Photochemical Synthesis of Pentacene
FULL PAPER
[2] A. Kraft, ChemPhysChem 2001, 2, 163–165.
[3] C. D. Dimitrakopoulos, P. R. L. Malenfant, Adv. Mater. 2002, 14, 99–
117.
[4] O. D. Jurchescu, J. Baas, T. T. M. Palstra, Appl. Phys. Lett. 2004, 84,
3061–3063.
[5] a) D. Knipp, R. A. Street, B. Krusor, R. Apte, J. Ho, J. Non-cryst.
Solids 2002, 299–302, 1042–1046; b) H. Klauk, M. Halik, U. Zschie-
schang, F. Eder, G. Schmid, C. Dehm, Appl. Phys. Lett. 2003, 82,
4175–4177; c) J. A. Nichols, D. J. Gundlach, T. N. Jackson, Appl.
Phys. Lett. 2003, 83, 2366–2368; d) N. Kitamura, T. Imada, Y. Ara-
kawa, Appl. Phys. Lett. 2003, 83, 3410–3412.
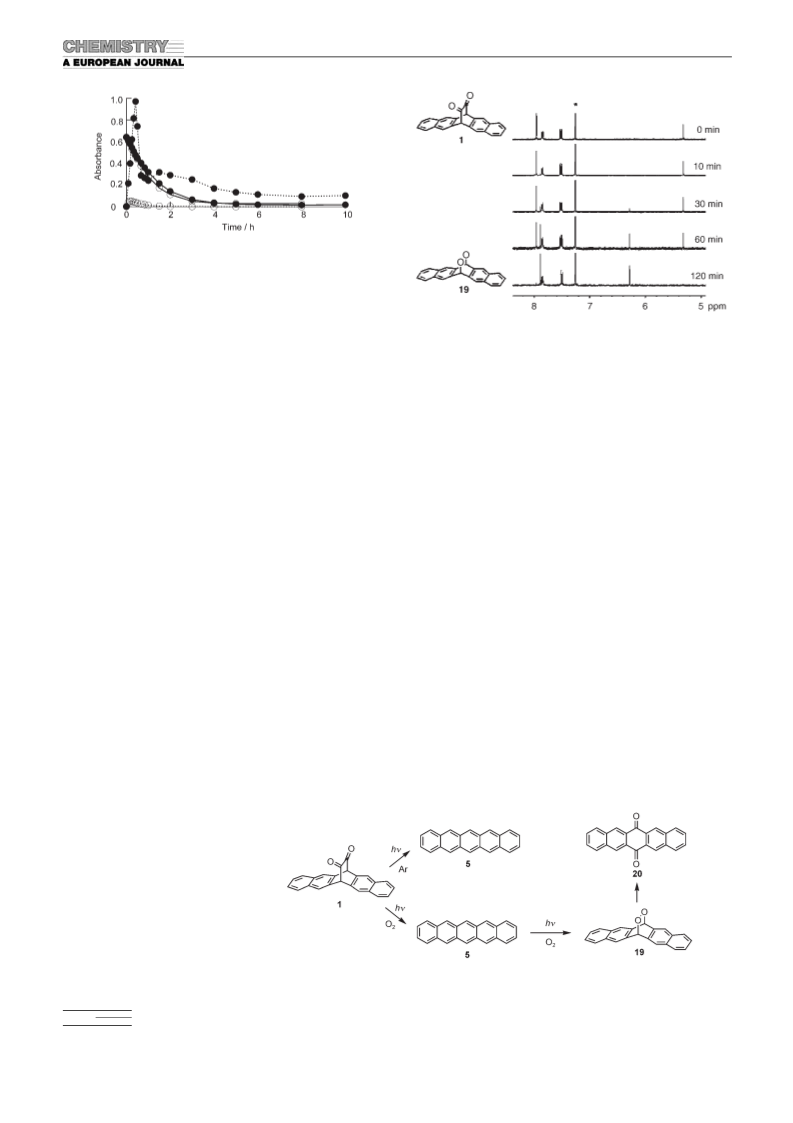
tolysis of the corresponding precursors, 2 and 3, respectively.
These results suggest that this photolysis reaction is very
useful for the preparation of a variety of acenes with low
solubility from soluble diketone precursors in a single step,
both in solution and in a film condition, and, therefore,
could contribute to the development of acenes with high
mobilities.
[6] a) T. Takahashi, M. Kitamura, B. Shen, K. Nakajima, J. Am. Chem.
