European Journal of Organic Chemistry
10.1002/ejoc.202000278
FULL PAPER
product was dissolved in 37% aq HCl 0.5 mL). TIS (2 drops) was added
and the reaction was stirred at r.t. for 4 h. The solution was freeze-dried
1
affording the title compound as a white powder (15 mg, 48% yield). H-
2
NMR (600 MHz, D O): δ= 8.68 (m, 1H, H6), 7.43 (s, 1H, H4), 4.17 (m, 1H,
H2), 4.06 (m, 1H, H15), 3.99 (m, 1H, H12), 3.50 (dd, J = 5.1, 15.7 Hz, 1H,
H1), 3.46-3.36 (m, 3H, H’1+H10+H’10), 2.75-2.63 (m, 2H, H18+H’18),
1
3
2
.41 (m, 2H, H11+H’11), 2.27 (m, 2H, H17+H’17). C-NMR (151 MHz,
O): δ= 176.5 (C19), 171.4 (C16), 171.0 (C14), 170.4 (C8), 134.1 (C6),
D
2
Figure 4. Pseudopaline numbering for attribution.
1
2
C
26.4 (C3), 118.0 (C4), 60.5 (C15), 59.9 (C2), 59.0 (C12), 43.8 (C10),
9.6 (C18), 26.4 (C11), 24.9 (C17), 24.6 (C1). MS(ESI): m/z calcd for
+
+
15
22 4 8
H N O : 386.1; found: 386.9(100) [M+H] , 369.1(60) [M-18+H] ,
+
(
R)-2-(((S)-1-carboxy-3-(((S)-1-carboxy-2-(1H-imidazol-4-
3
51.0(82) [M-36+H] . This compound corresponds to 4b.
yl)ethyl)amino)propyl)amino) pentanedioic acid trihydrochloride
1
(
4a*3HCl) H NMR (400 MHz, D
2
O): δ= 8.69 (d, J = 1.2 Hz, 1H, H6), 7.45
(
s, 1H, H4), 4.28 (dd, J = 5.2, 7.3 Hz, 1H, H2), 4.20-4.13 (m, 2H,
Acknowledgements
H15+H12), 3.54 (dd, J = 5.2, 15.9 Hz, 1H, H1), 3.49-3.39 (m, 3H,
H’1+H10+H’10), 2.68 (t, J = 7.0 Hz, 2H, H18+H’18), 2.40 (dt, J = 6.8, 7.4
1
3
Hz, 2H, H11+H’11), 2.27 (m, 2H, H17+H’17). C NMR (101 MHz, D
2
O):
We thank the association Vaincre la Mucoviscidose (VLM, grant
RFI20160501495) for financial support.
δ= 179.7 (C19), 176.4 (C16), 172.4 (C14), 169.9 (C8), 134.2 (C6), 126.2
(
C3), 118.2 (C4), 60.5 (C2), 59.2 (C15), 53.9 (C12), 44.0 (C10), 29.0
C18), 25.5 (C11), 24.4 (C1), 23.7 (C17). MS(ESI): m/z calcd for
(
+
+
Keywords: Natural products • Amino acids • Opines •
C
15 22 4 8
H N O : 386.1; found: 387.1(30) [M+H] , 369.1(95) [M-18+H] ,
+
20
3
51.0(98) [M-36+H] , 153.2(100). [α]
D
-9.1 (c 0.55, H
2
O). HRMS(ES+):
Pseudopaline • Total synthesis
+
m/z calcd for C15H N O : 387.1516; found: 387.1517 [M+H] (Δ = 0.3
23 4 8
ppm).
[
1]
a) A. Petit, C. David, G. A. Dahl, J. G. Ellis, P. Guyon, F. Casse-Delbart,
J. Tempé, Mol. Gen. Genet. 1983, 190 (2), 204-214; b) Y. Dessaux, A.
Petit, J. Tempé, Phytochemistry 1993, 34 (1), 31-38.
