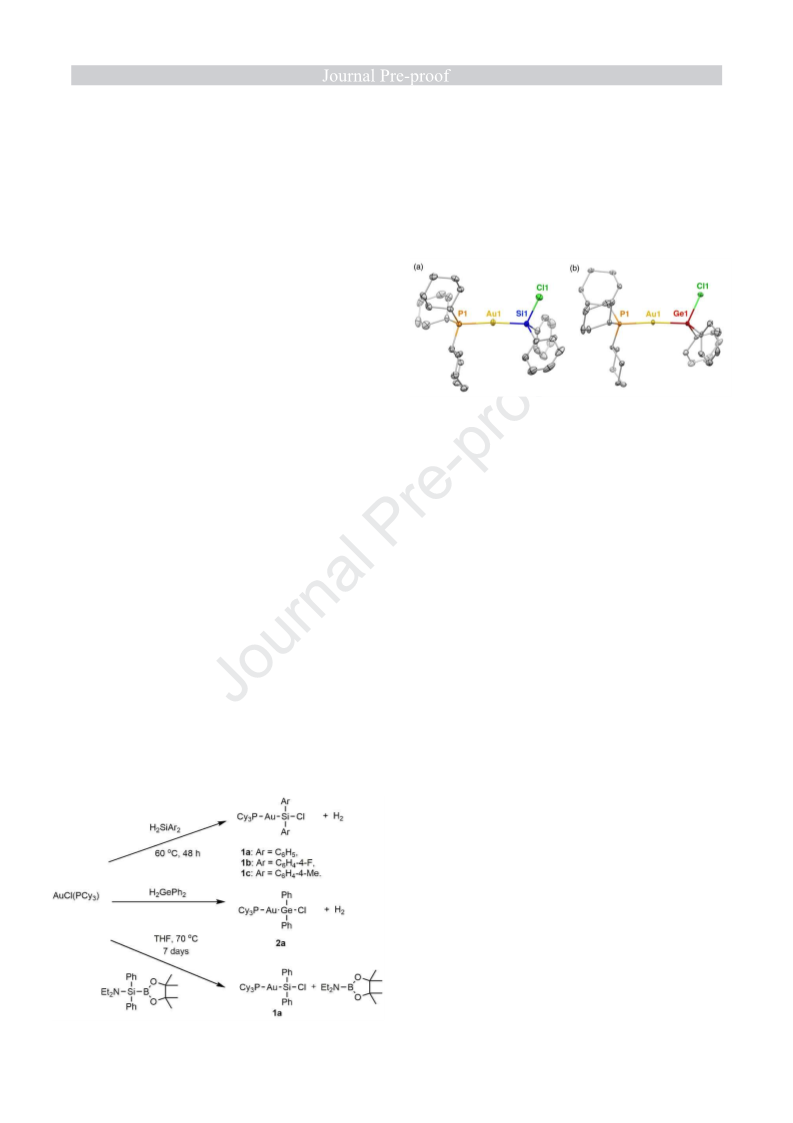
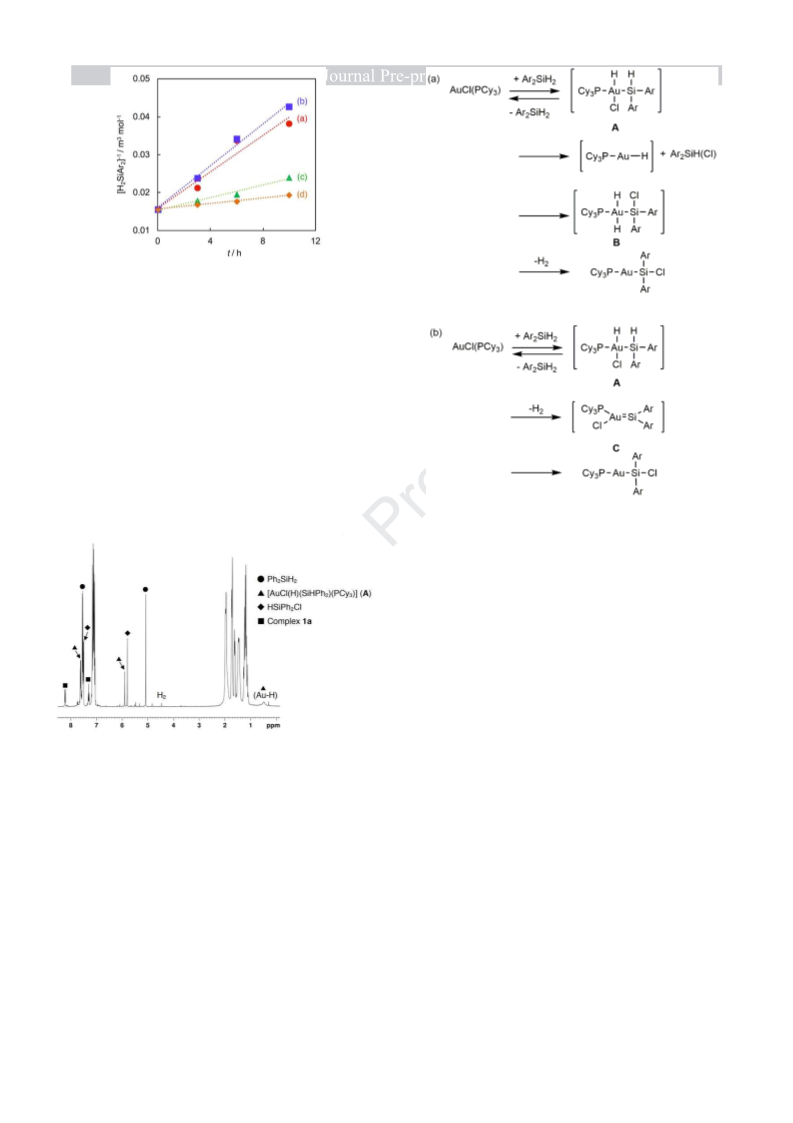
6
Tetrahedron
vacuo to give [Au(SiArF Cl)PCy3] (1b) as a white solid (29.9
standard: 4.2 mg) was added to the reaction mixture. D2SiPh2
2
mg, 7%). 1H NMR (400 MHz, C6D6, rt): δ 7.98 (q, JHH = 6.36 Hz,
JFH = 8.28 Hz, 4H), 6.97 (t, JHH = 8.88 Hz, 4H), 1.83 (br, 9H),
1.61 (br, 6H), 1.53 (br, 3H), 1.36 (br, 6H), 1.05 (br, 9H). 19F{1H}
NMR (C6D6, 376 MHz, rt): δ -113.5 (s, (4-F-C6H4)). 31P{1H}
NMR (161 MHz, C6D6, rt): δ 69.7, (s, PCy3). 13C{1H} NMR (126
(9.0 mg, 0.032 mmol) was injected to the reaction mixture via 10
o
µL micro syringe. The reaction mixture was stirred at 60 C for
24 h, which was monitored by 1H NMR, to afford
[Au(SiPh2Cl)(PCy3)] as the product. By comparing with the
signals corresponding to reactant (δΗ 7.50) and product (δΗ 8.22),
the conversion of the reactant and the NMR yield of the product
were determined (15% yield, k = 1.11×10-7 m3 mol-1 s-1, kH/kD =
6.36).
MHz, C6D6, rt): δ 141.5 (d, JCF = 3.7 Hz, ipso-C), 137.6 (d, JCF
7.6 ortho-C), 115.2 (d, JCF 18.9 para-C), 33.8 (br,
=
=
PCH(CH2CH2)CH2), 30.9 (s, PCH(CH2CH2)CH2), 27.4 (s,
PCH(CH2CH2)CH2), 26.3 (s, PCH(CH2CH2)CH2). The SiArF
2
(meta) signal was not assigned clearly due to overlapping with
solvent signal.
