Article
Inorganic Chemistry, Vol. 49, No. 24, 2010 11371
AlO(OH), to screen the cation-cation repulsions. Besides, the
metal oxide-hydroxide layer is puckered in AlO(OH). There is
therefore immense scientific interest in the synthesis of Al-
(OH)3-based LDHs containing different metal ions and in the
investigation of their structures. Further Al(OH)3 itself crystal-
lizes in a number of polymorphic modifications of which the
most common are gibbsite7 and bayerite.15 The main difference
between the two polymorphs is in the stacking sequence of the
hydroxyl ions. In gibbsite the hydroxyl ions are stacked in an
eclipsed manner (AB BA AB3 3 3 ) whereas in bayerite the
stacking sequence is approximately AB AB AB3 3 3
From the point of view of applications, the Li-Al LDHs
exhibit shape selective catalysis.16-18 This selectivity has a struc-
tural basis and is thought to arise because of cation ordering.
In this paper, we report the synthesis and structure refine-
ment of an Al(OH)3 based II-III LDH containing Zn and Co.
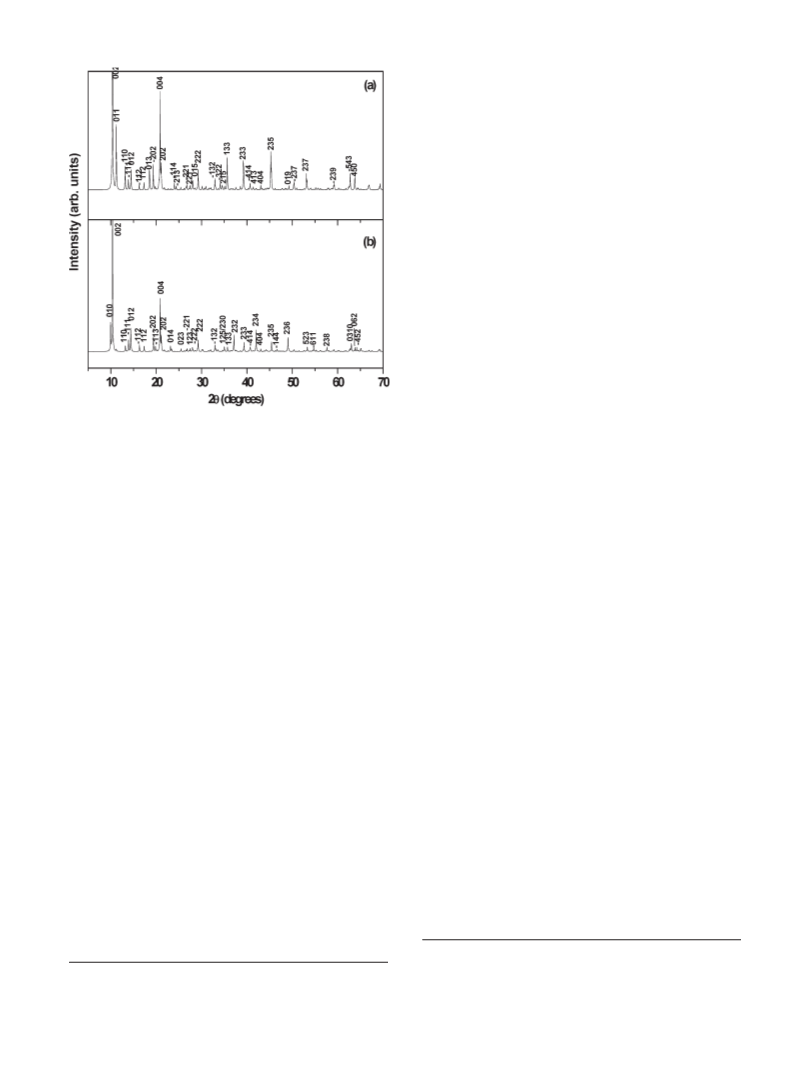
Hydrothermal treatment of bayerite in a ZnSO4 solution
results in a highly crystalline product whereas reaction of ZnO
with Al2(SO4)3 solution results in a compound with structural
disorder. While the incidence of polytypism and stacking
disorders is well understood in the hexagonal brucite-based
LDHs, similar phenomena in Al(OH)3-based LDHs are not
described.19 We investigate the potential for polytypism in the
Al(OH)3-based II-III LDHs and show that the anion ex-
change properties of these LDHs are distinctly different from
those of the brucite-based II-III LDHs.
