3530 J. Agric. Food Chem., Vol. 58, No. 6, 2010
Zhang et al.
(
(
(
(
6) Adams, J. B. Thermal degradation of anthocyanins with particular
reference to the 3-glycosides of cyanidin I. In acidified aqueous
solution at 100°C. J. Sci. Food Agric. 1973, 24, 747–762.
7) Sadilova, E.; Carle, R.; Stintzing, F. C. Thermal degradation of
anthocyanins and its impact on color and in vitro antioxidant
capacity. Mol. Nutr. Food Res. 2007, 51, 1461–1471.
8) Hrazdina, G.; Franzese, A. J. Oxidation products of acylated
anthocyanins under acidic and neutral conditions. Phytochemistry
1
974, 13, 231–234.
9) Tsuda, T.; Ohshima, K.; Kawakishi, S.; Osawa, T. Oxidation
products of cyanidin 3-O-β- -glucoside with a free radical initiator.
Lipids 1996, 31 (2), 1259–1263.
10) Tsuda, T.; Horio, F.; Osawa, T. Absorption and metabolism
of cyanidin-3-O-β- -glucoside in rats. FEBS Lett. 1999, 449,
79–182.
D
(
(
D
1
11) Kader, F.; Haluk, J. P.; Nicolas, J. P.; Metche, M. Degradation
of cyanidin-3-glucoside by blueberry polyphenol oxidase:
kinetics study and mechanisms. J. Agric. Food Chem. 1998, 46,
3
060–3065.
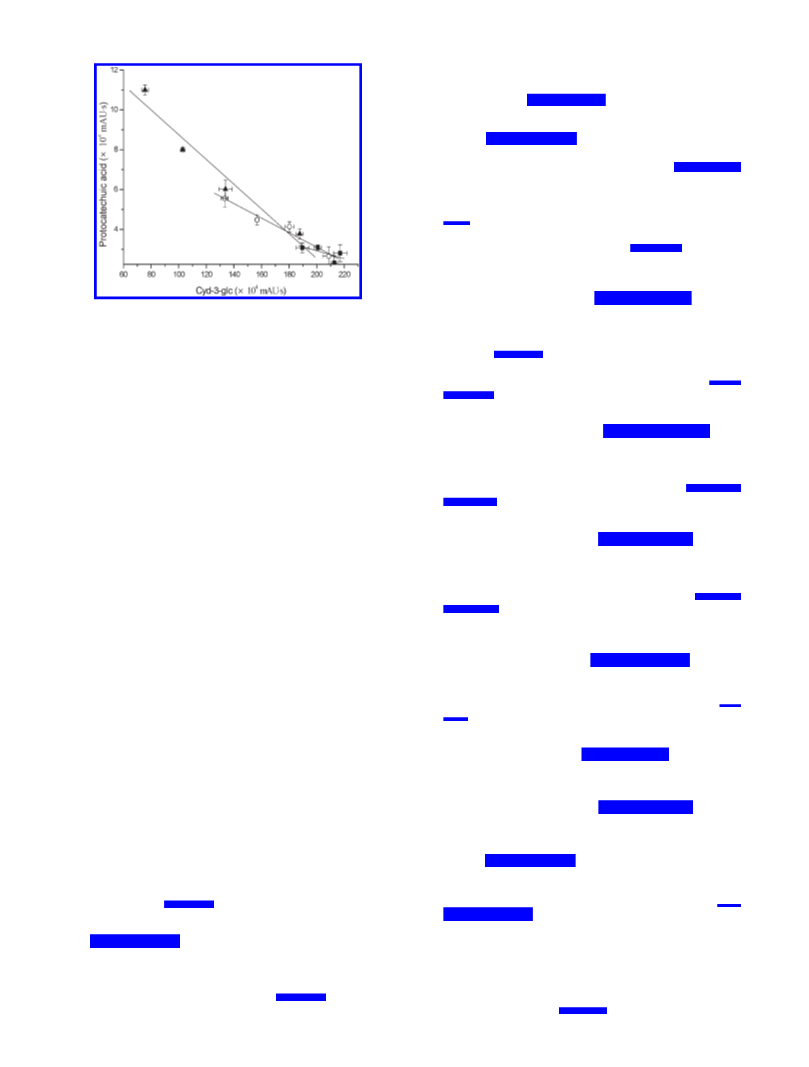
Figure 7. Correlation between the degradation of Cyd-3-glc and the
formation of protocatechuic acid exposed to PEF at (9) 7, (O)
(
(
(
12) Jiang, Y. M.; Duan, X. U.; Joyce, D.; Zhang, Z. Q.; Li, J. R.
Advances in understanding of enzymatic browning in harvested
litchi fruit. Food Chem. 2004, 88, 443–446.
14 kV/cm, and (2) 22 kV/cm of the electric field strength.
