M. Bierenstiel, M. Schlaf
FULL PAPER
a PEG column (30 m ϫ 0.25 mm). The GC FID was calibrated
for cyclohexanol using naphthalene as an internal standard. All
experimental preparations were conducted in a dry-box under Ar
and/or using usual Schlenk technique on a vacuum line. Acetone,
cyclohexanone, and n-heptane were dried by distillation under Ar
from anhydrous CaCl2, anhydrous MgSO4, and potassium, respec-
tively, and then degassed and stored under Ar. DMF was dried
over BaO and distilled under reduced pressure, and then it was
degassed and stored under an Ar atmosphere. Cyclohexanol, cyclo-
hexanone, -galactose, -glucose, -mannose, naphthalene, tetra-
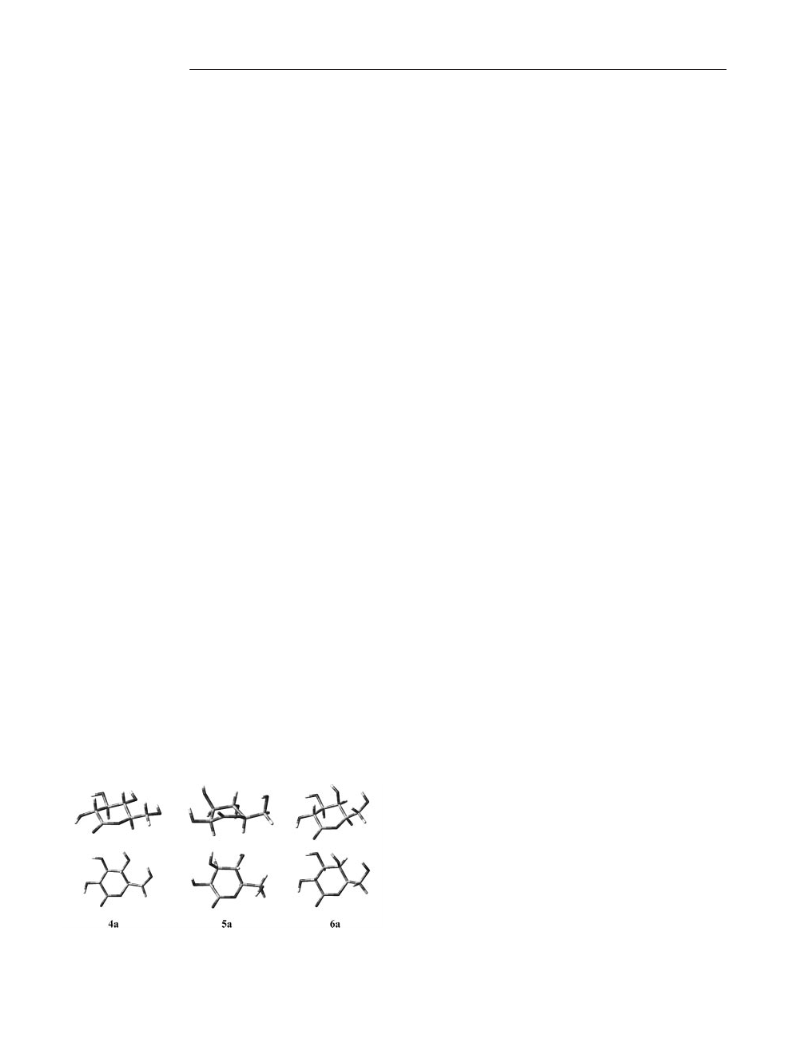
larities between the postulated ester hemiketal intermedi-
ates and the native conformation of the δ-lactones in
[D6]DMSO. The 4H3 conformation of 6a (Figure 2) is struc-
turally most closely related to that of the B1,4 conformation
of the intermediate requiring motion of only the C(1)ϭO
group [and, to a lesser extent, the C(3)ϪOH-3 group] from
below to above the plane defined by the C(2)ϪC(5)ϪO
atoms, i.e., the δ-galactonolactone is structurally ‘‘set-up’’
to attain the geometry required to isomerize to the γ-form,
which results in a low activation barrier for this process. phenyl-cyclopentadienone, acetone, chloroform, n-heptane, and
4
Ru3(CO)12 were purchased from commercial sources. All chemicals
were reagent grade and used as obtained without further purifi-
cation unless otherwise noted. Shvo’s catalyst (7) was prepared ac-
cording to literature procedures.[15] The best results were obtained
when the synthesis was carried out inside the dry-box. Oil-pump
vacuums applied in the isolation of the lactones were Յ 30 mTorr.
