H. Uno et al. / Tetrahedron Letters 46 (2005) 1981–1983
1983
Fritz, S. E.; Martin, S. M.; Frisbie, C. D.; Ward, M. D.;
Toney, M. F. J. Am. Chem. Soc. 2004, 126, 4084.
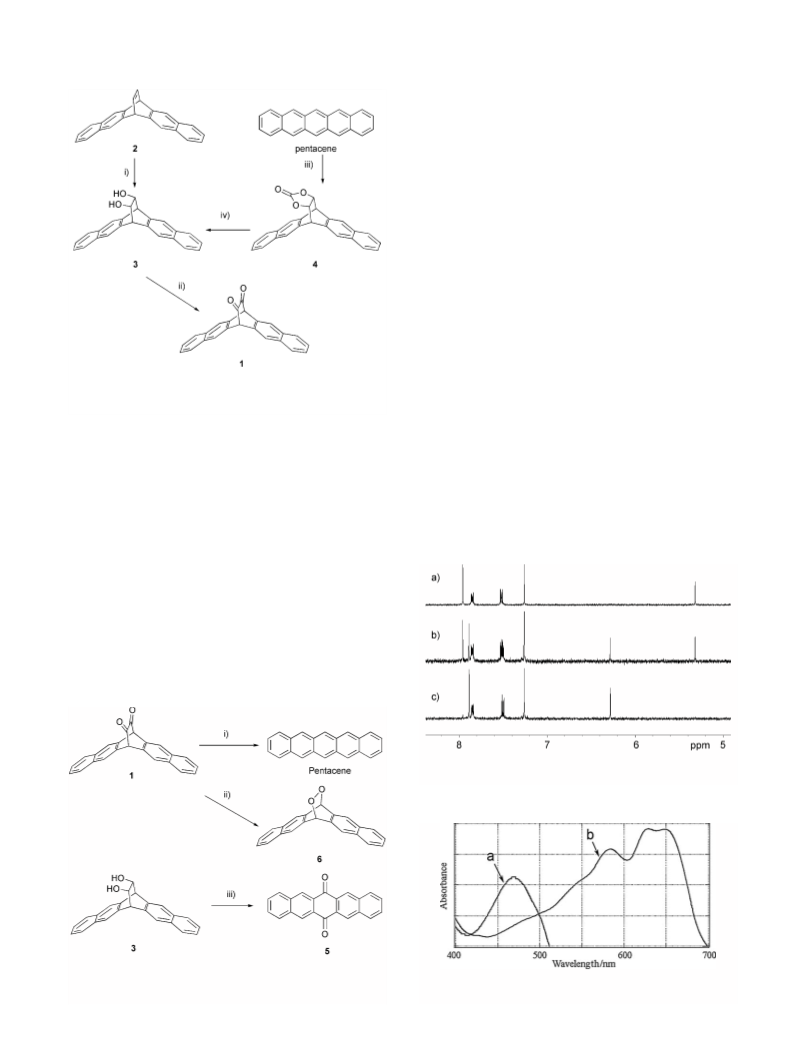
thermal decomposition of 3. When the sodium salt of 3
was heated at 100 °C in DMF,12 the obtained product
was 6,13-pentacenequinone (5, 89%) probably due to
air oxidation of pentacene formed.
3. Takahashi, T.; Kitamura, M.; Shen, B.; Nakajima, K. J.
Am. Chem. Soc. 2000, 122, 12876; Anthony, J. E.; Brooks,
J. S.; Eaton, D. L.; Parkin, S. R. J. Am. Chem. Soc. 2001,
123, 9482; Anthony, J. E.; Eaton, D. L.; Parkin, S. R. Org.
Lett. 2002, 4, 15; Meng, H.; Bendikof, M.; Mitchell, G.;
Helgeson, R.; Wudl, F.; Bao, Z.; Siegrist, T.; Kloc, C.;
Chen, C.-H. Adv. Mater. 2003, 15, 1090; Payne, M. M.;
Delcamp, J. H.; Parkin, S. R.; Anthony, J. E. Org. Lett.
2004, 6, 1609; Sakamoto, Y.; Suzuki, T.; Kobayashi, M.;
Gao, Y.; Fukai, Y.; Inoue, Y.; Sato, F.; Tokito, S. J. Am.
Chem. Soc. 2004, 126, 8138.
In conclusion, we achieved the first preparation of the
readily soluble and stable photo precursor of pentacene,
and showed the efficient conversion to pentacene both in
solid and in solution. This method provides not only a
new synthetic method of pentacene derivatives, which
are not easily accessible due to low solubility and stabil-
ity, but also new preparative methodology for the penta-
cene-based devices. Further studies about the device
preparation are under way.
