2
V.V. Patil et al. / Tetrahedron Letters xxx (2017) xxx–xxx
Previous reports
(Table 1, entry 19). The effect of other bromine sources such as
KBr and NaBr was also investigated under present conditions
(Table 1, entries 20, 21). In both the cases, traces of 2a formation
was observed after 24 h at room temperature.
Gl. acetic acid,
liq. Br2,
reflux 6 h
O
O
NH2
Br
O
O
NH2
After optimizing the solvent and oxidant equivalents we inves-
tigated the effect of other commonly used oxidizing agents on the
bromination of 1a (Table 2). To our surprise, m-CPBA showed
exceptionally high reactivity towards oxidative bromination of
1a. The oxidative bromination of 1a with m-CPBA was completed
within 10 min with 100% conversion at room temperature with
96% selectivity for 2a (Table 2, entry 1). To the best of our knowl-
edge, this is the fastest conversion obtained till date for the bromi-
nation of 1a.
This encourages us to develop another protocol for the same
transformation. Except these two oxidants, other oxidants failed
to give any remarkable results under present conditions (Table 2,
entries 2–9). Next to H2O2 and m-CPBA, Oxone was the only oxi-
dant which gives high selectivity for 2a but required 24 h (Table 2,
entry 9). Thus, after extensive screening of the reaction parame-
ters, the best results obtained for the oxidative bromination of 1a
are 2 equivalents of HBr, 2 equivalents of oxidant (H2O2 as well
as m-CPBA) in methanol at room temperature.
Similar to H2O2-HBr, we also optimized conditions for m-CPBA–
HBr system by taking 1a as substrate. Except methanol, all other
solvents failed to give high selectivity for 2a. The oxidant equiva-
lents study was also performed for 1, 1.5, 1.75 and 2.15 equivalents
of m-CPBA which gives 34%, 56%, 70%, and 97% selectivity respec-
tively for 2a in 10 min. When the other bromine sources such as
KBr and NaBr were used with m-CPBA, a mixture of 2a and 3a were
obtained. The reaction performed with KBr resulted in formation of
54% of 2a and 12% of 3a whereas NaBr gave 50% of 2a and 13% of
3a.
With optimized conditions in hand, a series of substituted
aminoanthracene-9,10-diones were executed by using both the
methods and the results obtained were summarized in Table 3.
All the reactions performed by using H2O2-HBr system required
8 h for completion, while that with m-CPBA-HBr completed within
10 min at room temperature to afford desired products in high
yields. The aminoanthracene-9,10-diones bearing –Cl substituent
1b and 1c (at 1- and at 5-position respectively) gave respective
brominated products in high yields at room temperature under
both the methods (Table 3, entries 2, 3). The substrate 1e which
contains two amino functionalities; viz. unsubstituted and substi-
tuted (-NH2 and –NHPh) afford selective di-bromination at ring
bearing free amino group (Table 3, entry 5). Similarly, the substrate
1g bearing –NH2 and –OCH3 groups gave selectively mono-bromi-
nation at position ortho with respect to –NH2 group under both the
methods (Table 3, entry 7). In order to check the selective mono
bromination of the substrates such as 1a, 1c, 1d and 1e, which gave
di brominated product we performed their reactions by taking 1
equivalent of HBr and 1 equivalent of H2O2 as well as m-CPBA.
But, in all the cases a mixture of mono and di brominated products
was obtained.
90% H2SO4, liq. Br2, I2,
50 °C for 4 h,80 °C for 2 h,
50 °C for 2h
Br
94% H2SO4,
liq. Br2,
100 °C, 10 h