4-5. Synthesis of O-functionalized organosilane
4.5.1 Reaction of 1a with H2O.
4.2.6 Preparation of 1c.
In a J. Young NMR tube under inert gas, to a THF-d8
(0.5 mL) solution of [Au(SiPh2Cl)(PCy3)] (1a) (10 mg, 0.014
mmol) and dibenzyl (3.7 mg) was added H2O (1.8 ꢀL, 0.10
mmol). The reaction mixture was stirred at room temperature for
15 h. The conversion of the reactant and the NMR yield of the
product were determined by comparing integration of the
methylene signal of the internal standard (dibenzyl, δH 2.88) with
the signals corresponding to reactant (1a: δH 7.67) and product
In
a glovebox, to THF (10 mL) solution of
[AuCl(PCy3)] (200 mg, 0.39 mmol) was added (4-MeC6H4)2SiH2
(83 mg, 0.39 mmol). The reaction mixture was stirred at 60 C
o
for 48 h. After the solvent was removed under reduced pressure,
the resulting material was washed with hexane (2 mL×2) and
dried in vacuo to give [Au(Si(C6H4Me-4)2Cl)(PCy3)] (1c) as a
gray solid (108 mg). 1H NMR (400 MHz, C6D6, rt): δ 8.17 (d, JHH
= 7.72 Hz, 4H), 2.12 (s, 3H), 1.97 (br, 9H), 1.67 (br, 6H), 1.52
(br, 3H), 1.26 (br, 6H), 1.14 (br, 9H). The Si(tol)2 (meta) signal
was not assigned clearly due to overlapping with solvent signal.
31P{1H} NMR (161 MHz, C6D6, rt): δ 69.8 (s, PCy3).
1
(HSiPh2OH: δH 7.60), The H and 31P{1H} NMR spectra of the
reaction mixture exhibited quantitative conversion to give
HSiPh2OH (83% NMR yield).
4.5.2 Reaction of 1a with excess H2O.
4.2.7 Preparation of 2a.
In a 25 mL Schlenk tube under inert gas, to a THF (2
mL) solution of [Au(SiPh2Cl)(PCy3)] (1a) (20 mg, 0.029 mmol)
was added H2O (50 ꢀL, 2.8 mmol). After the reaction mixture
was stirred at room temperature for 39 h, the solvent was
removed under reduced pressure. The resulting material was
washed by hexane (2 mL × 2) and dried in vacuo to give
[AuCl(PCy3)] as a white solid. 31P{1H} NMR (161 MHz, C6D6,
rt): δ 53.1 (s, PCy3).
In a glovebox, to a toluene solution (10 mL) of
[AuCl(tht)] (200 mg, 0.62 mmol) was added PCy3 (438 mg, 1.6
mmol), forming [AuCl(PCy3)] in situ. Ph2GeH2 (357 mg, 1.56
mmol) was injected to the reaction mixture via micro syringe.
The reaction mixture was stirred at 110 oC for 5 days. The
solvent was removed under reduced pressure. The resulting
material was washed with hexane (6 mL×2), acetone (1 mL×2)
and dried in vacuo to give [Au(GePh2Cl)(PCy3)] (2a) as a white
solid (209 mg, 25%). Recrystallization by slow diffusion of
CH2Cl2/hexane (1:10) in a glovebox afforded a colorless crystal
of [Au(GePh2Cl)(PCy31)] suitable for X-Ray crystallography
(CCDC No. 1957766). H NMR (400 MHz, C6D6, rt): δ 8.21 (d,
JHH = 6.96 Hz, 4H), 7.30 (t, JHH = 7.44 Hz, 4H), 1.80 (br, 9H),
1.60 (br, 6H), 1.31 (br, 6H), 1.05 (br, 9H). 31P{1H} NMR (161
MHz, C6D6, rt): δ 64.7 (s, PCy3). 13C{1H} NMR (126 MHz,
C6D6, rt):δ150.6 (s, ipso-C), 134.8 (s, ortho-C), 128.6 (s, meta-
4.5.3 Reaction of 1a with EtOH.
In a J. Young NMR tube under inert gas, to a C6D6 (0.5
mL) solution of [Au(SiPh2Cl)(PCy3)] (1a) (8 mg, 0.012 mmol)
and dibenzyl (3.7 mg) was added EtOH (0.7 ꢀL, 0.015 mmol).
