Journal of Materials Chemistry A
Paper
high-performance cathodes reported;30 in this case, the volume
ratio of BSSC4224 in this composite is almost the threshold
value for the charge carrier conduction. Thus, it was suggested
that the triple phase boundary of the BSSC4224/BCPY/gas phase
is the electrochemically active reaction site and BSSC4224
serves as the active site for the dissociative adsorption of
oxygen. However, the design guide for this composite system
has not been established yet. It is required to verify the validity
of the proposed electrode microstructure as well as the reaction
mechanism. Moreover, this composite system has a potential to
achieve higher ORR activity by modifying the chemical
composition, optimizing the fabrication process, changing the
mixing materials, etc. In this study, then, the effect of elemental
substitution, especially the Ba/Sr ratio and rare-earth elements,
in (Ba,Sr)6RE2Co4O15 and the composite effect with BCPY on the
electrocatalytic activity were studied. We also focused on
elemental interdiffusion in (Ba,Sr)6RE2Co4O15–Ba(Ce,Pr,Y)O3
composites to clarify the reason for the high ORR activity.
Cell preparation
The symmetric cells consisting of various electrodes and a BCY
disk electrolyte were fabricated as follows. The BCY powder was
uniaxially pressed at 40 MPa into a pellet, followed by cold
isostatic pressing at 300 MPa. The obtained pellet was red at
1600 ꢁC for 10 h in air, and subsequently polished to be 1 mm in
thickness. The diameter of the pellet was ca. 19 mm. The rela-
tive density of the sintered BCY disk was higher than 98%. Some
powders were mechanically mixed to form composites with
varying the weight ratio. The slurries of electrodes were
prepared by adding polyethylene glycol (molecular weight 400,
Wako Pure Chemical Industries, Ltd.) to electrode powders. The
slurry was screen-printed onto both sides of the BCY electrolyte,
and then red at 1000–1200 ꢁC for 5 h in air. The diameter and
thickness of the electrodes were 6 mm and ca. 20 mm, respec-
tively. The electrode microstructure observation was performed
by using a dual-beam focused ion beam-scanning electron
microscope (FIB-SEM, NVision 40, Carl-Zeiss-SIINT) equipped
with an EDX (Oxford).
Experimental
Electrochemical measurements
Powder preparation and characterization
The symmetrical cells were used to evaluate the ORR activity.
The electrode performance was measured in 3 vol% humidied
synthetic air (21% O2–79% N2) at 450–700 ꢁC by impedance
spectroscopy (CellTest system; potentiostat/galvanostat 1470E
and frequency response analyzer 1455, Solartron Analytical,
UK). The applied frequency was in the range of 0.1 Hz to 105 Hz
with a voltage amplitude of 10 mV. The humidication was
conducted by bubbling the gas through the water. Note that the
electrode performance included the inuence of the electronic
leakage current because of the usage of symmetrical cells with
a BCY electrolyte in the oxidizing atmosphere. However, the
difference in the electrode performance obtained can be
compared fairly. This is because the experimental conditions,
such as the electrolyte thickness and the atmosphere, were xed
throughout this study. The electrode design concept obtained
in this study can also be applied to cells using other electrolytes,
such as BaZrO3-based oxides.