13) Keppler, K.; Humpf, H. Metabolism of anthocyanins and their
phenolic degradation products by the intestinal microflora. Bioorg.
Med. Chem. 2005, 13, 5195–5205.
14) Zhong, K.; Chen, F.; Wang, Z. F.; Wu, J. H.; Liao, X. J.; Hu, X. S.
Inactivation and kinetic model for the Escherichia coli treated
by a co-axial pulsed electric field. Eur. Food Res. Technol. 2005,
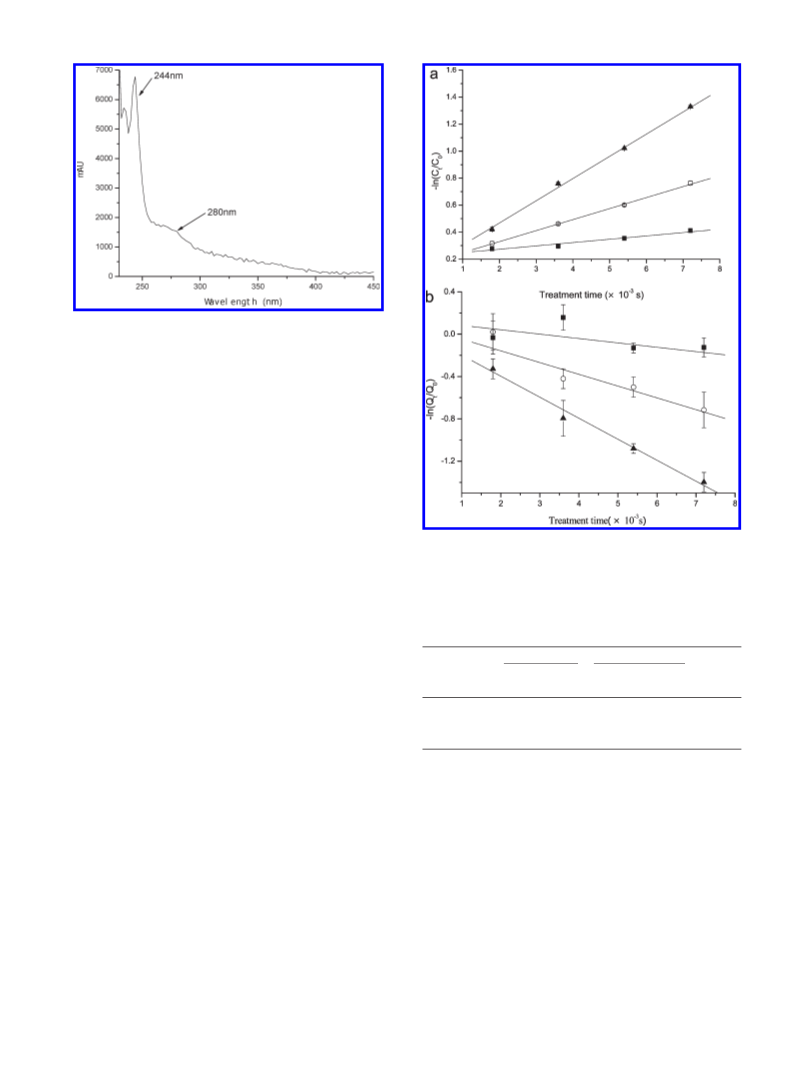
Possible Mechanism of Cyd-3-glc Exposed to PEF. The absence
of cyanidin in PEF treated samples indicated that the first step
of Cyd-3-glc degradation induced by PEF was not the hydrolysis
of a glycosidic bond. According to previous reports, PEF could
induce electrochemical reactions and lead to changes of tempera-
ture, pH, and chemical composition (25-27): for example,
2
21, 472–478.
(
(
(
(
15) Vega-Mercado, H.; Martın-Belloso, O.; Qin, B. L.; Chang, F. J.;
G oꢁ ngora-Nieto, M. M.; Barbosa-C ꢁa novas, G. V.; Swanson, B. G.
Non-thermal food preservation: pulsed electric fields. Trends Food
Sci. Technol. 1997, 8, 151–157.
´
2þ
3þ
the release of Fe /Fe from stainless steel electrodes (25, 28),
the increase of oxygen dissolution (27), and even the presence of
hydrogen peroxide caused by a lower pH value of sample
solution (29). These may be the possible reasons resulting in the
absence of cyanidin in PEF treated sample. In the investigation
16) Hye, W. Y.; Streaker, C. B.; Zhang, Q. H.; Min, D. B. Effects of
pulsed electric fields on the quality of orange juice and com-
parison with heat pasteurization. J. Agric. Food Chem. 2000, 48,
of Tsuda et al. (9), the oxidation products of cyanidin 3-O-β-
glucoside with 2,2 -azobis(2,4-dimethylvaleronitrile) were
D
-
4
597–4605.
