European Journal of Organic Chemistry
10.1002/ejoc.201700498
COMMUNICATION
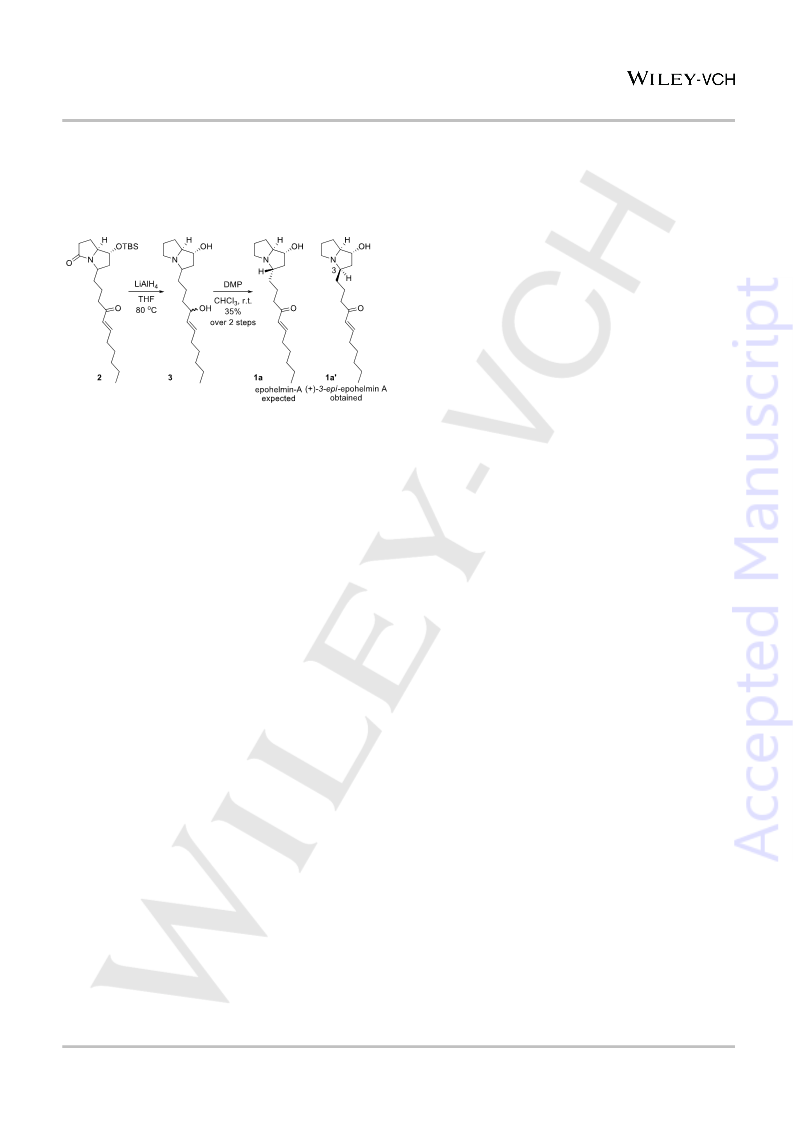
Weinreb amide 5 to obtain the corresponding enone-lactam
intermediate 2. With enone-lactam 2 in hand, we attempted its
A solution of the D-A cyclopropane 8 (690.0 mg, 3.09 mmol), n-
Bu
3
SnH (3.5 mL, 12.37 mmol) and a catalytic amount of AIBN
4
reduction using LiAlH . Interestingly, not only the lactam and the
(
50.0 mg, 0.30 mmol) in benzene (15 mL) was refluxed for 3 h.
enone was reduced, but it also led to deprotection of TBS group
to give the diol 3. To complete the synthesis, the allylic hydroxy
group of diol 3 was chemoselectively oxidized using Dess-Martin
Periodinane (DMP) (Scheme 5).
The reaction mixture was cooled, evaporation of the solvent and
purification of the residue on a silica gel column using ethyl
acetate-pet. ether (1:1) as eluent furnished the pyrrolidone 18
(
680.0 mg, 97%) as a colourless liquid.
Supporting Information (see footnote on the first page of this
article): Experimental procedures and characterization data along
1
13
with copies of the H and C NMR spectra for all new compounds.
Acknowledgements
We thank the SERB, New Delhi, CSIR, New Delhi and MoES,
New Delhi for financial support. We thank Mr. Darshan Mhatre of
the X-ray facility of the Department of Chemistry, IIT Bombay for
collecting the crystallographic data and IRCC, IIT Bombay for
funding central facilities. We are grateful to CSIR, New Delhi, for
the award of research fellowships to LNN.