4. (a) Brown, A. R.; Pomp, A.; de Leeuw, D. M.; Klaassen,
D. B. M.; Havinaga, E. E.; Herwig, P.; Mullen, K. J. Appl.
¨
Phys. 1996, 79, 2136; Herwig, P. T.; Mullen, K. Adv.
¨
Mater. 1999, 11, 480; (b) Miao, Q.; Nguyen, T.-Q.;
Someya, T.; Blanchet, G. B.; Nuckolls, C. J. Am. Chem.
Soc. 2003, 125, 10284; Vets, N.; Smet, M.; Dehaen, W.
Tetrahedron Lett. 2004, 45, 7287.
Acknowledgements
Partial financial support from Canon inc. is greatly
acknowledged. We also thank Professor Nagao Azuma
for permission of our use of an ultra-high Hg lamp.
5. Very recently, deposition of pentacene from a precursor by
photochemically generated proton and its application for
an EFT device were reported, see: Weidkamp, K. P.;
Afzali, A.; Tromp, R. M.; Hamers, R. J. J. Am. Chem.
Soc. 2004, 126, 12740.
References and notes
6. Fleming, I. Pericyclic Reactions; Oxford University Press:
Oxford, 1999.
7. Strating, J.; Zwanenburg, B.; Wagenaar, A.; Udding, A.
C. Tetrahedron Lett. 1969, 125.
1. Lin, Y. Y.; Gundlach, D. J.; Jackson, T. N. Annu. Device
Res. Conf. Dig. 1996, 54, 80; Lin, Y. Y.; Gundlach, D. J.;
Nelson, S.; Jackson, T. N. IEEE Trans. Electron. Dev.
1997, 44, 1325.
8. Chollet, A.; Wismer, M.; Vogel, P. Tetrahedron Lett. 1976,
47, 4271; Gabioud, R.; Vogel, P. Tetrahedron 1980, 36,
149.
2. Brown, R.; Pomp, A.; Hart, C. M.; de Leeuw, D. M.
Science 1995, 270, 972; Nelson, S. F.; Lin, Y.-Y.;
Gundlach, D. J.; Jackson, T. N. Appl. Phys. Lett. 1998,
72, 1854; Dimitrakopoulos, C. D.; Purushothaman, S.;
Kymissis, J.; Callegari, A.; Shaw, J. M. Science 1999, 283,
822; Meyer zu Heringdorf, F.-J.; Reuter, M. C.; Tromp,
R. M. Nature 2001, 412, 517; Dimitrakopoulos, C. D.;
Malenfant, P. R. L. Adv. Mater. 2002, 14, 99; Baude, P.
F.; Ender, D. A.; Maase, M. A.; Kelley, T. W.; Muyres, D.
V.; Theiss, S. D. Appl. Phys. Lett. 2003, 82, 3964; Lee, J.;
Hwang, D. K.; Park, C. H.; Kim, S. S.; Im, S. Thin Solid
Films 2004, 12, 451; Park, J. H.; Kang, C. H.; Kim, Y. J.;
Lee, Y. S.; Choi, J. S. Mater. Sci. Eng., C 2004, 24, 27;
1
9. Selected data for 1: Yellow crystals, mp 320 °C; H NMR
d (CDCl3) 7.94 (4H, s), 7.84 (4H, m), 7.52 (4H, m) and
5.31 (2H, s); 13C NMR d (CDCl3) 185.2, 133.6, 131.9,
127.9, 127.0, 125.4, and 60.6; IR (KBr) 1755, 1736, and
756 cmÀ1; UV–vis (e, CHCl3) 463 (1400) nm.
10. Mancuso, J.; Swern, D. Synthesis 1981, 165.
11. Sparfel, D.; Gobert, F.; Rigaudy, J. Tetrahedron 1980, 36,
2225.
12. Similar lowering of the decomposition temperature was
observed, see: Uno, H.; Ishikawa, T.; Hoshi, T.; Ono, N.
Tetrahedron Lett. 2003, 44, 5163; Rajanbabu, T. V.;
Eaton, D. F.; Fukunaga, T. J. Org. Chem. 1983, 48, 652.

 Uno, Hidemitsu
Uno, Hidemitsu
 Yamashita, Yuko
Yamashita, Yuko
 Kikuchi, Makoto
Kikuchi, Makoto
 Watanabe, Hikaru
Watanabe, Hikaru
 Yamada, Hiroko
Yamada, Hiroko
 Okujima, Tetsuo
Okujima, Tetsuo
 Ogawa, Takuji
Ogawa, Takuji
 Ono, Noboru
Ono, Noboru