Simulations of the powder X-ray diffraction (PXRD) pat-
terns were carried out using the program DIFFaX.22,23 Within
the DIFFaX formalism, a solid is treated as a stacking of layers
of atoms and the PXRD pattern computed by integrating the
diffraction intensity layer by layer. This is ideally suited for
layered materials where the layers exist naturally as a conse-
quence of anisotropic bonding. The transformation matrices
required to define a monoclinic cell in DIFFaX as well as the
input used for the simulation are given as Supporting Informa-
tion, SI.1. and SI.2, respectively.
The LDH composition was determined by a combination of
chemical and instrumental methods. Al3þ content was estimated
gravimetrically as Al2O3. The divalent metal ion content was
estimated by Atomic Absorption Spectroscopy (Varian AA240)
and sulfate by Ion Chromatography (Metrohm Model 861
Advanced Compact ion chromatography fitted with a Metrosep
A SUPP5 anion column and conductivity detector). The results
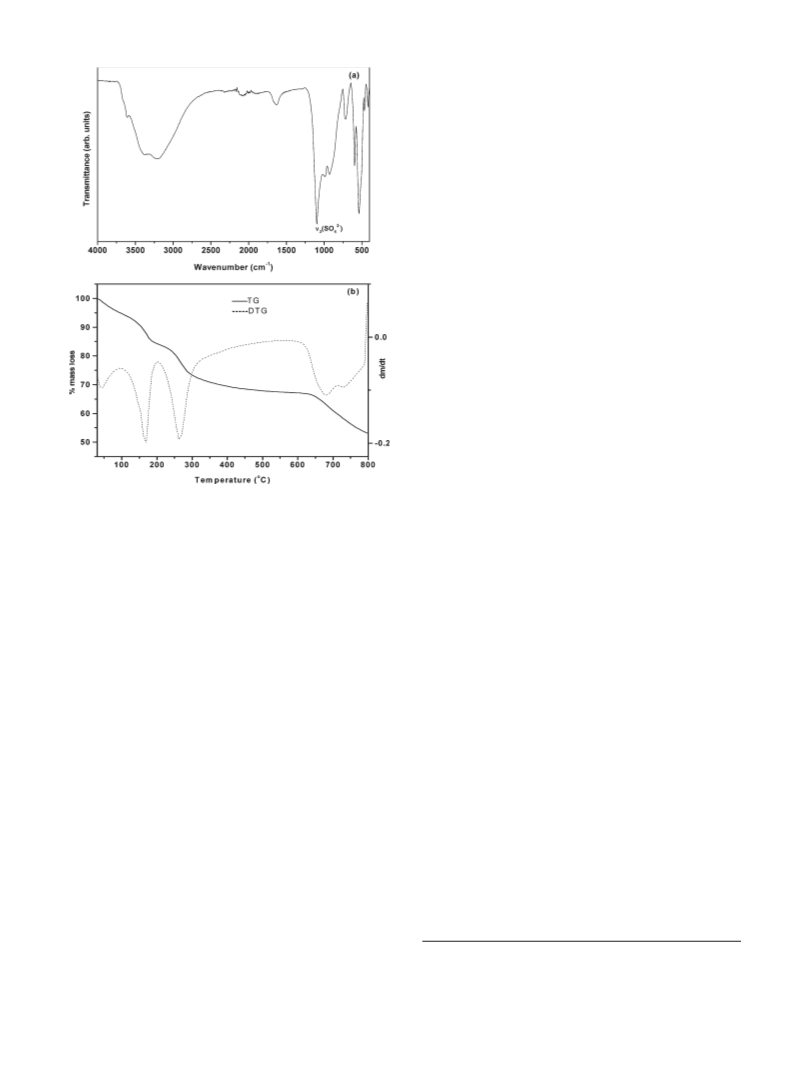
are given in Table 1. The intercalated water content is deter-
mined from thermogravimetry (TGA) data (Mettler Toledo
TG/SDTA Model 851e system, 30-800 °C, heating rate
5° min-1, flowing air). IR spectra were recorded using a Bruker
Alpha-P FTIR spectrometer (ATR mode, 400-4000 cm-1
,
4 cm-1 resolution).
