Angewandte
Chemie
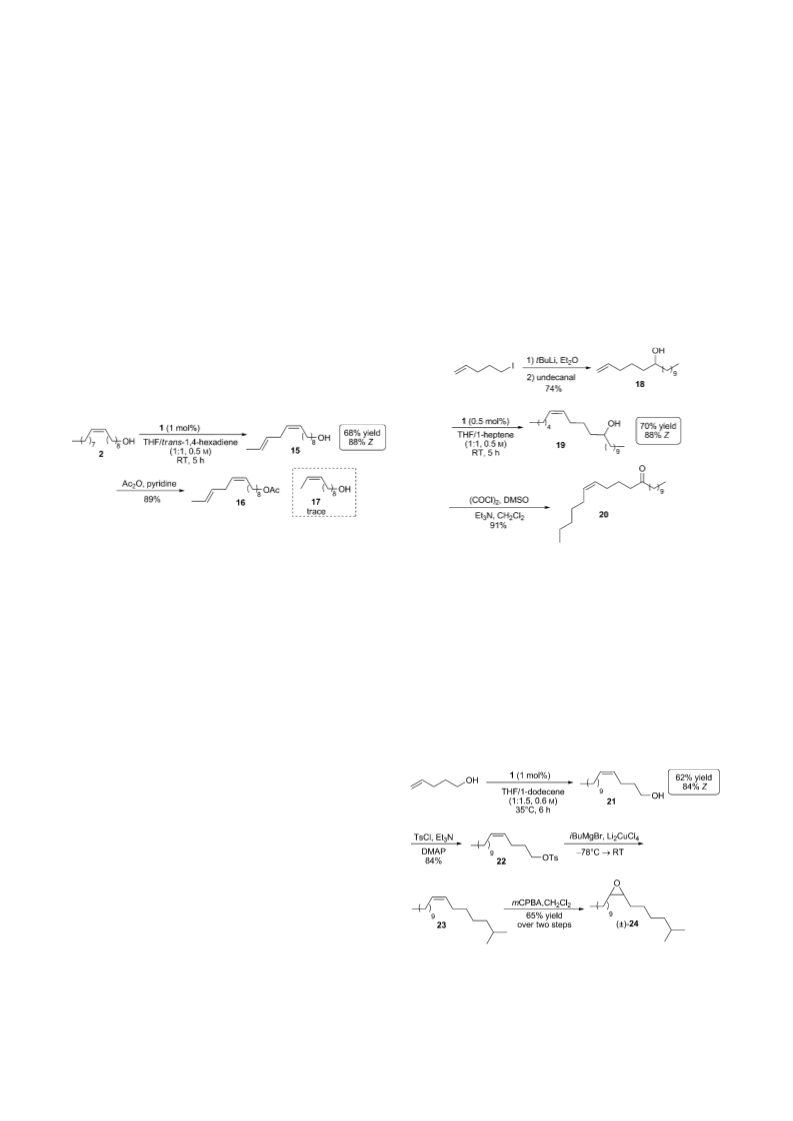
dodecene afforded 21 in moderate yield (62%) and high
Z selectivity (84%).[21] Alcohol 21 was tosylated and reacted
with isobutylmagnesium bromide to produce the desired alkyl
intermediate 23.[22] Subsequent epoxidation of 23 with
mCPBA gave (Æ)-24 in four steps from commercially
available starting materials.
[1] D. Pimintel, Chem. Br. 1991, 27, 646.
[3] a) S. C. Welter, C. Pickel, J. Millar, F. Cave, R. A. Van Steenwyk,
J. Dunley, J. Calif. Agric. 2005, 59, 17; b) P. E. Howse, I. D. R.
Stevens, O. T. Jones, Insect Pheromones and their Use in Pest
Management, Chapman & Hall, New York, 1998, pp. 314 – 338.
[4] R. L. Pederson, I. M. Fellows, T. A. Ung, H. Ishihara, S. P.
[5] The syntheses of alkenyl alcohols, acetates, and aldehydes
containing a Z olefin using classical approaches have been
widely reported: a) E. A. Petrushkina, V. N. Kalinin, Russ. J.
b) B. K. Keitz, K. Endo, M. B. Herbert, R. H. Grubbs, J. Am.
[7] Separation of E isomers from the desired Z isomers was beyond
the scope of this paper, however, the undesired E pheromone is
generally inert for pest control applications. If isolation of the
pure Z isomer is necessary, chromatography with silver nitrate
has been shown to be effective at this type of separation: C. M.