O
NH2
Chlorobenzene,liq. Br2,
60 °C, 24 h
Br
O
O
NH2
98% H2SO4,
Gl. acetic acid,
liq. Br2,60 °C for 5 h,
70-75 °C,
O
O
NH2
Br
100 °C for 9 h
Br
90% H2SO4,
liq. Br2,
50 to 80 °C, 8 h,
then 100 °C for 16 h
O
Present work
R
H2O2
8h
R
R
O
NH
Br
O
O
NH
Br
O
NH
Br
or
HBr
MeOH, rt
-CPBA
m
O
O
10 min
Where, -R= -H
Where, -R=-CH3, -C2H5
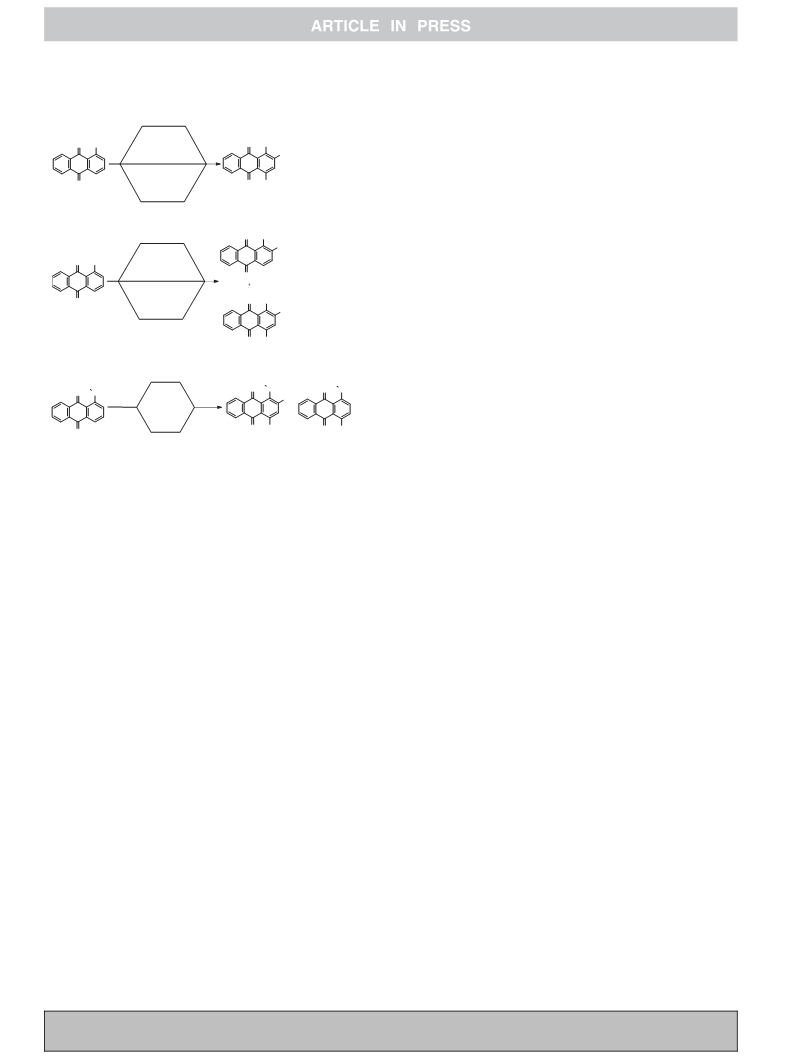
Scheme 1. Previous reports and present work.
consumption of bromine by oxidizing it to bromonium ion which
lack in the traditional bromination protocols. Thus, the oxidative
bromination serve as an efficient approach which overcomes the
shortcomings of the conventional processes used for bromination
of these intermediates.
Thus, in continuation of our previous work to develop efficient
and selective oxidation protocols32–38 herein we report two mild
and easily amenable protocols viz. H2O2-HBr and m-CPBA-HBr for
the oxidative bromination of deactivated aminoanthracene-9,10-
diones in methanol at room temperature. The use of commercially
available entities, such as H2O2, m-CPBA, HBr and methanol at
ambient temperature with easy operational procedure enhances
the commercial feasibility of both the protocols.
Results and discussion
The optimization of reaction parameters was carried out by tak-
ing 1-aminoanthracene-9,10-dione 1a (1 equiv.) as key substrate,
H2O2 (2 equiv.), and HBr (2 equiv.) (Table 1). The solvent screening
study revealed that the bromination of 1a efficiently proceeds in
acetic acid and methanol under present conditions. Acetic acid
gives 97% conversion for 2a (Table 1, entry 2) whereas methanol
gives 95% conversion for 2a within 8 h at room temperature
(Table 1, entry 9). The use of acetic acid was then ruled out as it
gives a mixture of products during bromination of N-alkylated
amine. In all other solvents, mixture of 2a and 3a was obtained
(Table 1, entries 1, 3–7, 9–11). Unlike methanol, other alcohols
failed to give high selectivity for 2a under present conditions
(Table 1, entries 10–16). In the case of ethanol only 23% selectivity
was obtained for 2a but the higher selectivity (77%) was observed
for 3a (Table 1, entry 10).
The regio-selective bromination of N-alkylated aminoan-
thracene-9,10-diones to p-bromo proceeds by using pyridine-
bromine,39 aluminum trichloride-liquid bromine-nitro benzene
system,40 liquid bromine with acetic acid41 or mixture of acetic
acid with propionic acid.42 It was noticeable that the N-alkylated
aminoanthracene-9,10-diones, 1i and 1j undergo regio-selective
bromination under both the conditions at room temperature to
give p-brominated product with excellent yields (Table 2, entries
9, 10). Even when 2 equivalents of HBr and 2 equivalents of H2O2
(as well as m-CPBA) were employed, only mono brominated pro-
duct was obtained in both the methods. The substrate 1i with
H2O2-HBr system gives 86% yield within 8 h while with m-CPBA-
HBr system afford 87% yield of 2i within 10 min (Table 3, entry
The H2O2 equivalents were varied to investigate its effect on the
yield of 2a. It was observed that, the yield of 2a was increased with
increasing the oxidant equivalents from 1.50 to 1.75 equivalents
(Table 1, entries 17, 18) Further increase in the oxidant equivalents
2.15 did not show any significant influence on the yield of 2a

 Patil, Vilas V.
Patil, Vilas V.
 Gayakwad, Eknath M.
Gayakwad, Eknath M.
 Patel, Khushbu P.
Patel, Khushbu P.
 Shankarling, Ganapati S.
Shankarling, Ganapati S.