RSC Advances
Communication
The performance of rare earth extraction with these dia- of magnitude higher than bLu/Dy of P247 and P2361. The
lkylphosphinic acids was then explored. The extraction of Ln3+ difference of bLu/Dy for various dialkylphosphinic acids in the
from the aqueous phase was studied for Ln (0.01 M), HL (0.2 M), extraction of heavy rare earth is closely related to the steric
(Na, H)Cl (1 M) and pH ¼ 3. Dialkylphosphinic acid exists as hindrance of extractants. The increase in steric hindrance of the
a dimer in aliphatic solvent. Hence, the dialkylphosphinic acid alkyl chain leads to a decrease in separation efficiency in heavy
extractant can be considered to occur as (HL)2 in rare earth rare earth separation. Furthermore, the inuence of branching
extraction. The equilibrium with dialkylphosphinic acid can be of the alkyl group can be found in Table 3 (entries 3 and 7).
represented as follows:
There are little differences exist between methyl and ethyl in
steric hindrance between P247 and P272, but the Kex of P272 in
the extraction equilibrium are of a 1 to 2 orders of magnitude
higher than those of P247 (Table 3, entries 3 and 7). This result
demonstrates that the effect of steric hindrance is greater than
that from branching of the alkyl group. It should be mentioned
that P272 which branched on the terminal carbon has good
separation efficiency, but the terminal branched group also
leads to a decrease in saturation capacity.7,8 In summary, steric
hindrance is important for rare earth metal separation, espe-
cially for heavy rare earths separation. The results revealed that
ethyl and propyl substituents in the b-position of dia-
lkylphosphinic acids are most suitable for rare earth separation.
A computational study was conducted to understand the
Ln3+ + 3(HL)2(o) # [Ln(HL2)3](o) + 3H+
(1)
(a)
(a)
In this equation, subscripts a and o denote species in the
aqueous and organic phases, respectively. The extraction
concentration equilibrium constant, Kex, is given by:
Â
Ã
Â
Ã
Â
Ã
3
Hþ
3
D Hþ
LnðHL2Þ
ðaÞ
3
ðaÞ
ðoÞ
Kex ