Page 5 of 6
PleaseRd So Cn oA t da vd aj un s ct ems argins
DOI: 10.1039/C6RA14060B
Journal Name
of compound 40 as compared with compounds 1b and 39
COMMUNICATION
.
compound 40 is being carried out in our laboratory. Results
Apparently, compounds 39 and 40 are useful hits for the will be reported in due course.
molecular design of inhibitors that specifically bind to the ATP
pocket of PDKs. Further work on hit expansion based on
6B
6
A
6C
6D
Fig. 6 Molecular modelling of compounds in the ATP binding pocket of PDK2. 6A: compound 1b and ATP in the ATP binding pocket of PDK2; 6B: compound
8 and ATP in the ATP binding pocket of PDK2; 6C: compound 39 in the ATP binding pocket of PDK2; 6D: compound 40 in the ATP binding pocket of PDK2
PDB code: 2BU2). All the amino acid residues and compounds are shown as stick models, H-bonds are shown as yellow dashed lines. The 3D graphical
3
(
presentations were drawn by PyMol.
6
7
S.-L. Zhang, X. H. Hu, W. Zhang, H. K. Yao and K. Y. Tam, Drug
Discov. Today, 2015, 20, 1112.
S. C. Tso, X. Qi, W. J. Gui, C. Y. Wu, J. L. Chuang, I. Wernstedt-
Asterholm, L. K. Morlock, K. R. Owens, P. E. Scherer, N. S.
Williams, U. K. Tambar, R. M. Wynn and D. T. Chuang, J. Biol.
Chem., 2014, 289, 4432.
Conclusions
In this paper, we descried the synthesis and identification of
1
novel PDK1 inhibitors. Their structures were confirmed by H
1
3
8
9
1
1
1
1
W. Zhang, S.-L. Zhang, X. H. Hu and K. Y. Tam, Int. J. Biol. Sci.,
2
NMR, C NMR, and HRMS. Biological assays revealed that
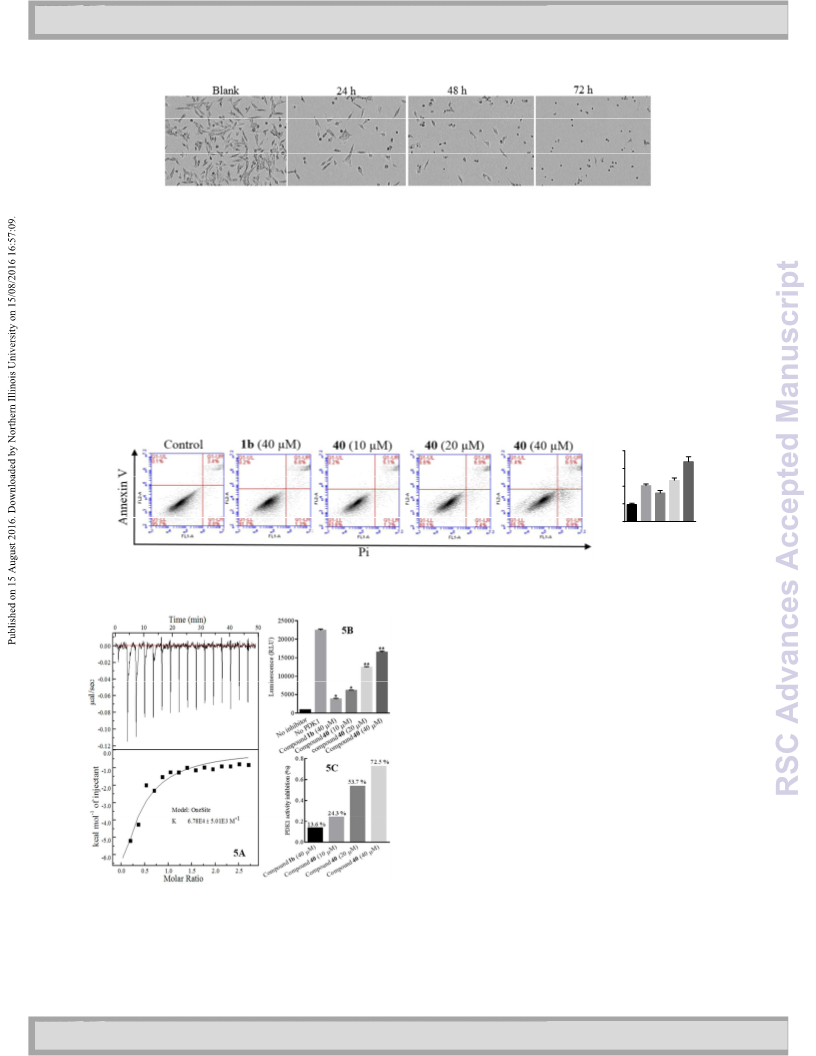
compound 40 inhibited the cancer cell proliferation with an
IC50 value of 8.21 µM against the SF188 cancer cells. ITC
experiment suggested that compound 40 bound to PDK1 with
015, 11, 1390.
S. M. Wigfield, S. C. Winter, A. Giatromanolaki, J. Taylor, M.
L. Koukourakis and A. L. Harris, Br. J. Cancer, 2008, 98, 1975.
0 M. I. Koukourakis, A. Giatromanolaki, G. Bougioukas and E.
Sivridis, Cancer Biol. Ther., 2007, 6, 1476.