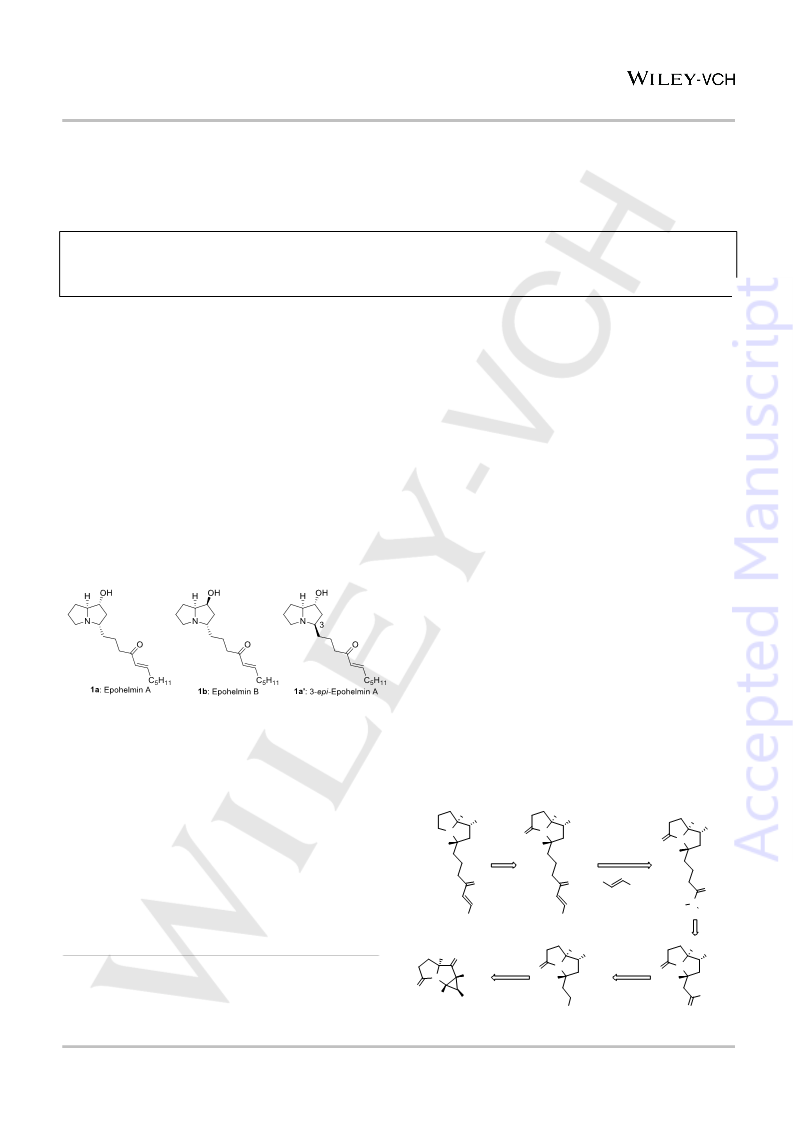
Scheme 5. Synthesis of (+)-3-epi-epohelmin A 1a'
Surprisingly, the data of the obtained enone did not match with
that reported for epohelmin A (1a). Hence, the data was
compared with all other isomers of epohelmin natural products
and its epimers. Careful analysis revealed that the obtained
spectral data was in good agreement with 3-epi-epohelmin A (1a')
Keywords: D-A cyclopropane; epohelmin; pyrrolizidine; lactam;
[
7a]
Weinreb amide
reported by Wei et al.
sequence, the C3 stereocentre underwent epimerization to give
+)-3-epi-epohelmin A (1a'). While exact step in which the
It implies that during the reaction
[
1]
Review on pyrrolizidine alkaloids, see: J. Robertson, K. Stevens, Nat.
Prod. Rep. 2014, 31, 1721.
(
epimerization occurred is not clear at the moment, one can
speculate that this could have happened either during the
oxidation of the alcohol 7 to the aldehyde 6 or during its Wittig
olefination to get amide 21. It is worth noting that the amine moiety
is b to the carbonyl group in the aldehyde 6, so equilibration
through the retro-aza-Michael followed by Michael addition is a
very likely possibility, which might lead to the isomerization of the
C3 stereocentre. Further investigation to ascertain this is on-going
in the laboratory and will be reported in due course.
[2]
For biological activity, see: a) A. Belal, B. El-Dien M, El-Gendy, Bioorg.
Med. Chem. 2014, 22, 46; b) G. Casiraghi, F. Zanardi, G. Rassu, L. Pinna,
Org. Prep. Proc. Int. 1996, 28, 643; c) J. R. Liddell, Nat. Prod. Rep. 2001,
1
8, 441; d) H. Yoda, Curr. Org. Chem. 2002, 6, 223.
[
3]
Recent synthetic methods on pyrrolizidines, see: a) K. Savaspun, C. W.