Results and Discussion
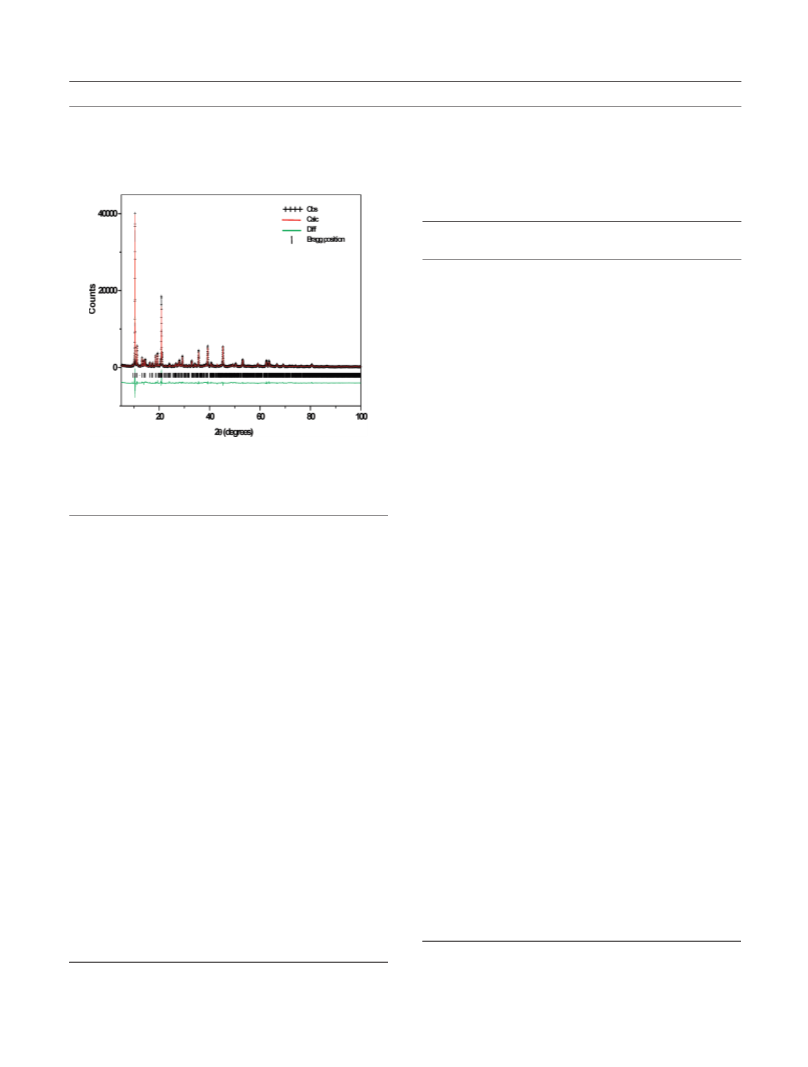
The PXRD pattern of the sample (ZA-1) obtained by
hydrothermally treating bayerite in ZnSO4 solution (Figure 1)
is indexed to a monoclinic cell derived from the structure of
the mineral nickelalumite24 (Space group: P21/n, cell param-
Experimental Section
˚
˚
˚
eters: a = 10.305(3) A, b = 8.911(2) A, c = 17.141(2) A, β =
95.68(2)°) (See Supporting Information, SI.3 for the list of
d-spacings and their assignment). The sharp and symmetric
peaks in the PXRD pattern of sample ZA-1 is characteristic
of a high degree of crystallinity. We therefore first refine the
structure of this compound by the Rietveld method.
LDH Synthesis. Al(OH)3-based II-III LDHs were prepared
by soaking 0.5 g of bayerite (prepared using a literature procedure)10
in ∼2.8 M (40 mL) ZnSO4 solution and hydrothermally treating the
suspension in a Teflon lined autoclave (50% filling) at 150 °C for
24 h. We call this sample as ZA-1. Other compounds were similarly
prepared using MSO4 or M(NO3)2 solutions (M = Co2þ, Ni2þ).
For comparison a sample, ZA-2, was prepared by soaking 1.96 g
of ZnO in 20 mL of 0.75 M Al2(SO4)3 solution and stirring the
suspension at 80 °C for 24 h. All the products were washed 4-5times
with distilled water, once with isopropanol, and dried in an air oven
at 65 °C.
