324
J.B. Wu et al. / Journal of Alloys and Compounds 431 (2007) 321–325
undergoes a dissolving process resulting in blue Co(II) complex
ion in alkaline electrolyte. These Co(II) complex ions precipitate
on -Ni(OH)2 particles as -Co(OH)2 while standing and then
converted to -CoOOH conductive network after first charging
process. The standing procedure, as Oshitani reported [4], will
last several days for CoO to dissolve thoroughly. The nanoscale
CoO is of advantage to dissolve completely due to its tiny
size. Therefore a more integrated conductive network can be
fabricated after the first charge in the positive electrode with
nanoscale CoO. This integrating network improved the real reac-
tion surface and the utility of the active material -Ni(OH)2, thus
the specific capacity was improved subsequently.
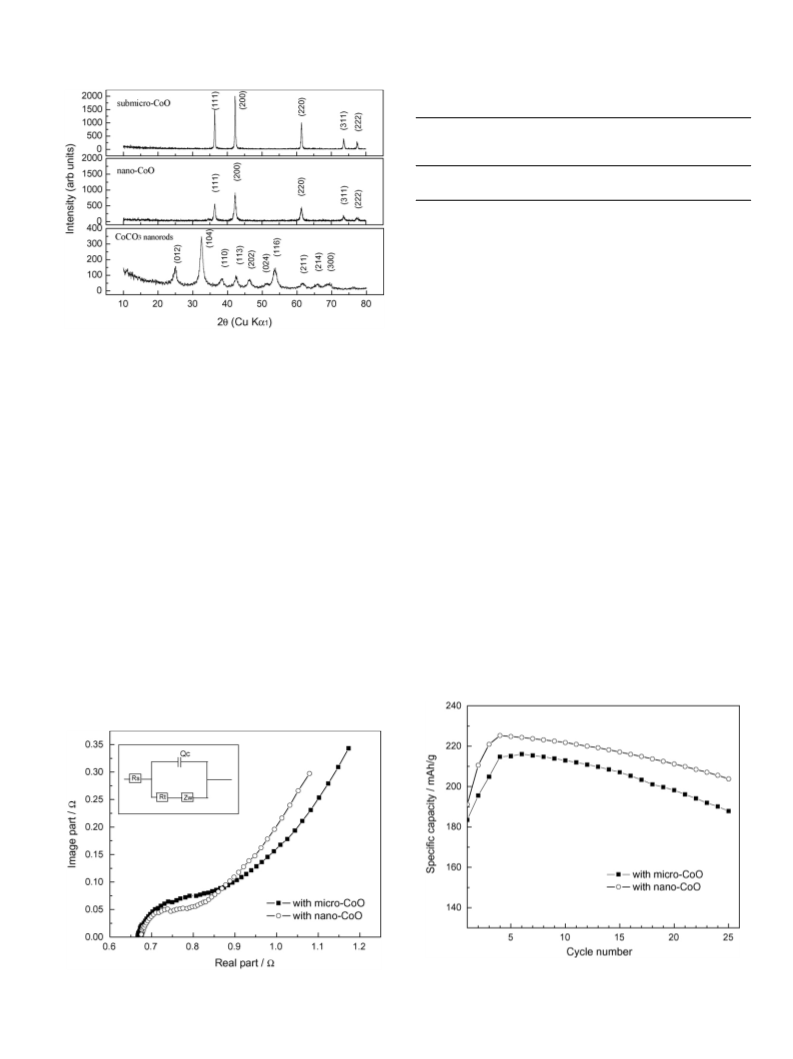
It also can be seen in Fig. 4 that the specific capacity of the
with nanoscale CoO. It was reported that the open circuit voltage
of the batteries is mainly related with the RS in the equivalent
with the RT [11]. Lower is RT of the electrode with nanoscale
CoO, more slightly is the deterioration of the real reaction sur-
face, and more slightly is the decay of the specific capacity.
Fig. 5 shows the typical charge–discharge profiles of the elec-
trodes discharged at 0.1 C. The electrode with nanoscale CoO
had lower charge voltage plateau and higher discharge voltage
plateau than that with usual submicron CoO. It attributes to the
homogenous conductive network transformed from nanoscale
CoO. It is known that the voltage of the electrodes lies on total
polarization, and the high total polarization leads to low load
voltage. The integrated conductive network helps to remove the
charge efficiently and diminish the polarization, which lowers
the charge voltage plateau and improves the discharge voltage.
Higher discharge voltage plateau means more energy the elec-
trodes can release, and it helps to improve the power of the
batteries.
Fig. 6. Rate discharge capability of the two groups of batteries discharged at
different current rates (1 C = 2200 mA).
teries with submicron CoO, which presents the rate capability
of 93.7%. Followed with the increase of discharge rate, the rate
capability of the two kinds of batteries both decreased. Com-
pared to the batteries with nanoscale CoO, the batteries with
submicron CoO deteriorated more seriously. Especially at 10 C
the rate capability only remains 77% for the batteries with sub-
micron CoO, which is remarkably lower than 89% for the other
group of batteries.
Because two groups of batteries have the same negative elec-
trodes, distinguish of the rate discharge capability attributes to
the positive electrodes. As we know the discharge kinetics of
Ni(OH)2 electrode is contributed both from proton diffusion and
from charge-transfer owing to the rapid discharge process [1].
According to the former analysis, a superior conductive network
transformed from nanoscale CoO is formed as current collector
between the active materials and the nickel substrate. Though
the network has no effect on the proton diffusion process which
depends mainly on the sphere sizes and the lattice structure of
-Ni(OH)2, it assures the fluent transfer of charges. Therefore,
the addition of nanoscale CoO improves the discharge kinetics
under high-rate discharge condition and it is of advantage to the
discharged capacity of Ni/MH batteries even at 10 C.
The rate discharge capability of the sealed batteries with two
kinds of positive electrode, respectively, was presented in Fig. 6.
ThebatterywithnanoscaleCoOinthepositiveelectrodeexhibits
rate capability of 95.4% at 1 C. It is slightly higher than the bat-
4. Conclusion
By adding dispersive nanoscale CoO instead of usual sub-
micron CoO to the positive electrodes of Ni/MH batteries, a
more integrated conductive network of -CoOOH can be formed
between the active materials and the substrate. As a result, the
ohmic resistance of the electrode and the impedance of the
electrochemical reaction are decreased. Thus-prepared Ni(OH)2
electrode has higher specific capacity, high discharge voltage
plateau and deteriorates more slightly. The sealed batteries with
nanoscale CoO in the positive electrode exhibit a modified rate
discharge capability. Compared with usual submicron CoO, the
nanoscale CoO is better as additive considering the overall per-
formance of the Ni/MH rechargeable batteries.
Fig. 5. Charge/discharge profiles of Ni(OH)2 electrodes with nanoscale and
submicron CoO at 0.1 C rate.

 Wu
Wu
 Tu
Tu
 Zhang
Zhang
 Huang
Huang