1 S. Fujiwara, Y. Kawano, H. Yuki, Y. Okuno, K. Nosaka, H.
Mitsuya and H. Hata, Br. J. Cancer, 2013, 108, 170.
2 H. Hur, Y. Xuan, Y. B. Kim, G. Lee, W. Shim, J. Yun, I. H. Ham
and S. U. Han, Int. J. Oncol., 2013, 42, 44.
3 B. Daniel, R. Uta, H. Jannis, K. Hans, E. Jan, C. Hannes, M.
Kurt, S. Martin and W. Steffen, World J. Urol., 2013, 31,
a K
compound 40 in 10, 20, and 40 µM reduced PDK1 activity by
4.3, 53.7, and 72.5 %, respectively. Molecule modeling study
d
value of 14.7 µM. Kinase activity assay showed that
2
indicated that compound 40 bound to ATP pocket of PDKs,
potentially forming four hydrogen bonds with amino acid
residues Glu 243, Asp 282, Thr 320, and Thr 346. Compound 40
could be a probe molecule to explore the pharmacology of
PDK1.
1
191.
1
1
4 J. A. Morrell, J. Orme, R. J. Butlin, T. E. Roche, R. M. Mayers
and E. Kilgour, Biochem. Soc. Trans., 2003, 31, 1168.
5 T. D. Aicher, R. C. Anderson, G. R. Bebernitz, G. M. Coppola,
C. F. Jewell, D. C. Knorr, C. Liu, D. M. Sperbeck, L. J. Brand, R.
J. Strohschein, J. Gao, C. C. Vinluan, S. S. Shetty, C. Dragland,
E. L. Kaplan, D. DelGrande, A. Islam, X. Liu, R. J. Lozito, W. M.
Maniara, R. E. Walter and W. R. Mann, J. Med. Chem., 1999,
Acknowledgments
We thank the financial support from the Science and
Technology Development Fund, Macao S.A.R. (FDCT) (project
reference no. 086/2014/A2). This work was supported partly
by the startup of South University of Science and Technology
of China and by Science, Technology and Innovation
Commission of Shenzhen Municipality (Grant No.
JCYJ20140417105742705) to Z.W.
4
6 P. Gahlot and R. Kakkar, Int. Res. J. Pharm., 2011, 1, 33.
2, 2741.
1
1
7 J. D. Moore, A. Staniszewska, T. Shaw, J. D'Alessandro, B.
Davis, A. Surgenor, L. Baker, N. Matassova, J. Murray, A.
Macias, P. Brough, M. Wood, P. C. Mahon, Oncotarget, 2014,
5
, 12862.
8 M. Kato, J. Li, J. L. Chuang and D. T. Chuang, Structure, 2007,
, 992.
1
1
1
5
Notes and references
9 P. W. Stacpoole, T. L. Kurtz, Z. Han and T. Langaee, Adv.
Drug. Deliv. Rev., 2008, 60, 1478.
1
2
3
4
D. A. Tennant, R. V. Durán and E. Gottlieb, Nat. Rev. Cancer,
010, 10, 267.
S.-L. Zhang, X. H. Hu, W. Zhang and K. Y. Tam, J. Med. Chem.,
016, 59, 3562.
R. J. DeBerardinis, J. J. Lum, G. Hatzivassiliou and C. B.
Thompson, Cell Metab., 2008, , 11.
S. Bonnet, S. L. Archer, J. Allalunis-Turner, A. Haromy, C.
Beaulieu, R. Thompson, C. T. Lee, G. D. Lopaschuk, L.
Puttagunta, S. Bonnet, G. Harry, K. Hashimoto, C. J. Porter,
M. A. Andrade, B. Thebaud and E. D. Michelakis, Cancer Cell,
2
20 M. H. Norman, N. Chen, Z. D. Chen, C. Fotsch, C. Hale, N.
Han, R. Hurt, T. Jenkins, J. Kincaid, L. B. Liu, Y. L. Lu, O.
Moreno, V. J. Santora, J. D. Sonnenberg and W. Karbon, J.
Med. Chem., 2000, 43, 4288.
21 N. Baindur, N. Chadha and M. R. Player, J. Comb. Chem.,
2003, 5, 653.
2
7
2
007, 11, 37.
5
E. D. Michelakis, G. Sutendra, P. Dromparis, L. Webster, A.
Haromy, E. Niven, C. Maguire, T.-L. Gammer, J. R. Mackey, D.
Fulton, B. Abdulkarim, M. S. McMurtry and K. C. Petruk, Sci.
Transl. Med., 2010, 2, 31ra34.
This journal is © The Royal Society of Chemistry 20xx
J. Name., 2013, 00, 1-3 | 5
Please do not adjust margins

 Zhang, Shao-Lin
Zhang, Shao-Lin
 Zhang, Wen
Zhang, Wen
 Xiao, Qingpin
Xiao, Qingpin
 Yang, Zheng
Yang, Zheng
 Hu, Xiaohui
Hu, Xiaohui
 Wei, Zhiyi
Wei, Zhiyi
 Tam, Kin Yip
Tam, Kin Yip