(
S)-2-(((S)-1-carboxy-3-(((S)-1-carboxy-2-(1H-imidazol-4-
yl)ethyl)amino)propyl)amino) pentanedioic acid trihydrochloride
[2]
N. Kan-no, M. Sato, T. Yokoyama, E. Nagahisa, Y. Sato, Comp.
Biochem. Physiol. B, Biochem. Mol. Biol. 1998, 121 (3), 323-332.
J. Thompson, J. Donkersloot, Res. Microbiol. 1992, 143 (2), 127-131.
I. Vladimirov, T. Matveeva, L. Lutova, Russ. J. Genet. 2015, 51 (2),
121-129.
1
(
4b*3HCl) H NMR (400 MHz, D
2
O): δ= 8.67 (d, J = 1.4 Hz, 1H, H6), 7.43
(
s, 1H, H4), 4.30 (dd, J = 5.2, 7.5 Hz, 1H, H2), 4.16 (t, J = 6.5 Hz, 1H,
[3]
[4]
H15), 4.11 (t, J = 6.5 Hz, 1H, H12), 3.52 (dd, J = 5.2, 15.9 Hz, 1H, H1),
.48-3.39 (m, 3H, H’1+H10+H’10), 2.69 (dd, J = 7.1, 13.2 Hz, 2H,
H18+H’18), 2.43 (m, 2H, H11+H’11), 2.29 (dd, J = 3.7, 7.0 Hz, 1H, H17),
3
[5]
[6]
M. Harcet, D. Perina, B. Pleše, Biochem. Genet. 2013, 51 (9-10), 666-
676.
1
3
2
.26 (dd, J = 4.1, 7.0 Hz, 1H, H’17). C NMR (101 MHz, D
2
O): δ= 176.4
(
C19), 170.9 (C16), 170.3 (C14), 169.8 (C8), 134.2 (C6), 126.1 (C3),
G. Ghssein, C. Brutesco, L. Ouerdane, C. Fojcik, A. Izaute, S. Wang, C.
Hajjar, R. Lobinski, D. Lemaire, P. Richaud, R. Voulhoux, A. Espaillat, F.
Cava, D. Pignol, E. Borezée-Durant, P. Arnoux, Science 2016, 352
(6289), 1105-1109.
1
2
3
3
18.2 (C4), 59.9 (C15), 59.4 (C2), 58.2 (C12), 43.6 (C10), 29.5 (C18),
6.3 (C11), 24.7 (C17), 24.4 (C1). MS(ESI): m/z calcd for C15
22 4 8
H N O :
+
+
86.1; found: 387.1(67) [M+H] , 369.1(83) [M-18+H] , 351.0(100) [M-
+
20
6+H] . [α]
D
+18.9 (c 0.55,
H
2
O). HRMS(ES+): m/z calcd for
[7]
a) J. S. McFarlane, A. L., Lamb, Biochemistry 2017, 56 (45), 5967-
5971; b) S. Lhospice, N. O. Gomez, L. Ouerdane, C. Brutesco, G.
Ghssein, C. Hajjar, A. Liratni, S. Wang, P. Richaud, S. Bleves, G. Ball,
E. Borezée-Durant, R. Lobinski, D. Pignol, P. Arnoux, R. Voulhoux, Sci.
Rep. 2017, 7 (1), 17132.
+
15 23 4 8
C H N O
: 387.1516; found: 387.1517 [M+H] (Δ = 0.3 ppm).
