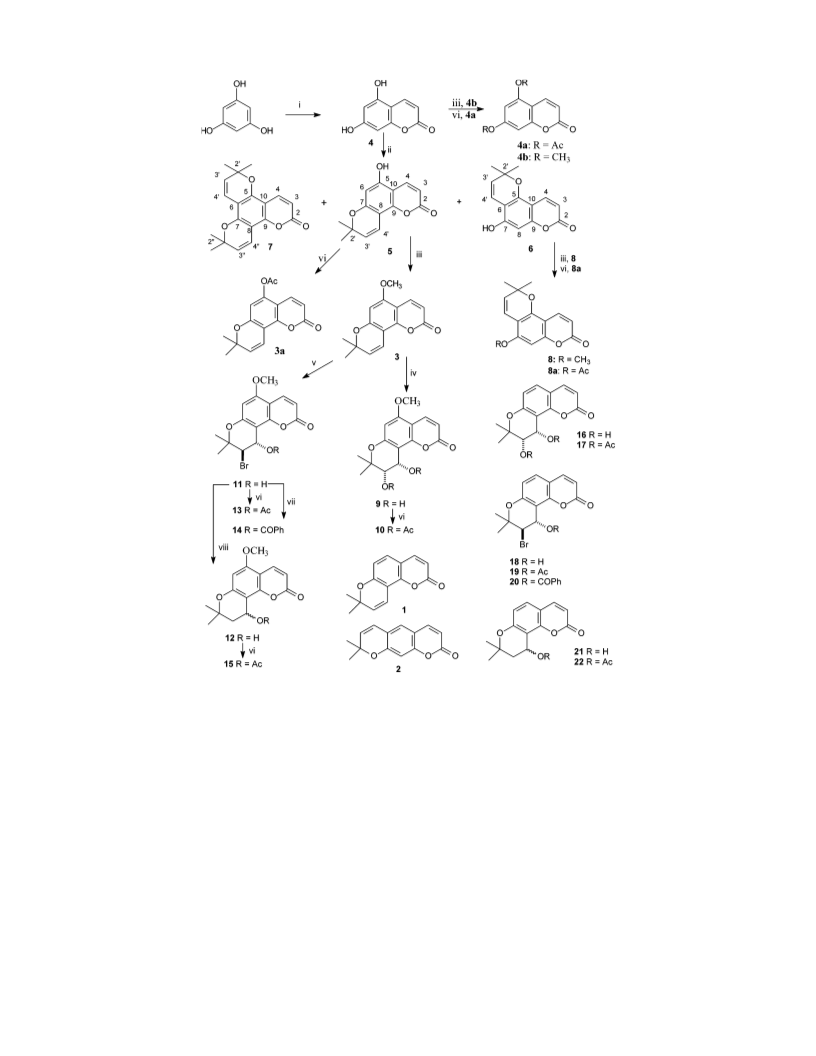
2,2-Dimethylpyranocoumarins
Journal of Natural Products, 2005, Vol. 68, No. 1 81
ref 10; 13C NMR (CDCl3, 50 MHz) δ 161.9 (C-2), 157.7 (C-7),
157.2 (C-5), 156.0 (C-9), 140.0 (C-4), 110.5 (C-3), 104.3 (C-8,-
10), 96.0 (C-6), 79.5 (C-2′), 71.7 (C-3′), 61.4 (C-4′), 56.4 (OCH3),
25.7 (CH3), 21.9 (CH3); MS-DCI m/z 293 (M + H)+.
m/z 461, 459 (M + H)+; anal. C 57.65%, H 4.21%, Br 17.55%
calcd for C22H19BrO6, C 57.53%, H 4.17%, Br 17.40%.