We use the structure of mineral nickelalumite24 as a model.
The nickelalumite structure is closely related to that of
bayerite/gibbsite and can be described as arising from the
incorporation of divalent cations into half the octahedral
vacancies of [Al20 (OH)6] to give a structure having the com-
position [M0.50
0.5Al2(OH)6][SO4]0.5 (M = Ni2þ). While the
All samples were characterized by powder X-ray diffraction
(Bruker D8 Advance Diffractometer, Cu KR radiation, λ =
composition of the ideal nickelalumite structure corresponds to
an Al/M2þ ratio of 4, elemental analysis (Table 1) indicates an
Al/Zn ratio of ∼3 for ZA-1. In the absence of any unitary phase
of Zn, the excess Zn is expected to partially substitute for Al3þ
˚
1.5418 A, reflection geometry). Data were collected at a con-
tinuous scan rate of 1° 2θ min-1 and a step size of 0.02° 2θ. For
Rietveld refinement, data were collected over 5-100° 2θ (step
size 0.02° 2θ, continuous scan rate of 10 s step-1).
yielding the layer composition [(Zn0.500.5)(Al1.8Zn0.100.1)-
Rietveld refinement was carried out using the GSAS software
package.20 For the refinement, a TCH-pseudo-Voigt line shape
function21 (Profile Function 2) with eight variables was used to fit
the experimental profile. A cosine Fourier series function with eight
terms was used to refine the background. Bond distance restraints
(OH)6)]0.6þ. This kind of substitution leads to a decrease
in the layer charge. The observed sulfate content matches
with the layer charge, and is less than the sulfate content
of the model structure. As the number of interlayer atoms
is determined by the crystallographically allowed sites, the
shortfall in the atom count because of sulfate deficiency is
made up by the inclusion of water molecules. The nickelalu-
mite structure of the nominal composition allows for 1.5 H2O
molecules/formula unit. The TGA data of ZA-1 indicates a
higher water content of 1.8 molecules. The oxygen atoms of
the additional water molecules are assigned to the same sites
as the oxygen atoms of the sulfate ions. This enhances the
SOFs of all the sulfate oxygen atoms to 1 to yield the formula
˚
˚
were imposed on Al-O (1.940 ( 0.05 A), Zn-O (2.050 ( 0.05 A),
˚
and S-O (1.48 ( 0.03 A) bonds to enable stable refinement. The
relative weights of these restraints were reduced as the refinement
progressed with the final soft restraint weight factor being 50.
(15) Rothbauer, R.; Zigan, F.; O’Daniel, H. Z. Kristallogr. 1967, 125, 317.
(16) Fogg, A. M.; Green, V. M.; Harvey, H. G.; O’Hare, D. Adv. Mater.
1999, 11, 1466.
(17) Fogg, A. M.; Dunn, J. S.; Shyu, S.-G.; Cary, D. R.; O’Hare, D.
Chem. Mater. 1998, 10, 351.
(18) Lei, L.; Millange, F.; Walton, R. I.; O’Hare, D. J. Mater. Chem. 2000,
10, 1881.
(19) Britto, S.; Thomas, G. S.; Kamath, P. V.; Kannan, S. J. Phys. Chem.
C 2008, 112, 9510.
(20) Larson, A. C.; Von Dreele, R. B. General Structure Analysis System
(GSAS); Los Alamos National Laboratory Report LAUR 86-748, 2004.
(21) Thomson, P.; Cox, D. E.; Hastings, J. B. J. Appl. Crystallogr. 1987,
20, 79.
(22) Treacy, M. M. J.; Deem, M. W.; Newsam, J. M. DIFFaX, Version
1.807.
(23) Treacy, M. M. J.; Newsam, J. M.; Deem, M. W. Proc. R. Soc. London
1991, A 433, 499.
(24) Uvarova, Y.; Sokolova, E.; Hawthorne, F. C.; Karpenko, V. V.;
Agakhanov, A. A.; Pautov, L. A. Can. Mineral. 2005, 43, 1511.

 Britto, Sylvia
Britto, Sylvia
 Kamath, P. Vishnu
Kamath, P. Vishnu