H. Yamada et al.
FULL PAPER
PDO (0.260 g, 0.770 mmol, 21%) as white crystals and 6,13-di-
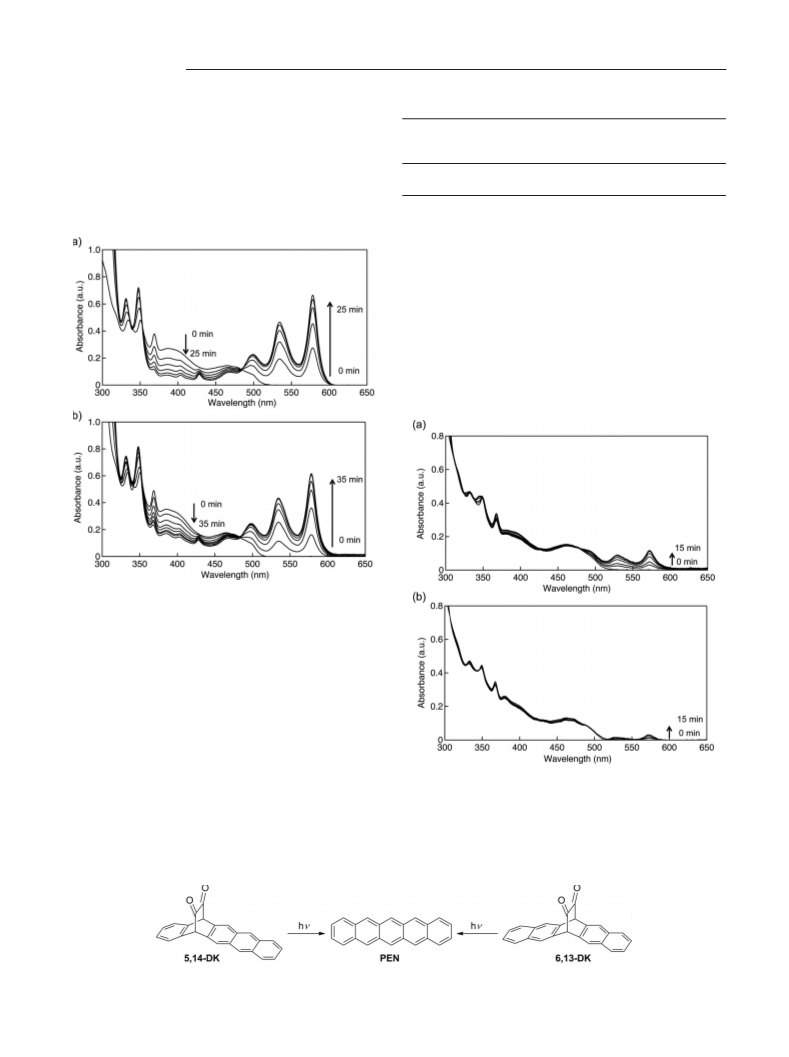
perimental conditions, both 5,14-DK or 6,13-DK absorbed essen-
tially all incident light. The photochemical reaction was monitored
by using a JASCO UV/Vis/NIR V-570 spectrophotometer. The
hydro-15,16-dihydroxy-6,13-ethanopentacene (6,13-PDO; 0.710 g,
1
2.10 mmol, 58%). 5,14-PDO: M.p. 275–277 °C.
H NMR
(400 MHz, CDCl
3
, TMS): δ = 2.18 (br., 2 H, OH), 4.23 (br., 2 H), quantum yields were determined from the increase in absorbance
4
7
.56 (br., 2 H), 7.25 (m, 2 H), 7.44–7.72 (m, 2 H), 7.89 (s, 2 H), due to PEN (578 nm) at the beginning of the reaction.
.97–7.95 (m, 2 H), 8.33 (s, 2 H) ppm. 1 C NMR (100 MHz,
3
Photochemical Reactions in Films: Compound 5,14-DK (10 mg) was
CDCl
27.00, 127.88, 136.80 ppm. MS (FAB): m/z = 339 [M+ + 1].
(338.41): calcd. C 85.18, H 5.36; found C 85.22, H 5.30.