0
17) Zhang, Y.; Liao, X. J.; Ni, Y. Y.; Wu, J. H.; Hu, X. S.; Wang, Z. F.;
Chen, F. Kinetic analysis of the degradation and its color of
cyanidin-3-glucoside exposed to pulsed electric field. Eur. Food
Res. Technol. 2007, 224, 597–603.
identified as 4,6-dihydroxy-2-O-β-D-glucosyl-3-oxo-2,3-dihydro-
benzofuran and protocatechuic acid. According to Hrazdina and
Franzes ꢀe ’s (8) investigation, upon nucleophilic attack of the H O2
2
at the 2-carbon of the molecule, the heterocyclic ring was cleaved
18) Odriozola-Serrano, I.; Soliva-Fortuny, R.; Gimeno-A oꢁ , V.;
between C and C to form o-benzoyloxyphenylacetic acid esters
Martın-Belloso, O. Kinetic study of anthocyanins, vitamin C, and
´
2
3
of the malvone type. These esters were further hydrolyzed under
alkaline conditions to the B-ring acid, the sugar substituent of the
antioxidant capacity in strawberry juices treated by high-
intensity pulsed electric fields. J. Agric. Food Chem. 2008, 56,
8387–8393.
3-position, and the elusive 2,4,6-trihydroxyphenylacetic acid (8).
(
19) Fossen, T.; Cabrita, L.; Andersen, O. M. Colour and stability of pure
The results mentioned above indicated that cyanidin could be
absent in the oxidized reaction of anthocyanin. Oxidation could
thus play an important role in the degradation of Cyd-3-glc
exposed to PEF. Moreover, great changes in pH in the vicinity of
electrodes (the acidification around the anode and alkalization at
the cathode) will break down the chemical equilibrium of the
Cyd-3-glc solution and directly lead to the formation of quino-
noidal anhydrobase or carbinol pseudobase, which was unstable
and further degraded to chalcones and coumarin glycosides.
e
anthocyanins influenced by pH including the alkaline region. Food
Chem. 1998, 63, 435–440.
(
20) Gao, L.; Girard, B.; Mazza, G.; Reynolds, A. G. Changes in
anthocyanins and color characteristics of Pinot Noir wines during
different vinification processes. J. Agric. Food Chem. 1997, 45, 2003–
2008.
(
(
21) Giusti, M. M.; Rodrıguez-Saona, L. E.; Wrolstad, R. E. Molar
´
absorptivity and color characteristics of acylated and non-acylated
pelargonidin-based anthocyanins. J. Agric. Food Chem. 1999, 47,
4631–4637.
22) Stintzing, F. C.; Stintzing, A. S.; Carle, R.; Frei, B.; Wrolstad, R. E.
Color and antioxidant properties of cyanidin-based anthocyanin
pigment. J. Agric. Food Chem. 2002, 50, 6172–6181.
LITERATURE CITED
(
(
(
1) Mazza, G.; Miniati, E. Anthocyanins in Fruits, Vegetables, And
Grains; CRC Press: Boca Raton, FL, 1993.
(23) Odriozola-Serrano, I.; Soliva-Fortuny, R.; Martın-Belloso, O.
´
2) Bridle, P.; Timberlake, C. F. Anthocyanins as natural food colours-
selected aspects. Food Chem. 1997, 58, 103–109.
3) Seeram, N. P.; Bourquin, L. D.; Nair, M. G. Degradation products
of cyanidin glycosides from tart cherries and their bioactivities.
J. Agric. Food Chem. 2001, 49, 4924–4929.
Impact of high-intensity pulsed electric fields variables on vitamin
C, anthocyanins and antioxidant capacity of strawberry juice. LWT
- Food Sci. Technol. 2009, 42 (1), 93–100.
(24) Zhao, W.; Yang, R. J; Wang, M.; Lu, R. R. Effects of pulsed
electric fields on bioactive components, colour and flavour of
green tea infusions. Int. J. Food Sci. Technol. 2009, 312 (44), 312–
321.
(25) Loomis-Husselbee, J. W.; Cullen, P. J.; Irvine, R. F.; Dawson, A. P.
Electroporation can cause artifacts due to solubilization of cations
from the electrode plates. Biochem. J. 1991, 277, 883–885.
(
4) Markakis, P. Anthocyanins as Food Colors; Academic Press:
New York, 1982; pp 163-180.
(
5) Sadilova, E.; Stintzing, F. C.; Carle, R. Thermal degradation of
acylated and non-acylated anthocyanins. J. Food Sci. 2006, 71 (8),
5
04–512.

 Zhang, Yan
Zhang, Yan
 Sun, Jianxia
Sun, Jianxia
 Hu, Xiaosong
Hu, Xiaosong
 Liao, Xiaojun
Liao, Xiaojun