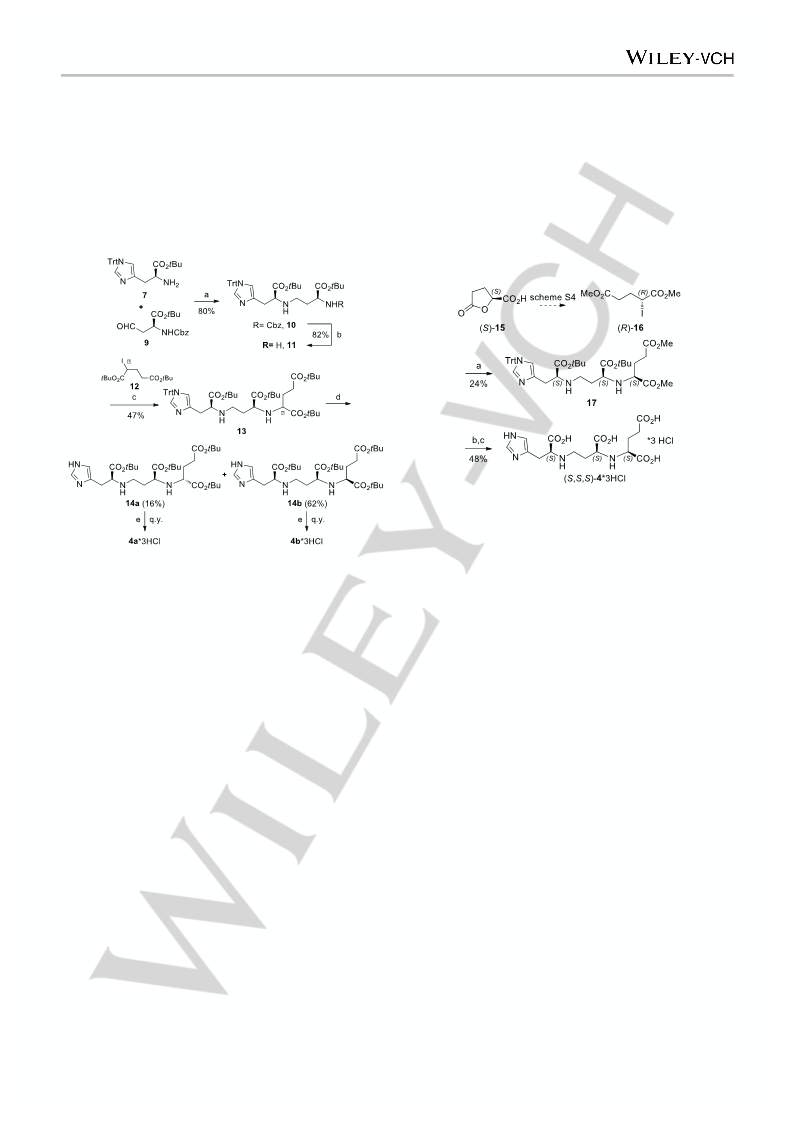
(
S)-Dimethyl 2-(((S)-1-(tert-butoxy)-4-(((S)-1-(tert-butoxy)-1-oxo-3-(1-
trityl-1H-imidazol-4-yl)propan-2-yl)amino)-1-oxobutan-2-
[
[
8]
9]
C. Curie, G. Cassin, D. Couch, F. Divol, K. Higuchi, M. Le Jean, J.
Misson, A. Schikora, P. Czernic, S. Mari, Ann. Bot. 2009, 103 (1), 1-11.
M. C. Mastropasqua, M. D'Orazio, M. Cerasi, F. Pacello, A. Gismondi,
A. Canini, L. Canuti, A. Consalvo, D. Ciavardelli, B. Chirullo, P.
Pasquali, A. Battistoni, Mol. Microbiol. 2017, 106 (4), 543-561.
yl)amino)pentanedioate (17) Amine 11 (255 mg, 0.417 mmol) was
3
dissolved in dry DMF (1 mL). NaHCO (42 mg, 0.500 mmol) and a
solution of iodure (R)-16 (143 mg, 0.500 mmol) in dry DMF (1 mL) were
added. The reaction was stirred at 45 °C until complete conversion.
Water was added and extracted twice with EtOAc. The pooled organic
[
10] a) M. Gi, K.-M. Lee, S. C. Kim, J.-H. Yoon, S. S. Yoon, J. Y. Choi, Sci.
Rep. 2015, 5, 14644; b) L. Remy, M. Carrière, A. Derré-Bobillot, C.