G. Au, S. G. Pyne, J. Org. Chem. 2014, 79, 4569; b) T. J. Donohoe, R.
E. Thomas, M. D. Cheeseman, C. L. Rigby, G. Bhalay, I. D. Linney, Org.
Lett. 2008, 10, 3615; c) L. Ramabaud, P. Compain, O. R. Martin,
Tetrahedron: Asymmetry 2001, 12, 1807; d) I. Izquierdo, M. T. Plaza, R.
Robles, F. Franco, Tetrahedron: Asymmetry 2002, 13, 1581; e) F.
Cardona, E. Faggi, F. Liguori, M. Cacciarini, A. Goti, Tetrahedron Lett.
2
003, 44, 2315; f) I. Izquierdo, M. T. Plaza, F. Franco, Tetrahedron:
Asymmetry 2003, 14, 3933; g) S. Desvergnes, S. Py, Y. Valee, J. Org.
Chem. 2005, 70, 1459; h) L. Chabaud, Y. Landais, P. Renaud, Org. Lett.
Conclusions
2
005, 7, 2587; i) I. Izquierdo, M. T. Plaza, J. A. Tamayo, F. Sanchaz-
Cantalejo, Tetrahedron: Asymmetry 2007, 18, 2211; j) I. Izquierdo, M. T.
Plaza, J. A. Tamayo, V. Yanez, D. L. Re, F. Sanchaz-Cantalejo,
Tetrahedron 2008, 64, 4613; k) T.-X. Zhang, L. Zhou, X.- P. Cao, Chem.
Res. Chin. Univ. 2008, 24, 469; l) I. Izquierdo, M. T. Plaza, J. A. Tamayo,
F. Franco, F. Sanchaz-Cantalejo, Tetrahedron 2010, 66, 3788; m) I.
Delso, T. Tejero, A. Goti, P. Merino, Tetrahedron 2010, 66, 1220; n) X.-
G. Hu, Y.-M. Jia, J. Xiang, C.-Y. Yu, Synlett 2010, 982; (o) J. A. Tamayo,
F. Franco, F. Sanchez-Cantalejo, Tetrahedron 2010, 66, 7262; p) W.
Zhang, K. Sato, A. Kato, Y. Jia, X. Hu, F. X. Wilson, R. Well, G. Horne,
G. W. J. Fleet, R. J. Nash, C. Yu, Org. Lett. 2011, 13, 4414; q) G.
D’Adamio, A. Goti, C. Parmeggiani, E. Moreno-Clavijo, I. Robina, F.
Cardona, Eur. J. Org. Chem. 2011, 7155; r) T. Sengoku, Y. Satoh, M.
Oshima, M. Takahashi, H. Yoda, Tetrahedron 2008, 64, 8052; s) T.
Sengoku, Y. Satoh, M. Takahashi, H. Yoda, Tetrahedron Lett. 2009, 50,
Nitrogen substituted D-A cyclopropane was synthesized starting
from L-pyroglutamic acid, which was successfully utilized in the
synthesis of (+)-3-epi-epohelmin A (1a’). The key reactions
involved in the synthesis were highly regio- and stereo-selective
cyclopropanation, regioselective ring opening of nitrogen
substituted D-A cyclopropane, stereoselective reduction of ketone
in the ring opening product, organolitium addition to Weinreb
amide and chemoselective oxidation of allylic alcohol. This
particular strategy will also be useful for the synthesis of other
class of pyrrolizidine natural products.
4
937; t) C. Ribes, E. Falomir, J. Murga, M. Carda, J. A. Marco,
Experimental
Tetrahedron 2009, 65, 6965; u) S. Chandrasekhar, B. B. Parida, C.
Rambabu, J. Org. Chem. 2008, 73, 7826; v) X.-K. Liu, S. Qiu, Y.-G. Xiang,
Y.-P. Ruan, X. Zheng, P.-Q. Huang, J. Org. Chem. 2011, 76, 4952; w) E.
A. Brock, S. G. Davies, J. A. Lee, P. M. Roberts, J. E. Thomson, Org.
Biomol. Chem. 2013, 11, 3187.
Synthesis of Ethyl 2-((3R,7aS)-1,5-dioxohexahydro-1H-
pyrrolizin-3-yl)acetate (18):
[
4]
For isolation, see: M. Shibuya, B. B. Snider, Y. Sakano, H. Tomoda, S.
Omura, Y. Ebizuka, J. Antibiot. 2005, 58, 599.
This article is protected by copyright. All rights reserved.

 Gharpure, Santosh J.
Gharpure, Santosh J.
 Nanda, Laxmi Narayan
Nanda, Laxmi Narayan
 Kumari, Dimple
Kumari, Dimple