_
J. Zabin´ski et al. / Journal of Molecular Structure 833 (2007) 74–81
76
New compounds 1 and 2 were obtained in the course
119.17 (C-7, C-70), 141.82 (C-1, C-10), 153.94 (C-2, C-20,
0
ꢀ
of a four-step synthesis (Scheme 1). It involves bromina-
tion of 4-hydroxybenzaldehyde 1a by the procedure given
in [11] yielding 97% of 4-hydroxy-3,5-dibromobenzalde-
hyde 1b, substitution of both bromine atoms by the
methoxy group [12] to afford 4-hydroxy-3,5-dimethoxy-
benzaldehyde 1c (yield 92%), and conversion of 1c into
4-hydroxy-3,5-dimethoxybenzonitrile 1d (yield 77%) [13].
The synthetic route completed O-alkylation of 1d with
bis(2-chloroethyl)ether (leading to 1) or 1,5-dibromopen-
tane (leading to 2) during our work-up procedure based
on [14,15]. All the literature procedures were slightly
modified.
C-6, C-6 ) ppm. IR (KBr): m ¼ 2947, 2843, 2222, 1582,
1501, 1454, 1335, 1242, 1130, 1072 cmꢀ1
.
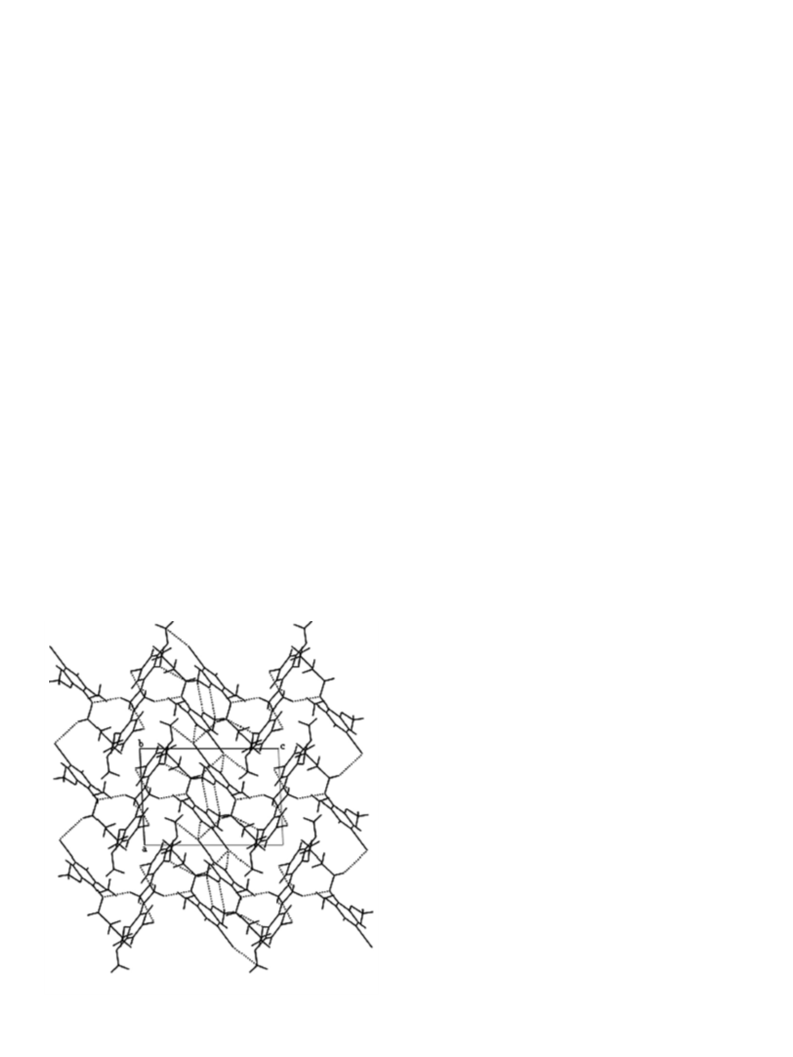
2.2. Crystallography
The crystals of 1 suitable for X-ray analysis were grown
from acetone by slow evaporation. Diffraction data were
collected on an Oxford Diffraction KM4CCD diffractome-
ter [16] at 296 K, using graphite-monochromated MoKa
radiation. A total of 1072 frames were measured in six sep-
arate runs. The x-scan was used with a step of 0.75ꢁ, two
reference frames were measured after every 50 frames, they
did not show any systematic changes either in peaks
positions or in their intensities. The unit cell parameters
were determined by least-squares treatment of setting
angles of 4571 highest-intensity reflections selected from
the whole experiment. Intensity data were corrected for
the Lorentz and polarization effects [17]. The structures
were solved by direct methods with the SHELXS-97 pro-
gram [18] and refined with full-matrix least-squares by
1,5-bis(4-cyano-2,6-dimethoxyphenoxy)-3-oxapentane 1
-4-hydroxy-3,5-dimethoxybenzonitrile 1d (1.79 g, 0.01 mol)
and bis(2-chlorethyl)ether (0.72 g, 0.005 mol) was added
to 25 ml of 1-methyl-2-pyrrolidone together with K2CO3
(3.46 g, 0.025 mol). The mixture was heated with stirring
in 150 ꢁC for 90 min, and then poured into water
(200 ml) with ice (50 g) to obtain brown precipitate. The
formed solid was filtered and washed with H2O. The pre-
cipitate was recrystallized from ethanol to give 1.89 g
(88% yield) of 1,5-bis(4-cyano-2,6-dimethoxyphenoxy)-3-
oxapentane 1, as the brown crystals. M.p. 124–125 ꢁC.