The powders of (Ba6ꢀxSrx)RE2Co4O15 (x ¼ 1–3, RE ¼ La, Pr, Nd,
Sm, Gd), BaCe0.5Pr0.3Y0.2O3ꢀd (BCPY), BaCe0.5ꢀxCoxPr0.3Y0.2O3ꢀd
(BCCPYx, x ¼ 0.05–0.25), and BaCe0.8Y0.2O3ꢀd (BCY) were
synthesized by the Pechini method. Stoichiometric amounts of
starting reagents were dissolved into distilled water: Ba(NO3)2
(Wako Pure Chemical Industries, Ltd.), Sr(NO3)2 (Wako Pure
Chemical Industries, Ltd.), Co(NO3)2$6H2O (Wako Pure Chem-
ical Industries, Ltd.), Ce(NO3)3$6H2O (Wako Pure Chemical
Industries, Ltd.), Pr(NO3)3$6H2O (Sigma-Aldrich, Co.),
Y(NO3)3$6H2O (Sigma-Aldrich, Co.), Sm(NO3)3$6H2O (Wako
Pure Chemical Industries, Ltd.), Gd(NO3)3$6H2O (Wako Pure
Chemical Industries, Ltd.), Nd(NO3)3$6H2O (Wako Pure
Chemical Industries, Ltd.), and La (NO3)3$6H2O (Wako Pure
Chemical Industries, Ltd.). Citric acid (C6H8O7$H2O, Wako Pure
Chemical Industries, Ltd.) and ethylene glycol (Wako Pure
Chemical Industries, Ltd.) were added to the resulting solution
at the molar ratio of citric acid/ethylene glycol/total metal
cations of 1.5/1.5/1. The pH value of the solution was controlled
to be ca. 8.0 with adding an aqueous ammonia solution (Wako
Results and discussion
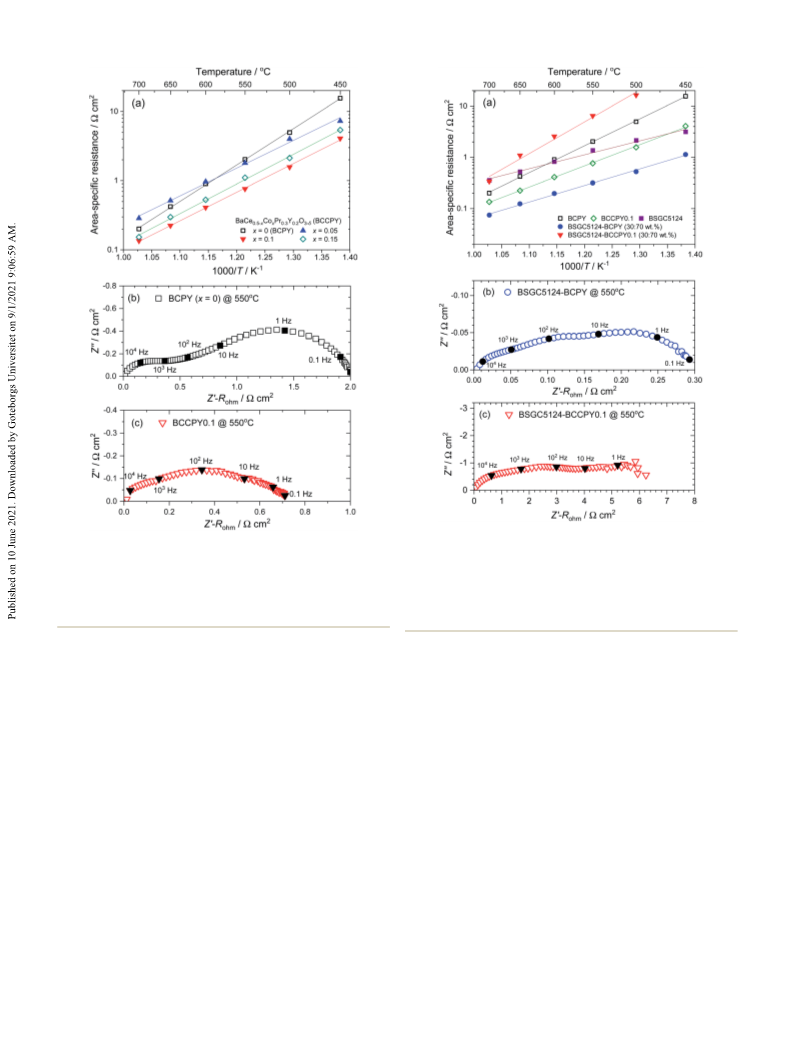
Pure Chemical Industries, Ltd.). Aer that the solution was First of all, the effect of elemental substitution, especially the
stirred at 80 ꢁC until the water was evaporated. The obtained gel Ba/Sr ratio and rare-earth elements, in (Ba,Sr)6RE2Co4O15 on the
ꢁ
was pre-heated at 350 C in air, and subsequently calcined at ORR activity was studied. Fig. 1 shows impedance spectra at
ꢁ
1100 ꢁC for 5 h in air. The crystal structures of resultant powders 550 C and the temperature dependence of area-specic resis-
were identied by X-ray diffraction (XRD, Rigaku Ultima IV X- tance (ASR) for (Ba6ꢀxSrx)Sm2Co4O15 (x ¼ 1–3) electrodes in
ray diffractometer, Cu Ka). The working conditions were 40 3 vol% humidied synthetic air. All oxides were red on the BCY
kV and 40 mA with a scanning rate of 20ꢁ minꢀ1. The diffraction disk with a symmetrical conguration at 1200 C. The polari-
ꢁ
patterns of (Ba6ꢀxSrx)RE2Co4O15 (x ¼ 1–3, RE ¼ La, Pr, Nd, Sm, zation resistance decreased with an increase in the Ba/Srꢁratio of
Gd) are summarized in Fig. S1 and S2 in the ESI.† The oxide (Ba6ꢀxSrx)Sm2Co4O15, which was prominent below 650 C. The
powder of Ba5SrGd2Co4O15 (BSGC5124) was mixed with BCPY to same tendency was conrmed for (Ba,Sr)6Gd2Co4O15, as shown
form the composite, and subsequently annealed at 1000 ꢁC for in Fig. S3.† Furthermore, the difference in the total electrical
5 h in air. Aer that, the local structure was analyzed using conductivity was conrmed; the conductivity for Ba5SrSm2Co4-
transmission electron microscopes (TEM), Tecnai G2 F20 (FEI O15 was one order of magnitude higher or more than that for
Co.) and ARM-200F (JEOL Ltd.) equipped with an energy Ba3Sr3Sm2Co4O15, as shown in Fig. S4.† These results indicate
dispersive X-ray spectrometer (EDX, JED-2300).
that the basicity, as well as the conductivity, strongly affects the
15200 | J. Mater. Chem. A, 2021, 9, 15199–15206
This journal is © The Royal Society of Chemistry 2021

 Eguchi, Koichi
Eguchi, Koichi
 Kamiuchi, Naoto
Kamiuchi, Naoto
 Kunimoto, Naoki
Kunimoto, Naoki
 Manriki, Kohei
Manriki, Kohei
 Matsui, Toshiaki
Matsui, Toshiaki
 Miyazaki, Kazunari
Miyazaki, Kazunari
 Muroyama, Hiroki
Muroyama, Hiroki