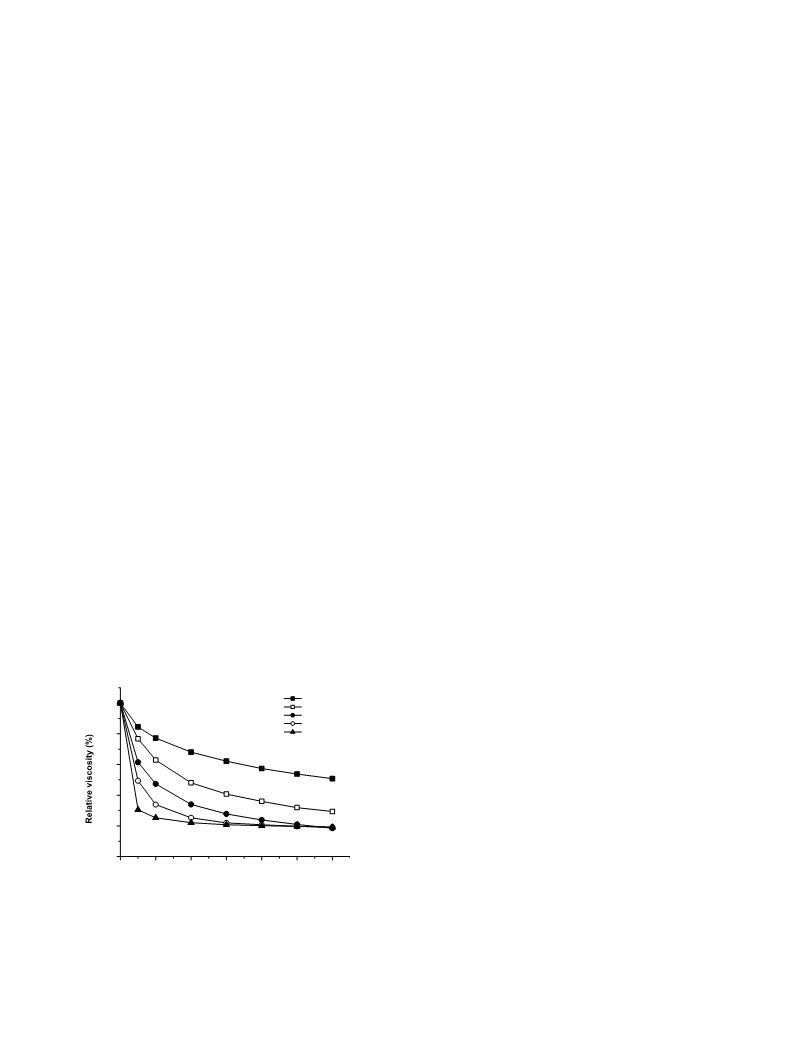
The high molecular weight of chitosan, which results in a poor solubility at neutral pH values and high viscosity aqueous solutions, limits its potential uses in the fields of food, health and agriculture. However, most of these limitations are overcome by chitosan oligosaccharides obtained by enzymatic hydrolysis of the polymer. Several commercial enzymes with different original specificities were assayed for their ability to hydrolyze a 93% deacetylation degree chitosan and compared with a chitosanase. According to the patterns of viscosity decrease and reducing end formation, three enzymes-cellulase, pepsin and lipase A-were found to be particularly suitable for hydrolyzing chitosan at a level comparable to that achieved by chitosanase. Unlike the appreciable levels of both 2-amino-2-deoxy-d-glucose and 2-acetamido-2-deoxy-d-glucose monomers released from chitosan by the other enzymes after a 20 h-hydrolysis (4.6-9.1% of the total product weight), no monomer could be detected following pepsin cleavage. As a result, pepsin produced a higher yield of chitosan oligosaccharides than the other enzymes: 52% versus as much as 46%, respectively. Low molecular weight chitosans accounted for the remaining 48% of hydrolysis products. The calculated average polymerization degree of the products released by pepsin was around 16 units after 20 h of hydrolysis. This product pattern and yield are proposed to be related to the bond cleavage specificity of pepsin and the high deacetylation degree of chitosan used as substrate. The optimal reaction conditions for hydrolysis of chitosan by pepsin were 40 °C and pH 4.5, and an enzyme/substrate ratio of 1:100 (w/w) for reactions longer than 1 h.
View More


Contact:+86-10-62651721
Address:29 Yongxing Road, Daxing District,Beijing China
Heliosense Biotechnologies, Inc.
Contact:+86-592-5667290
Address:Xiamen Torch Hi-tech Zone Venture Weiye Building S506
Jinan Jinguilin Chemical Co.,Ltd
Contact:+86-531-81188412
Address:3rd floor of Torch Building, Huanyuan Rd, City of Jinan
Hangzhou Neway Chemicals Co., Ltd.
Contact:+86-571-85095566
Address:Room 803, Qinglian Bldg, No 139 Qingchun Road, Hangzhou, Zhejiang China
Jiangsu Willing Bio-Tech Co ltd
website:http://www.willingbio.com/
Contact:+86 18796909136
Address:No 18 Guoqiao Road
Doi:10.1007/s13738-013-0296-3
(2014)Doi:10.1246/bcsj.66.1852
(1993)Doi:10.1016/j.jpcs.2010.10.040
(2011)Doi:10.1039/c8nj06508j
(2019)Doi:10.1016/j.apcata.2020.117631
(2020)Doi:10.1016/S0040-4020(99)00474-3
(1999)