-C22H24N2O7 (428.44): calcd C 61.68, H 5.65, N 6.54%;
found C 61.32, H 5.56, N 6.32%.–1H NMR (400.13 MHz
CDCl3): 3.857 (s broad, 16H, 13-OCH3, 12-OCH3, 130-
OCH3, 120-OCH3, H-9, H-90), 4.194 (t, J = 4.8 Hz, 4H,
H-8, H-80), 6.854 (s, 4H, H-3, H-30, H-5, H-50) ppm. 13C
NMR (100.62 MHz, CDCl3): 56.49 (OCH3), 70.56 (C-9,
C-90), 72.60 (C-8, C-80), 106.90 (C-4, C-40), 109.50 (C-3,
2
the SHELXL-97 program [19]. The function R w(jFoj ꢀ
2 2
jFcj ) was minimized with wꢀ1=[r2(Fo)2+(0.0920P)2],
where P ¼ ðF 2o þ 2F 2cÞ=3. All non-hydrogen atoms were
refined with anisotropic thermal parameters. The positions
of hydrogen atoms were generated geometrically and
refined as a riding model. Thermal parameters of all hydro-
gen atoms were calculated as 1.2 (1.5 for methyl groups)
times Ueq of the respective carrier carbon atom.
2.3. NMR spectra
C-30, C-5, C-50),0 119.16 (C-7, C-70), 141.55 (C-1, C-10),
153.80 (C-2, C-2 , C-6, C-6 ) ppm. – IR (KBr) m ¼ 2943,
1H NMR and 13C NMR 1D and 2D spectra in solu-
tion were recorded with a Bruker Avance DMX 400. The
solid state 13C CP/MAS NMR spectra were acquired on
a Bruker Avance DMX 400. Powdered samples were
spun at 10 kHz. Contact time of 2 ms, repetition time
of 8 s, and spectral width of 44 kHz were used for accu-
mulation of 1,000 scans. Chemical shifts d [ppm] were
references to TMS. Temperature measurements were per-
formed using a Bruker B-VT 1000 E unit with a 10 K
temperature increment in the range 263–358 K. Nonprot-
onated carbons and methyl groups were selectively
observed by dipolar-dephasing experiment with delay
time 50 ls.
0
ꢀ
2843, 2226, 1578, 1497, 1450, 1416, 1338, 1238, 1130,
1061 cmꢀ1
.
1,5-bis(4-cyano-2,6-dimethoxyphenoxy)pentane 2 - 4-hy-
droxy-3,5-dimethoxybenzonitrile 1d (1.79 g, 0.01 mol), 1,5-
dibromopentane (1.15 g, 0.005 mol) and K2CO3 (2.07 g,
0.015 mol) was added to 25 ml of 1-methyl-2-pyrrolidone.
The mixture was allowed to stand at 100 ꢁC for 90 min
while stirring. The resulting hot solution was added to
water (200 ml) with ice (50 g). The formed brown solid
was filtered and washed with H2O. The precipitate was
recrystallized from ethanol to give 1.84 g (86% yield) of
1,5-bis(4-cyano-2,6-dimethoxyphenoxy)pentane 2, as the
brown crystals. Analytical purity sample was obtained by
column chromatography (Merck Silicagel 60, 230–400
mesh ASTM) with C2H4Cl2 as eluent. M.p. 119–120 ꢁC.
C23H26N2O6 (426.47): calcd C 64.78, H 6.14, N 6.57%;
2.4. Molecular modeling details
Crystallographic atom coordinates for 1 and only the
optimized ones for 2 were used for computation of shield-
ing constants r [ppm] of 13C atoms as the help in assign-
ment of resonances in the solid state NMR spectra. We
employed the DFT method with B3LYP/6–311(d,p) hybrid
functional for structure optimization, and the CHF–GIAO
approach for the NMR shielding constants computations
using Gaussian 03 program [20]. To gain an insight into
conformational equilibria of compound 2 we employed
found
C
64.84,
H
6.06,
N
6.61%.– 1H NMR
(400.13 MHz, CDCl3): 1.642 (multiplet, 2H, H-10), 1.820
(quintet, J = 6.4 Hz, 4H, H-9, H-90), 3.858 (s, 12H,
11-OCH3, 12-OCH3, 110-OCH3, 120-OCH3), 4.045 (t,
J = 6.4 Hz, 4H, H-8, H-80), 6.862 (s, 4H, H-3, H-30, H-5,
H-50) ppm. 13C NMR (100.62 MHz, CDCl3): 22.10
(C-10), 29.87 (C-9, C-90), 56.49 (OCH3), 73.64 (C-8,
C-80), 106.66 (C-4, C-40), 109.60 (C-3, C-30, C-5, C-50)

 Zabiński, Jerzy
Zabiński, Jerzy
 Wolska, Irena
Wolska, Irena
 Maciejewska, Dorota
Maciejewska, Dorota