[8] a) U. Biermann, E. Bornscheuer, M. A. R. Meier, J. O. Metzger,
[10] See the Supporting Information for NMR conditions.
[11] Alternative methods, including pressurization of the reaction
vessel with 1-butene, and a slow stream of 1-butene into the
headspace of the reaction vessel, resulted in highly reduced
yields and Z selectivity. The cause of this reduced Z selectivity is
not known, but will be investigated in due course.
[12] A small amount of terminal olefin products derived from
ethenolysis of the seed-oil derivatives was generated during the
corresponding reaction with 1-hexene as well. The reaction of 9-
decenol with 1-butene to produce 7 produced analogous yields
and Z selectivities when compared to the reaction of oleyl
alcohol and 1-butene.
Current industrial syntheses of (Æ)-24 similarly proceed
through epoxidation of alkyl intermediate 23, which can be
formed either by Wittig or acetylenic routes.[5h] Use of the
Wittig reaction on an industrial scale is generally difficult
because of the large amount of phosphine oxide by-products
that must be removed, and problems cooling the reaction
mixture enough to promote adequate cis selectivity. Still, (Æ)-
24 can be formed in five steps with 60% overall yield and
88% Z olefin using this route. The industrial-scale synthesis
of (Æ)-24 using the poisoned hydrogenation of a disubstituted
acetylene can be completed in four overall steps with 98%
Z olefin. However, on an industrial scale, forming disubsti-
tuted acetylenes can be expensive and requires the use of
large amounts of liquid ammonia. In addition, temperature
control can be a problem and lead to incomplete reduction,
and migration of double bonds and isomerization can occur,
thus leading to impurities. The use of metathesis as the key
step to form (Æ)-24 is attractive because cheap a-olefins can
be used, unreacted starting material can be recycled, and all
reactions can be run at mild temperatures. It is envisioned
that all of the synthetic routes outlined above can be adapted
for large-scale syntheses.
In summary, the facile synthesis of nine lepidopteran
female sex pheromones has been achieved using ruthenium-
based Z-selective olefin metathesis. These pheromones are
approved by the EPA as pest control agents, however, it is
conceived that other analogous pheromones can be synthe-
sized in a similar manner, further promoting their use as
insecticide alternatives. The synthesis of these compounds
provides valuable insight into the reactivity and selectivity of
catalyst 1, which is markedly distinct from previous gener-
ations of metathesis catalysts. The development of new Z-
selective catalysts and operating on a larger scale should
make this methodology even more selective and efficient.
Compounds containing a variety of functional groups, includ-
ing alcohols, acetates, aldehydes, ketones, and epoxides were
easily prepared in a minimal number of steps from commer-
cial sources, including several seed-oil derivatives; the
syntheses of all pheromones described above are the most
concise to date. It has been demonstrated that ruthenium-
based Z-selective metathesis provides an attractive route to
form complex internal olefins in good yields with high cis
selectivity, and could emerge as a viable alternative to other
popular methods used to form cis olefins, such as the partial
hydrogenation of alkynes and the Wittig reaction.
[13] A much larger amount of the undesired homodimer was
detected for reaction of 8-nonenyl acetate compared to 8-
nonenol.
[14] a) G. Hornyꢂnszky, J. Rohꢂly, L. Novꢂk, Synth. Commun. 2008,
38, 1533; b) A. Ortiz, A. Quesada, A. Sanchez, J. Chem. Ecol.
2004, 30, 991; c) E. D. Matveeva, A. S. Erin, I. F. Leshcheva,
A. L. Kurts, Russ. J. Org. Chem. 2000, 36, 765.
[16] The Z-selective homodimerization of conjugated 1,3-dienes was
recently reported for molybdenum and tungsten olefin meta-
thesis catalysts: E. M. Townsend, R. R. Schrock, A. H. Hoveyda,
Received: July 30, 2012
Published online: October 10, 2012
[17] This phenomenon was first reported for a Z-selective molybde-
num metathesis catalyst: S. C. Marinescu, D. S. Levine, Y. Zhao,
Keywords: insect pheromones · metathesis · ruthenium ·
seed-oil derivatives · unconjugated dienes
.
Angew. Chem. Int. Ed. 2013, 52, 310 –314
ꢀ 2013 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
313

 Herbert, Myles B.
Herbert, Myles B.
 Marx, Vanessa M.
Marx, Vanessa M.
 Pederson, Richard L.
Pederson, Richard L.
 Grubbs, Robert H.
Grubbs, Robert H.