Starting from the same H3 conformation, the equatorial
position of the OH-4 unit in 4a requires that the 1,4-linkage
is established below the C(2)ϪC(5)ϪO plane to give a B1,4
conformation, which requires motion of both the C(1) and
C(4) centers and, thus, leads to a higher activation barrier
and a slower rearrangement. In comparison, an even-more-
extensive molecular motion is required for the B2,5 Ǟ B1,4
conformational change that 5a has to undergo to establish
the 1,4-linkage. In addition, there is an unfavorable cis in-
teraction between the OH-2 and OH-3 groups present in
the ester hemiketal intermediate that we also assume to be
General Procedure A — Oxidation of D-Galactose: -Galactose
(396 mg, 2.20 mmol), Shvo’s catalyst (1.25 mol %, 30 mg), dry
cyclohexanone (20 mL), and dry DMF (10 mL) were combined in
a 50-mL Schlenk flask under Ar. Naphthalene (50 mg) was added
as an internal GC standard. The reaction was stirred at 21 °C and
present in the actual transition state of the similar structure monitored by GC by following the appearance of a peak for cyclo-
hexanol. Once the reaction was complete, the mixture was trans-
ferred to a 50-mL one-necked flask and the solvents were evapo-
rated under an oil-pump vacuum at Յ 45 °C. Anhydrous acetone
(10 mL) was added to the remaining solids and then the mixture
was sonicated for 1 min, transferred to a centrifuge tube, and cen-
trifuged at 2500 rpm for 3 min. The supernatant solution was care-
fully removed using a Pasteur pipette. The acetone extraction was
repeated, usually three times, until the supernatant solution was
colorless. The residual white solid was dried under an oil-pump
that further raises the activation barrier and results in the
comparatively very slow rate of the 5a Ǟ 5b rearrangement
in [D6]DMSO.
Conclusions
δ-Galactonolactone can be prepared and isolated from
DMF solutions by transfer dehydrogenating -galactopyr- vacuum to constant weight. Isolated yield: 206.4 mg (54%). The
reaction can be performed at one- or two-thirds of this scale and
carried out analogously for α--mannose. See text for the γ/δ ratio.
anose using Shvo’s catalyst and cyclohexanone as the hy-
drogen acceptor. In [D6]DMSO, δ-galactonolactone exists
predominantly in the H3 conformation, a structure closely
4
General Procedure
B — Oxidation of D-Glucose: -Glucose
related to a bicyclic ester hemiketal postulated to be the
intermediate in its rearrangement to the corresponding γ-
lactone, which explains its instability against this isomeriz-
ation. In comparison, δ--glucono and δ--mannonolac-
tone are much more stable against this isomerization since
their native conformations do not, or less closely, resemble
the shapes of their corresponding ester hemiketal intermedi-
ates.
(132 mg, 0.73 mmol), Shvo’s catalyst (1.25 mol %, 10 mg), and dry
cyclohexanone (10 mL) were combined in a 15-mL Schlenk tube
under an Ar atmosphere. Naphthalene (50 mg) was added as an
internal GC standard. The suspension was stirred at 45 °C and
monitored by GC by following the appearance of a peak for cyclo-
hexanol. After completion of the reaction, the mixture was trans-
ferred to a centrifuge tube and centrifuged at 2500 rpm for 1 min.
The supernatant orange solution was carefully removed using a
Pasteur pipette and the residual white solid was dried under an
oil-pump vacuum. In cases where the solids were slightly colored,
anhydrous acetone (10 mL) was added and the extraction pro-
cedure repeated. See the text for isolated yields and γ/δ ratios.
Experimental Section
NMR spectra (400 MHz, 1H; 100 MHz, 13C) were measured in
[D6]DMSO with DMSO (δ ϭ 2.49 ppm, 1H; δ ϭ 39.5 ppm, 13C)
and deuterated chloroform (δ ϭ 7.24 ppm, 1H; δ ϭ 77.0 ppm, 13C)
as internal references. DMSO was stored inside a dry-box under
General Procedure for the NMR Spectroscopy Experiments: Sugar
lactone (10 mg) was dissolved in [D6]DMSO (0.6 mL; stored under
inert amosphere over activated 4-A molecular sieves). The 1H
˚
˚
NMR spectra were recorded at the desired temperature after al-
lowing the solution to equilibrate for approximately 10 min at
each temperature.
Ar over 4-A activated molecular sieves. For variable-temperature
measurements, the spectrometer temperature controller unit was
calibrated using a bimetal thermometer directly inserted into the
probe. The γ:δ ratios of the lactones were determined by integration
1
of their signals in their H NMR spectra. Simulations of NMR
Supporting Information (see also the footnote on the first page of
this article): A comprehensive collection of 1H and 13C NMR spec-
troscopic data (COSY, HSQC), with images of spectra for all lac-
tones. Atomic-coordinate data from the Gaussian 98/03 DFT cal-
culations for all lactones (total 65 pages).
spectra were carried out using the SpinWorks program (Version
2.2).[25] DFT calculations were carried on a PC using the Gaussian
98 and Gaussian 03 suite of programs. No imaginary frequencies
were observed in the calculations. GC analyses were performed on
1480
2004 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Eur. J. Org. Chem. 2004, 1474Ϫ1481

 Bierenstiel, Matthias
Bierenstiel, Matthias
 Schlaf, Marcel
Schlaf, Marcel