Martini, M. Sanguinetti, E. Borezée-Durant, Mol. Microbiol. 2013, 87 (4),
phase was dried over anhydrous Na
removed under reduced pressure. Purification column chromatography
on SiO gel with 98:2 to 95:5 DCM/MeOH + 2 drops of 28% aq. NH
afforded pure title compound as a yellow oil (76 mg, 24% yield). R = 0.32
). H NMR (400 MHz,
): δ= 7.35-7.28 (m, 10H), 7.14-7.08 (m, 6H), 6.60 (d, J = 1.1 Hz,
2 4
SO , filtered and the solvent was
2
3
7
30-743.
f
1
[
[
11] A. Folkesson, L. Jelsbak, L. Yang, H. K. Johansen, O. Ciofu, N. Høiby,
S. Molin, Nat. Rev. Microbiol. 2012, 10, 841.
(
96:4 DCM/MeOH + 2 drops of 28% aq. NH
3
CDCl
3
12] C. Hajjar, R. Fanelli, C. Laffont, C. Brutesco, G. Cullia, M. Tribout, D.
Nurizzo, E. Borezée-Durant, R. Voulhoux, D. Pignol, J. Lavergne, F.
Cavelier, P. Arnoux, J. Am. Chem. Soc. 2019, 141 (13), 5555-5562.
13] J. Zhang, S. Wang, Y. Bai, Q. Guo, J. Zhou, X. Lei, J. Org. Chem. 2017,
1
3
=
2
1
1
6
H), 3.67 (s, 3H), 3.62(s, 3H), 3.44 (m, 1H), 3.22 (dd, J = 5.5, 7.1 Hz, 1H),
.13 (dd, J = 5.0, 7.7 Hz, 1H), 2.90 (dd, J = 6.4, 14.5 Hz, 1H), 2.80 (dd, J
6.4, 14.5 Hz, 1H), 2.76-2.66 (m, 1H), 2.65-2.54 (m, 1H), 2.48-2.32 (m,
H) 2.10 (brs, 2H), 2.01-1.81 (m, 2H), 1.80-1.68 (m, 1H), 1.68-1.57 (m,
[
[
[
1
3
8
2 (24), 13643-13648.
3
H), 1.42 (s, 9H), 1.36 (s, 9H). C NMR (101 MHz, CDCl ): δ= 174.5,
14] M. b. Bouazaoui, M. Larrouy, J. Martinez, F. Cavelier, Eur. J. Org.
Chem. 2010, 2010 (34), 6609-6617.
73.8, 173.7, 142.5, 138.4, 137.2, 129.8, 128.0, 119.3, 81.3, 81.0, 75.2,
1.8, 59.2, 59.1, 51.9, 51.6, 44.6, 33.8, 32.0, 30.1, 28.1, 28.1. MS(ESI):
+
15] a) M.-R. Abdo, P. Joseph, R.-A. Boigegrain, J.-P. Liautard, J.-L.
Montero, S. Köhler, J.-Y. Winum, Biorg. Med. Chem. 2007, 15 (13),
m/z calcd for C44
H
56
N
4
O
8
: 768.4; found: 769.5(30) [M+H] , 527.4(100)
+
[
M-Trt+H] .
4
6
427-4433; b) J. Xu, J. C., Yadan, J. Org. Chem. 1995, 60 (20), 6296-
301.
(
S)-2-(((S)-1-Carboxy-3-(((S)-1-carboxy-2-(1H-imidazol-4-
[
16] J. Zhang, T. Zhao, R. Yang, I. Siridechakorn, S. Wang, Q. Guo, Y. Bai,
yl)ethyl)amino)propyl)amino)pentanedioic acid trihydrochloride
H. C. Shen, X. Lei, Chem. Sci. 2019, 10, 6635-6641.
[
(S,S,S)-4*3HCl] Compound 17 (49 mg, 63.7 µmol) was dissolved in THF
(
0.14 mL) and 1 M aq. LiOH (0.14 mL) was added. After stirring at room
temperature for 2 h, the crude was purified by HPLC. The obtained
5
This article is protected by copyright. All rights reserved.

 Arnoux, Pascal
Arnoux, Pascal
 Cavelier, Florine
Cavelier, Florine
 Cullia, Gregorio
Cullia, Gregorio
 Fanelli, Roberto
Fanelli, Roberto
 Voulhoux, Romé
Voulhoux, Romé