A R T I C L E S
Mondal et al.
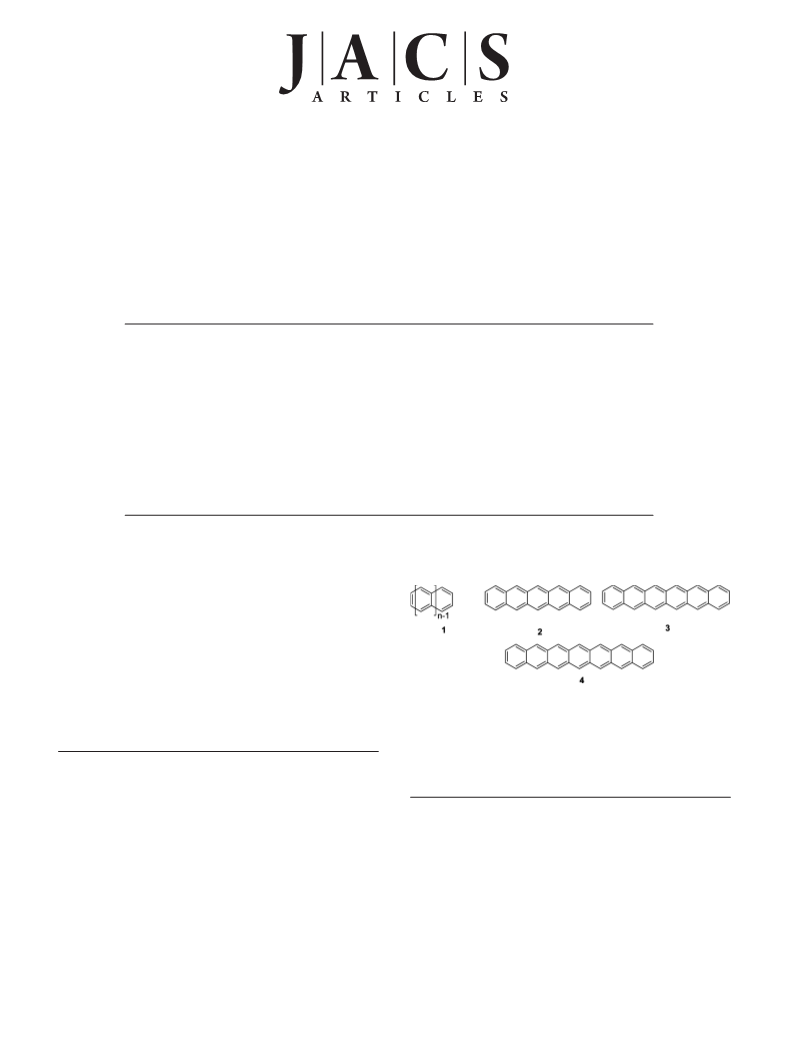
Scheme 1. Photogeneration of Higher Acenes 2-4 from the
results in HOMO-LUMO energy gaps that are smaller than
for other hydrocarbons with a similar number of aromatic rings.1
The energy gap quickly decreases with the length of the
conjugated π system, as seen in the bathochromic shift of the
absorption spectra1 and the decreasing singlet-triplet energy
splitting.27 This has two decisive and interrelated consequences:
the acenes are turning from insulators to organic molecular
p-type semiconductors with increasing length, and at the same
time they turn from prototypical stable aromatic compounds to
reactive species.31 The decrease in the reorganization energies,32
the increase in the charge carrier mobilities, and the band widths
make tetracene and the higher acenes useful materials for organic
electronic applications.2-5,33-35 Because of the quickly increas-
ing reactivity of acenes, only the members up to the size of
pentacene are characterized well. Hexacene already slowly
decomposes in solution at room temperature.36 The existence
of heptacene was controversial since the 1942 report by Clar,37
as repeated synthetic attempts at heptacene failed until 2006.
The lack of experimental information on the thermal stability
of heptacene led to several reinvestigations of the synthesis,
and the common conclusion was that heptacene appears to be
the limiting acene with respect to stability.37-40 Though
kinetically stabilized heptacene derivatives were obtained in
2005 by Payne et al.41 and very recently by Chun et al.42 and
Kaur et al.,43 the synthesis of the parent heptacene was only
reported recently and required the use of stabilizing matrices.44,45
However, an analysis of its IR and electronic absorption spectra
is lacking and no information is available on its singly charged
radical ions.
Diketones 5-7 and Photochemically Induced Charge Transfer To
Yield the Acene Radical Anions and Radical Cations Investigated
in the Present Work
(5-7).36,44,45,47 These compounds have two advantages in acene
synthesis: they are reasonably soluble in organic solvents, and
photolysis can be used efficiently in matrix isolation experi-
ments.45 Asreportedhere,theutilizationoftheStrating-Zwanenburg
reaction allowed us to investigate and compare the thermal
stabilities, spectral properties, and the photoinduced radical ion
formation of the three largest known acenes, pentacene (2),
hexacene (3), and heptacene (4), under identical conditions for
the first time (Scheme 1).