3
, TMS): δ = 51.26, 68.81, 122.79, 125.11, 125.70, 126.54,
dissolved in hot CHCl
coated on glass at 1000 rpm for 20 s. The absorption spectrum of
,14-DK in the film was measured. Then the film was irradiated
3
(1 mL), and the solution (100 μL) was spin-
1
24 18 2
C H O
5
Synthesis of 5,14-Dihydro-5,14-ethanopentacene-15,16-dione (5,14-
DK): Trifluoroacetic anhydride (2.1 mL, 15.1 mmol) was added
dropwise to a mixture of dry DMSO (1.0 mL, 14.0 mmol) and dry
with a 460 W metal halide lamp through a blue filter in a glove box
for 90 min, and the absorption spectrum of PEN was measured.
X-ray Analysis: Single crystals of 5,14-DK suitable for X-ray dif-
fraction analysis were obtained by slow diffusion of heptane into a
solution of 5,14-DK in CH Cl . The crystals were mounted in Litho
2 2
Loops (purchased from Protein Wave). The diffraction data was
collected at 25 °C with a Rigaku VariMaxRAPID/a imaging plate
CH
,14-PDO (0.306 g, 0.905 mmol) dissolved in a mixture of dry
DMSO (10 mL) and dry CH Cl (7 mL) was added dropwise. After
stirring for 90 min, N,N-diisopropylethylamine (4.50 mL,
5.8 mmol) was added dropwise to the reaction mixture. The solu-
2 2
Cl (10 mL) at –60 °C under argon. After stirring for 10 min,
5
2
2
2
diffractometer with graphite-monochromated Cu-K
with a Rigaku Mercury-8 diffractometer with graphite-monochro-
mated Mo-K radiation equipped with a CCD detector. The dif-
fraction data were processed with CrystalStructure of the Rigaku
α
radiation or
tion was stirred at –60 °C for 60 min and warmed to room temp.
before 3 m HCl (50 mL) was added to the mixture. The mixture
α
was extracted with CH
washed with water and brine and dried with Na
of the solvent in vacuo, the residue was purified by column
chromatography on silica gel with CH Cl and recrystallized from
toluene to give 5,14-DK as yellow crystals (0.157 g, 0.469 mmol,
2
Cl
2
, and the combined organic layers were
2
SO . After removal
4
[25]
program, solved with the SIR-97 program, and refined with the
SHELX-97 program.[
26]
2
2
Supporting Information (see footnote on the first page of this
article):·TD-DFT calcuration, change in absorption spectra before
and after photolysis in film, CV and DPV of 5,14-DK; change in
absorption spectra of 6,13-DK during photolysis in solution.
5
5
2
2%). M.p. Ͼ 300 °C. 1H NMR (400 MHz, CDCl
.19 (s, 2 H), 7.39–7.41 (m, 2 H), 7.48–7.52 (m, 4 H), 7.99–8.01 (m,
H), 8.07 (s, 2 H), 8.42 (s, 2 H) ppm. 13C NMR (75 MHz, CDCl
3
, TMS): δ =
3
,
TMS): δ = 60.46, 125.45, 126.03, 126.36, 126.50, 128.13, 129.60,
30.95, 131.21, 132.16, 134.90, 184.84 ppm. MS (FAB): m/z = 336
1
+
[M
+ 1]. C24
6.50, H 4.59.
H
14
O
2
(334.37): calcd. C 86.21, H 4.22; found C Acknowledgments
8
We thank Prof. Atsushi Wakamiya, Institute of Chemical Research,
Kyoto University for his valuable discussion on TD-DFT calcula-
tions. This work was partially supported by the Ministry of Educa-
tion, Culture, Sports, Science and Technology (MEXT), Japan
through a Grants-in-Aid (No. 22350083 to H. Y.) and the Green
Photonics Project in NAIST sponsored by the Ministry of Educa-
tion, Culture, Sports, Science and Technology, MEXT, Japan.
Leigh McDowel is gratefully acknowledged for proofreading this
manuscript.
Theoretical Calculations: All DFT calculations were achieved with
[
22]
the Gaussian 09 program package. The geometry was fully opti-
mized at the Becke’s three-parameter hybrid functional combined
with the Lee–Yang–Parr correlation functional abbreviated as the
B3LYP level of density functional theory with 6-31G(d) basis set.