Soc. 2000, 122, 12876–12877; b) J. E. Anthony, J. S. Brooks, D. L.
Eaton, S. R. Parkin, J. Am. Chem. Soc. 2001, 123, 9482–9483;
c) J. E. Anthony, D. L. Eaton, S. R. Parkin, Org. Lett. 2002, 4, 15–
18; d) C. D. Sheraw, T. N. Jackson, D. L. Eaton, J. E. Anthony, Adv.
Mater. 2003, 15, 2009–2011; e) M. J. Joung, J. H. Ahn, S. Y. Kang,
K. H. Baek, S. D. Ahn, L. M. Do, C. A. Kim, G. H. Kim, I. K. You,
S. M. Yoon, K. S. Suh, Bull. Korean Chem. Soc. 2003, 24, 1862–
1864; f) M. M. Payne, S. A. Odom, S. R. Parkin, J. E. Anthony, Org.
Lett. 2004, 6, 3325–3328; g) M. M. Payne, S. R. Parkin, J. E. Antho-
ny, C.-C. Kuo, T. N. Jackson, J. Am. Chem. Soc. 2005, 127, 4986–
4987; h) S. H. Chan, H. K. Lee, Y. M. Wang, N. Y. Fu, X. M. Chen,
Z. W. Cai, H. N. C. Wong, Chem. Commun. 2005, 66–68.
[7] a) H. Meng, M. Bendikov, G. Mitchell, R. Helgeson, F. Wudl, Z.
Bao, T. Siegrist, C. Kloc, C.-H. Chen, Adv. Mater. 2003, 15, 1090–
1093; b) Y. Sakamoto, T. Suzuki, M. Kobayashi, Y. Gao, Y. Fukai, Y.
Inoue, F. Sato, S. Tokito, J. Am. Chem. Soc. 2004, 126, 8138–8140.
[8] a) A. R. Brown, A. Pomp, D. M. de Leeuw, D. B. M. Klaassen, E. E.
Havinga, P. Herwig, K. Müllen, J. Appl. Phys. 1996, 79, 2136–2138;
b) P. T. Herwig, K. Müllen, Adv. Mater. 1999, 11, 480–483; c) A.
Afzali, C. D. Dimitrakopoulos, T. L. Breen, J. Am. Chem. Soc. 2002,
124, 8812–8813; d) N. Vets, M. Smet, W. Dehaen, Tetrahedron Lett.
2004, 45, 7287–7289.
[9] K. P. Weidkamp, A. Afzali, R. M. Tromp, R. J. Hamers, J. Am.
Chem. Soc. 2004, 126, 12740–12741.
[10] A. Afzali, C. D. Dimitrakopoulos, T. O. Graham, Adv. Mater. 2003,
15, 2066–2069.
[11] a) J. Strating, B. Zwanenburg, A. Wagenaar, A. C. Udding, Tetrahe-
dron Lett. 1969, 10, 125–128; b) D. Sülzle, T. Weiske, H. Schwarz,
Int. J. Mass Spectrom. Ion Processes 1993, 125, 75–79; c) H. Chen,
J. L. Holmes, Int. J. Mass Spectrom. Ion Processes 1994, 133, 111–
119; d) M. B. Rubin, M. Kapon, J. Photochem. Photobiol. A 1999,
124, 41–46.
[12] H. Uno, Y. Yamashita, M. Kikuchi, H. Watanabe, H. Yamada, T.
Okujima, T. Ogawa, N. Ono, Tetrahedron Lett. 2005, 46, 1981–1983.
[13] M. B. Rubin, Top. Curr. Chem. 1985, 129, 1–56.
Experimental Section
General: Melting points were measured by using a Yanaco M500-D melt-
ing point apparatus. 1H NMR and 13C NMR spectra were recorded by
using a JEOL JNM-AL 400 spectrometer with tetramethylsilane as an in-
ternal standard. IR spectra were measured by using a Hitachi 270–30
with KBr disks. FAB and DI-EI mass spectra were measured by using a
JEOL JMS-MS700 V spectrometer. MALDI-TOF mass spectra were
measured by using a Voyager DE Pro (Applied Biosystems). Elemental
analyses were performed by using a Yanaco MT-5 elemental analyzer.
Nanosecond transient absorption measurements were made by using a
conventional nanosecond laser-photolysis system (UNISOKU TSP-1000
based). A dye-laser (UNISOKU TSP-611) operated with a courmarin-
110 ethanol solution excited by a Nd-YAG laser (Continuum Surelight-I,
THG 355 nm, 2 Hz) was used as the excitation light source (around
460 nm) for the transient absorption experiment.