The reaction mixture was stirred at room temperature for 16 h.
The conversion of the reactant and the NMR yield of the product
were determined by comparing integration of the methylene
signal of the internal standard (dibenzyl, δH 2.74) with the signals
corresponding to reactant (1a: δH 8.21) and product (HSiPh2OEt:
C), 128.4 (s, para-C), 33.5 (d, JPC
PCH(CH2CH2)CH2), 30.7 (s, PCH(CH2CH2)CH2), 27.1 (s,
PCH(CH2CH2)CH2), 26.0 (s, PCH(CH2CH2)CH2).
=
20.7 Hz,
1
δH 5.66). The H and 31P{1H} NMR spectra of the reaction
mixture exhibited quantitative conversion to give HSiPh2OEt
(77% NMR yield).
4-3. Kinetics study for formation of 1a, 1b, 1c.
In a J. Young NMR tube, to a C6D6 solution (0.5 mL) of
4.5.4 Reaction of H2SiPh2 with H2O catalyzed by 1a in NMR
scale.
[AuCl(tht)] (10 mg, 0.032 mmol) was added PCy3 (9.0 mg, 0.032
mmol) forming [AuCl(PCy3)] in situ. Dibenzyl (internal
standard: 4.2 mg) was added to the reaction mixture. H2SiAr2
(0.032 mmol) was injected to the reaction mixture via 10 µL
micro syringe. The reaction mixture was stirred at 60 oC for 24 h,
which was monitored by 1H NMR, to afford [Au(SiAr2Cl)(PCy3)]
as the product. By comparing integration of the methylene signal
of the internal standard (δΗ 2.74) with the Si-H signals of reactant
(δΗ 5.07 (1a); δΗ 4.88 (1b); δΗ 5.16 (1c)) and product (δΗ 8.22
(1a); δΗ 7.98 (1b); δΗ 8.17 (1c)), the conversion of the reactant
and the NMR yield of the product were determined (1a, 26%
yield, k = 6.56×10-7 m3 mol-1 s-1; 1b, 38% yield, k = 7.65×10-7
m3 mol-1 s-1; 1c, 6% yield, k = 2.30×10-7 m3 mol-1 s-1).
In a J. Young NMR tube under inert gas, to a THF-d8
(0.5 mL) solution of [Au(SiPh2Cl)(PCy3)] (1a) (5.3 mg, 0.0076
mmol), H2SiPh2 (28 mg, 0.15 mmol), and dibenzyl (4.0 mg) was
added H2O (1.1 ꢀL, 0.061 mmol). The reaction mixture was
o
stirred at 60 C for 118 h. The conversion of the reactant and the
NMR yield of the product were determined by comparing
integration of the methylene signal of the internal standard
(dibenzyl, δH 2.88) with the signals corresponding to reactant
1
(H2SiPh2: δH 4.89) and product ((HSiPh2)2O: δH 5.60), The H
NMR spectra of the reaction mixture exhibited 85% conversion
to give and (HSiPh2)2O (56% NMR yield).
4.5.5 Reaction of H2SiPh2 with H2O catalyzed by 1a in large
scale.
4-4. Deuterium labeling study.
In a J. Young NMR tube, to a C6D6 solution (0.5 mL) of
[AuCl(tht)] (10 mg, 0.032 mmol) was added PCy3 (9.0 mg, 0.032
mmol) forming [AuCl(PCy3)] in situ. Dibenzyl (internal
In a 25 mL Schlenk tube under inert gas, to a THF (2
mL) solution of [Au(SiPh2Cl)(PCy3)] (1a) (18 mg, 0.025 mmol),
H2SiPh2 (93 mg, 0.51 mmol) was added H2O (8.3 ꢀL, 0.46

 Kanda, Atsushi
Kanda, Atsushi
 Osakada, Kohtaro
Osakada, Kohtaro
 Tsuchido, Yoshitaka
Tsuchido, Yoshitaka