(()-4′-Hydroxy-3′,4′-dihydro-5-methoxyseselin (12). Com-
pound 11 (78 mg, 0.22 mmol) was dissolved in anhydrous
toluene (10 mL), and the solution was refluxed for 15 min
under argon. Then AIBN (azo-bis-2,2′-(methyl-2-propionitrile))
(10 mg) was added, and after 5 min, a solution of tributyltin
hydride (0.5 mL in 4 mL of toluene) was added over a period
of 40 min. The reaction mixture was refluxed for 1 h. The
solvent was evaporated and the residue was purified by flash
chromatography on silica gel with cyclohexane-EtOAc (80:
20) to give compound 12 (35 mg, 59%): UV (CHCl3) λmax (log
ꢀ) 330 (3.90), 261 (3.66), 251 (3.60) nm; IR (CHCl3) νmax 3350,
1630, 1604, 1220 cm-1; 1H NMR (CDCl3, 400 MHz) δ 8.02 (1H,
d, J ) 9.8 Hz, H-4), 6.24 (1H, s, H-6), 6.18 (1H, d, J ) 9.8 Hz,
H-3), 5.20 (1H, m, H-4′), 3.89 (3H, s, OCH3), 3.27 (1H, d, J )
3.0 Hz, OH), 2.12 (1H, d, J ) 4.9, H-3′), 1.51 (3H, s, CH3),
1.43 (1H, s, CH3); 13C NMR (CDCl3, 50 MHz) δ 161.4 (C-2),
157.8 (C-7), 156.7 (C-5), 139.4 (C-4), 109.9 (C-3), 104.2 (C-8),
104.1 (C-10), 95.7 (C-6), 77.0 (C-2′), 59.0 (C-4′), 55.9 (OCH3),
40.2 (C-3′), 28.0 (CH3), 26.5 (CH3); MS-DCI m/z: 277 (M +
H)+; anal. C 65.33%, H 5.76%, calcd for C15H16O5, C 65.21%,
H 5.84%.
(()-cis-3′,4′-Diacetoxy-3′,4′-dihydro-5-methoxyseselin
(10). Treatment of 9 (24 mg, 0.08 mmol) in conditions similar
to those described for the preparation of 4a afforded 10 (28
mg, 92%): UV (CHCl3) λmax (log ꢀ) 344 (sh), 326 (4.15), 259
(3.95), 249 (3.92) nm; IR (CHCl3) νmax 1747, 1729 (sh),1629,
1
1603, 1236 cm-1; H NMR (CDCl3, 200 MHz) δ 7.92 (1H, d, J
) 9.5 Hz, H-4), 6.44 (1H, d, J ) 5.0 Hz, H-4′), 6.22 (1H, s,
H-6), 6.13 (1H, d, J ) 9.5 Hz, H-3), 5.25 (1H, d, J ) 5.0 Hz,
H-3′), 3.86 (3H, s, OCH3), 2.20 (3H, s, COCH3), 2.09 (6H, s,
COCH3), 1.42 (3H, s, 2×CH3); 13C NMR (CDCl3, 50 MHz) δ
171.0 (COCH3), 161.9 (C-2), 157.7 (C-5), 157.2 (C-7), 156.0 (C-
9), 139.7 (C-4), 112.8 (C-3), 104.3 (C-10), 100.0 (C-8), 96.7 (C-
6), 78.5 (C-2′), 71.5 (C-3′), 62.3 (C-4′), 57.3 (OCH3), 26.4 (CH3),
25.4 (CH3), 21.8 (COCH3); MS-DCI m/z 377 (M + H)+; anal. C
60.75%, H 5.39%, calcd for C19H20O8, C 60.64%, H 5.36%.
(()-trans-3′-Bromo-4′-hydroxy-3′,4′-dihydro-5-methoxy-
seselin (11). To a solution of 3 (140 mg, 0.54 mmol) in THF
(5 mL) and H2O (5 mL) was added N-bromosuccinimide (100
mg, 0.56 mmol). The reaction mixture was stirred for 1 h at 0
°C, and then the reaction mixture was extracted with NaCl
(sat.)-Et2O and the organic layer was collected. The solvent
was removed under reduced pressure, and compound 11 was
purified by crystallization with Et2O (110 mg, 57%): UV
(CHCl3) λmax (log ꢀ) 331 (3.99), 261 (3.75), 251 (3.71) nm; IR
(()-4′-Acetoxy-3′,4′-dihydro-5-methoxyseselin (15). Treat-
ment of compound 12 (23 mg, 0.08 mmol) in conditions
essentially similar to those described for the preparation of
4a afforded 15 (24 mg, 92%): UV (CHCl3) λmax (log ꢀ) 345 (sh),
329 (4.02), 261 (3.77), 251 (3.74) nm; IR (CHCl3) νmax 1732,
1630, 1604, 1220 cm-1; 1H NMR (CDCl3, 400 MHz) δ 7.98 (1H,
d, J ) 9.7 Hz, H-4), 6.24 (1H, s, H-6), 6.23 (1H, t, J ) 4.9,
H-4′), 6.17 (1H, d, J ) 9.7, H-3), 3.90 (3H, s, OCH3), 2.12 (2H,
1
(CHCl3) νmax 3350, 1630, 1605, 1225 cm-1; H NMR (DMSO,
200 MHz) δ 8.02 (1H, d, J ) 9.7 Hz, H-4), 6.28 (1H, s, H-6),
6.20 (1H, d, J ) 9.7 Hz, H-3), 5.35 (1H, d, J ) 5.1 H-4′), 4.25
(1H, s, br, OH), 4.29 (1H, d, J ) 5.1, H-3′), 3.90 (3H, s, OCH3),
1.65 (3H, s, CH3), 1.55 (3H, s, CH3); 13C NMR (DMSO, 50 MHz)
δ 161.6 (C-2), 157.3 (C-5), 156.9 (C-7), 155.2 (C-9), 139.9 (C-
4), 110.9 (C-3), 104.9 (C-10), 104.4 (C-8), 96.0 (C-6), 79.2 (C-
2′), 67.2 (C-4′), 58.2 (C-3′), 56.4 (OCH3), 26.5 (CH3), 25.3 (CH3);
MS-DCI m/z 357, 355 (M + H)+; anal. C 50.61%, H 4.19%, Br
22.45% calcd for C15H15BrO5, C 50.72%, H 4.26%, Br 22.50%.