Equilibrium geometries were verified by frequency calculations,
where no imaginary frequency was found. Based on the B3LYP/6-
3
1G(d)-optimized geometry, TD-DFT calculations were conducted
[23]
at the CAM-B3LYP/6-31G(d) level of theory.
Electrochemical Measurements: The cyclic voltammetry measure-
ments of investigated compounds were performed with a BAS elec-
trochemical analyser in deaerated acetonitrile containing
[
1] a) M. Bendikov, F. Wudl, D. F. Perepichka, Chem. Rev. 2004,
104, 4891–4945; b) J. E. Anthony, Chem. Rev. 2006, 106, 5028–
5048.
–
1
4 6
nBu NPF as a supporting electrolyte at 298 K (100 mVs ). The
[
[
2] D. Knipp, R. A. Street, B. Krusor, R. Apte, J. Ho, J. Non-
Cryst. Solids 2002, 299–302, 1042–1046.
glassy carbon working electrode was polished with BAS polishing
alumina suspension and rinsed with acetone before use. The
counter electrode was a platinum wire. The measured potentials
3] a) H. Klauk, M. Halik, U. Zschieschang, G. Schmid, W. Rad-
lik, W. Weber, J. Appl. Phys. 2002, 92, 5259–5263; b) H. Klauk,
M. Halik, U. Zschieschang, F. Eder, G. Schmid, C. Dehm,
Appl. Phys. Lett. 2003, 82, 4175–4177.
were recorded with respect to Ag/AgNO
Fc .
3
and normalized to Fc/
+
[
[
4] J. A. Nichols, D. J. Gundlach, T. N. Jackson, Appl. Phys. Lett.
Photochemical Reactions: The photocleavage reactions were carried
out in a quartz UV cell, which was irradiated with monochromatic
excitation light through a monochromator (Ritsu MC-10N) from
a 500 W xenon lamp (Ushio XB-50102AA-A), and monitored by
an OCEAN OPTICS HR-4000 high-resolution spectrometer sys-
tem with light source DH-2000-BAL. A standard actionometer
2003, 83, 2366–2368.
5] M. Kitamura, T. Imada, Y. Arakawa, Appl. Phys. Lett. 2003,
83, 3410–3412.
[6] D. Kumaki, M. Yahiro, Y. Inoue, S. Tokito, Appl. Phys. Lett.
2007, 90, 133511-3.
[
7] S. Yoo, B. Domercq, B. Kippelen, Appl. Phys. Lett. 2004, 85,
[
24]
5427–5429.
(K
3
[Fe(C
2
O
4
)
3
])
was used for quantum yield determination of
[
[
8] J. Yang, T.-Q. Nguyen, Org. Electron. 2007, 8, 566–574.
9] J. Sakai, T. Taima, T. Yamanari, Y. Yoshida, A. Fujii, M.
Ozaki, Jpn. J. Appl. Phys. 2010, 49, 032301–7.
10] K. Nomura, T. Oku, A. Suzuki, K. Kikuchi, G. Kinoshita, J.
Phys. Chem. Solids 2010, 71, 210–213.
the photochemical reactions of 5,14-DK and 6,13-DK in acetoni-
trile and toluene. A square quartz cuvette (10 mm i.d.) that con-
3
tained a deaerated solution (3.0 cm ) of 5,14-DK and 6,13-DK was
[
irradiated with monochromatized light (λ = 405 or 468 nm)
through a monochromator (Ritsu MC-10N) by using a 500 W
xenon lamp (Ushio XB-50102AA-A). Under the actinometry ex-
[11] a) A. R. Brown, A. Pomp, D. M. de Leeuw, D. B. M. Klaassen,
E. E. Havinga, P. Herwig, K. Müllen, J. Appl. Phys. 1996, 79,
1728
www.eurjoc.org
© 2012 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
Eur. J. Org. Chem. 2012, 1723–1729

 Aotake, Tatsuya
Aotake, Tatsuya
 Ikeda, Shinya
Ikeda, Shinya
 Kuzuhara, Daiki
Kuzuhara, Daiki
 Mori, Shigeki
Mori, Shigeki
 Okujima, Tetsuo
Okujima, Tetsuo
 Uno, Hidemitsu
Uno, Hidemitsu
 Yamada, Hiroko
Yamada, Hiroko