Materials: All solvents and chemicals were of reagent grade quality, ob-
tained commercially, and used without further purification, except as
noted. For photoreactions or spectral measurements, spectral grades of
toluene and chloroform were purchased from Nacalai Tesque. Thin-layer
chromatography (TLC) and column chromatography were performed by
using Art. 5554 (Merck) and Silica Gel 60N (Kanto Chemical), respec-
tively.
Photoreactions: The photoreactions were performed by using Pyrex
round-bottomed flasks under an Ar atmosphere. Typically, a solution of
diketone 1 in toluene (2.0 mm) was irradiated through a thermal cut-off
filter and a blue filter to cut out UV light (transmission range>390 nm)
by using a super-high-pressure mercury lamp (Hamamatsu Photonics,
500 W). To monitor the reaction by recording UV/Vis spectra, the dike-
tone solution (0.53 mm) in a quartz UV cell was irradiated with mono-
chromatic excitation light through a monochrometer (Ritsu MC-10N) by
using a 500 W Xenon lamp (Ushio XB-50102 AA-A). The deposition of
a diketone on glass was performed by using a standard spin-coating
method (Oshigane, SC-200, 1000 rpm, 30 s) using a diketone solution dis-
solved in CHCl3. The film was irradiated by using a metal-halide lamp
(Nippon P. I. PCS-UMX250, 330 W) in a dry box.
[14] K. Ito, T. Suzuki, Y. Sakamoto, D. Kubota, Y. Inoue, F. Sato, S.
Tokito, Angew. Chem. 2003, 115, 1191–1194; Angew. Chem. Int. Ed.
2003, 42, 1159–1162.
[15] H. Uno, H. Watanabe, Y. Yamashita, N. Ono, Org. Biomol. Chem.
2005, 3, 448–453.
Acknowledgements
[16] Molecular packing in the crystal structure of diol 10 is shown in Fig-
ure S1 of the Supporting Information.
The authors are grateful to Prof. Nagao Azuma of Ehime University for
his help with using a super-high-pressure mercury lamp for the photore-
actions, and to Prof. Shin-ichi Nagaoka of Ehime University for his help
in operating a conventional nanosecond laser-photolysis system. This
work was partially supported by Canon Inc., and by Grants-in-Aid for
the Scientific Research C (16550016) and Scientific Research on Priority
Areas “Application of Molecular Spins: Nanomagnets to Biological Spin
Systems” (Area No. 769, 15087104) from the Japanese Ministry of Educa-
tion, Culture, Sports, Science, and Technology (MEXT). In addition, H.Y.
would like to thank the Hayashi Memorial Foundation for Female Natu-
ral Scientists for their financial support.
[17] M. M. Wright, M. E. Welker, J. Org. Chem. 1996, 61, 133–141.
[18] CCDC 270597 contains the supplementary crystallographic data for
this paper. These data can be obtained free of charge from The
data_request/cif.
[19] M. Nagano, T. Hasegawa, M. Norikatsu, J. Yamaguchi, K. Itaka, H.
Fukumoto, T. Yamamoto, H. Koinuma, Jpn. J. Appl. Phys. Part 2,
2004, 43, L315–L316.
[20] D. Sparfel, F. Gobert, J. Rigaudy, Tetrahedron 1980, 36, 2225–2235.
[21] Y. Kamura, I. Shirotani, H. Inokuchi, Y. Maruyama, Chem. Lett.
1974, 627–630.
[22] The same reaction was performed in an Ar atmosphere, and NMR
spectroscopy was used in an attempt to monitor the pentacene
peaks. Although the color of the reaction mixture changed to purple
during photoirradiation, precipitates of pentacene appeared during
[1] F. Würthner, Angew. Chem. 2001, 113, 1069–1071; Angew. Chem.
Int. Ed. 2001, 40, 1037–1039.
Chem. Eur. J. 2005, 11, 6212 –6220
ꢀ 2005 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
6219

 Yamada, Hiroko
Yamada, Hiroko
 Yamashita, Yuko
Yamashita, Yuko
 Kikuchi, Makoto
Kikuchi, Makoto
 Watanabe, Hikaru
Watanabe, Hikaru
 Okujima, Tetsuo
Okujima, Tetsuo
 Uno, Hidemitsu
Uno, Hidemitsu
 Ogawa, Takuji
Ogawa, Takuji
 Ohara, Keishi
Ohara, Keishi
 Ono, Noboru
Ono, Noboru