(()-trans-3′-Bromo-4′-acetoxy-3′,4′-dihydro-5-methoxy-
seselin (13). Treatment of 11 (35 mg, 0.10 mmol) in conditions
essentially similar to those described for the preparation of
4a afforded 13 (36 mg, 92%): UV (CHCl3) λmax (log ꢀ) 344 (sh),
327 (4.11), 260 (3.90), 251 (3.82) nm; IR (CHCl3) νmax 1733,
1631, 1604, 1220 cm-1; 1H NMR (CDCl3, 400 MHz) δ 7.95 (1H,
d, J ) 9.7 Hz, H-4), 6.44 (1H, d, J ) 4.2 Hz, H-4′), 6.28 (1H,
s, H-6), 6.18 (1H, d, J ) 9.7, H-3), 4.33 (1H, d, J ) 4.2 Hz,
H-3′), 3.90 (3H, s, OCH3), 2.15 (3H, s, COCH3), 1.59 (6H, s,
2×CH3); 13C NMR (CDCl3, 50 MHz) δ 170.4 (COCH3), 160.6
(C-2), 157.7 (C-5), 157.6 (C-7), 155.4 (C-9), 138.9 (C-4), 111.8
(C-3), 104.9 (C-10), 98.5 (C-8), 96.1 (C-6), 78.3 (C-2′), 67.4 (C-
4′), 56.4 (OCH3), 54.3 (C-3′), 26.4 (2×CH3), 21.3 (COCH3); MS-
DCI m/z 397, 395 (M + H)+; anal. C 51.41%, H 4.27%, Br
20.22% calcd for C17H17BrO6, C 51.40%, H 4.31%, Br 20.12%.
(()-trans-3′-Bromo-4′-benzoyloxy-3′,4′-dihydro-5-meth-
oxyseselin (14). To a solution of 11 (28 mg, 0.08 mmol) in
dry pyridine (1.5 mL) was added benzoic anhydride (62 mg,
0.29 mmol). The reaction mixture was stirred for 48 h at room
temperature, and the reagents were removed under reduced
pressure. The residue mixture was extracted with EtOAc-
NaHCO3 (sat.), and the organic layer was collected. The solvent
was removed under reduced pressure, and the remaining
residue was purified by flash chromatography on silica gel with
cyclohexane-CH2Cl2 (1:1) to give compound 14 (23 mg, 63%):
UV (CHCl3) λmax (log ꢀ) 346 (sh), 329 (4.10), 297 (sh), 284 (sh),
258 (3.69), 242 (3.98) nm; IR (CHCl3) νmax 1736, 1629 (sh),
1608, 1604, 1220 cm-1; 1H NMR (CDCl3, 200 MHz) δ 8.02 (2H,
d, J ) 7.5, H-2′′,6′′), 7.98 (1H, d, J ) 9.5, H-4), 7,57 (1H, t, J
) 7.5 Hz, H-4′′), 7.43 (2H, t, J ) 7.5 Hz, H-3′′,5′′), 6.70 (1H, d,
J ) 2.9, H-4′), 6.36 (1H, s, H-6), 6.16 (1H, d, J ) 9.5, H-3),
4.53 (1H, d, J ) 2.9, H-3′), 3.94 (3H, s, OCH3), 1.67 (3H, s,
CH3), 1.66 (3H, s, CH3); 13C NMR (CDCl3, 50 MHz) δ 165.6
(COPh-9), 160.7 (C-2), 158.0 (C-5), 157.8 (C-7), 155.6 (C-9),
133.7 (C-4′′), 130.3 (C-2′′,6′′), 129.8 (C-1′), 129.5 (C-3′′,5′′), 111.5
(C-3), 104.5 (C-10), 97.8 (C-8), 95.5 (C-6), 77.6 (C-2′), 67.3 (C-
4′), 56.1 (OCH3), 53.5 (C-3′), 28.4 (CH3), 24.7 (CH3); MS-DCI
d, J ) 4.9, H-3′), 2.12 (3H, s, OCOCH3), 1.46 (1H, s, CH3); 13
C
NMR (CDCl3, 50 MHz) δ 170.8 (COCH3), 161.3 (C-2), 158.0
(C-7), 157.6 (C-5), 155.3 (C-9), 138.6 (C-4), 110.8 (C-3), 104.1
(C-10), 100.6 (C-8), 95.5 (C-6), 76.6 (C-2′), 61.3 (C-4′), 55.9