The successful syntheses of the reactive higher parent acenes
relied on the photochemical bisdecarbonylation, known as the
Strating-Zwanenburg46 reaction, of bridged R-diketones
Experimental Section
The R-diketones (5-7) were synthesized as described
previously.36,44,47 Matrix isolation experiments were carried out
according to standard techniques48 with APD CSW-20 displex
closed-cycle helium cryostats. The photoprecursors (5-7) were
sublimed out of a quartz tube that was resistively heated by a
tantalum coil to 163-170 °C, 193-198 °C, and 220-227 °C,
respectively. As the UV/vis spectrum of tetracene in argon is
not available, this was measured by subliming tetracene (Aldrich,
98%) at 75-80 °C (see Supporting Information, Figure S1). The
gaseous materials were trapped onto cold CsI (IR experiments)
or sapphire spectroscopic windows with a large excess of argon
(Messer Griesheim, 99.9999%) or xenon (Air Liquide, 4.0) gas
that were dosed to 2.0 sccm by a mass flow controller (MKS
Mass Flo type 247 four-channel read out). The experiments
performed in xenon are not discussed further here for the sake
of conciseness. The windows were kept at 30 and 55 K during
deposition of argon and xenon, respectively, by resistive heating
using an Oxford ITC 503 temperature controller. Photolysis of
the diketones and the photogenerated acenes were carried out
using a high-pressure mercury lamp equipped with 350-450 nm
mirror and a low pressure mercury lamp (Gra¨ntzel lamp) that
irradiates 185 and 254 nm wavelength light. IR spectra were
measured on Bruker IFS 66 and IFS 66/S spectrometers using a
resolution of 0.5 cm-1, while electronic absorption spectra were
measured using a Cary 5000 spectrometer.
(21) Houk, K. N.; Lee, P. S.; Nendel, M. J. Org. Chem. 2001, 66, 5517.
(22) Marian, C. M.; Gilka, N. J. Chem. Theory Comput. 2008, 4, 1501.
(23) Grimme, S.; Parac, M. ChemPhysChem 2003, 4, 292.
(24) Bendikov, M.; Duong, H. M.; Starkey, K.; Houk, K. N.; Carter, E. A.;
Wudl, F. J. Am. Chem. Soc. 2004, 126, 7416.
(25) Kadantsev, E. S.; Stott, M. J.; Rubio, A. J. Chem. Phys. 2006, 124,
134901.
(26) Sony, P.; Shukla, A. Phys. ReV. B 2007, 75, 155208.
(27) Hachmann, J.; Dorando, J. J.; Avile´s, M.; Chan, G. K.-L. J. Chem.
Phys. 2007, 127, 134309.
(28) Malloci, G.; Mulas, G.; Cappellini, G.; Joblin, C. Chem. Phys. 2007,
340, 43.
(29) Jiang, D.; Dai, S. J. Phys. Chem. A 2008, 112, 332.
(30) Qu, Z.; Zhang, D.; Liu, C.; Jiang, Y. J. Phys. Chem. A 2009, 113,
7909.
(31) Biermann, D.; Schmidt, W. J. Am. Chem. Soc. 1980, 102, 3163.
(32) Winkler, M.; Houk, K. N. J. Am. Chem. Soc. 2007, 129, 1805.
(33) Wu¨rthner, F. Angew. Chem., Int. Ed. 2001, 40, 1037.
(34) Dimitrakopoulos, C. D.; Malenfant, P. R. L. AdV. Mater. 2002, 14,
99.
(35) Kitamura, M.; Arakawa, Y. J. Phys.: Condens. Matter 2008, 20,
184011.
(36) Mondal, R.; Adhikari, R. M.; Shah, B. K.; Neckers, D. C. Org. Lett.
2007, 9, 2505.
(37) Clar, E. Chem. Ber. 1942, 75, 1330.
(38) Bailey, W. J.; Liao, C.-W. J. Am. Chem. Soc. 1955, 77, 992.
(39) Marschalk, C. Bull. Soc. Chim. 1943, 10, 511.
(40) Boggiano, B.; Clar, E. J. Chem. Soc. 1957, 2681.
(41) Payne, M. M.; Parkin, S. R.; Anthony, J. E. J. Am. Chem. Soc. 2005,
127, 8028.
(42) Chun, D.; Cheng, Y.; Wudl, F. Angew. Chem., Int. Ed. 2008, 47, 8380.
(43) Kaur, I.; Stein, N. N.; Kopreski, R. P.; Miller, G. P. J. Am. Chem.
Soc. 2009, 131, 3424.
(46) Strating, J.; Zwanenburg, B.; Wagenaar, A.; Udding, A. C. Tetrahedron
Lett. 1969, 125.
(47) Yamada, H.; Yamashita, Y.; Kikuchi, M.; Watanabe, H.; Okujima,
T.; Uno, H.; Ogawa, T.; Ohara, K.; Ono, N. Chem.sEur. J. 2005, 11,
6212.
(44) Mondal, R.; Shah, B. K.; Neckers, D. C. J. Am. Chem. Soc. 2006,
128, 9612.
(45) Bettinger, H. F.; Mondal, R.; Neckers, D. C. Chem. Commun. 2007,
5209.
(48) Dunkin, I. R. Matrix-Isolation Techniques; Oxford University Press:
Oxford, 1998.
9
14282 J. AM. CHEM. SOC. VOL. 131, NO. 40, 2009

 Mondal, Rajib
Mondal, Rajib
 Toenshoff, Christina
Toenshoff, Christina
 Khon, Dmitriy
Khon, Dmitriy
 Neckers, Douglas C.
Neckers, Douglas C.
 Bettinger, Holger F.
Bettinger, Holger F.