(OCH3), 38.4 (C-3′), 28.7 (CH3), 25.8 (CH3), 21.1 (COCH3); MS-
DCI m/z 319 (M + H)+; anal. C 64.09%, H 5.66%, calcd for
C17H18O6, C 64.14%, H 5.70%.
Antibacterial Activity. Antibacterial activities of com-
pounds were determined using the diffusion technique of
Bauer-Kirby (disk method) by measuring the zone of inhibi-
tion against two Gram-positive bacteria, Staphylococcus au-
reus (ATCC 25923) and Staphylococcus epidermidis (ATCC
12228), and five Gram-negative bacteria, Pseudomonas aerugi-
nosa (ATCC 27853), Escherichia coli (ATCC 25922), Entero-
bacter cloacae (ATCC 13047), Klebsiella pneumoniae (ATCC
13883), and Proteus mirabilis. Netilmicin, amoxycillin, and
clavulanic acid were used as control antibiotics. The results
were reported as the diameter of the zone of inhibition around
each disk (in mm), and evaluation of inhibition corresponds
to <7 mm (-), 7-10 mm (+), 11-16 mm (++), >16 mm (+++).
The compounds were dissolved in DMSO. For each experiment
control disks with solvent were used as negative control. All
paper disks had a diameter of 6 mm and were deposited on
the surface of the seeded trypticase Muller-Hinton agar. Petri
dishes were previously inoculated with the organisms to give
a final cell concentration of 107 cell/mL. Volumes of 10 µL of
the above solutions were required to wet the test paper disks.
The incubation conditions used were 24 h at 37 °C. The
experiments were repeated three times, and the results were
expressed as average values. All strains were standard refer-
ence strains (American Type Culture Collection).
References and Notes
(1) Murray, R. D. H. In Progress in the Chemistry of Organic Natural
Products; Herz, W., Falk, H., Kirby, G. W., Moore, R. E., Eds.;
Springer-Verlag: Wien, 2002; Vol. 83, pp 1-529.
(2) Magiatis, P.; Melliou, E.; Skaltsounis, A. L.; Mitaku, S.; Le´once, S.;
Renard, P.; Pierre´, A.; Atassi, G. J. Nat. Prod. 1998, 61, 982-986.
(3) Xie, L.; Takeuchi, Y.; Cosentino, L. M.; Lee, K.-H. J. Med. Chem. 1999,
42, 2662-2672.
(4) Kayser, O.; Kolodziej, H. Z. Naturforsch. 1999, 54c, 169-174.
(5) Magiatis, P.; Mitaku, S.; Pierre´, A.; Atassi, G. Heterocycles 2002, 57,
341-352.
(6) Melliou, E.; Magiatis, P.; Mitaku, S.; Skaltsounis, A. L.; Pierre´, A.;
Atassi, G.; Renard, P. Bioorg. Med. Chem. 2001, 9, 607-612.

 Melliou, Eleni
Melliou, Eleni
 Magiatis, Prokopios
Magiatis, Prokopios
 Mitaku, Sofia
Mitaku, Sofia
 Skaltsounis, Alexios-Leandros
Skaltsounis, Alexios-Leandros
 Chinou, Efrosini
Chinou, Efrosini
 Chinou, Ioanna
Chinou, Ioanna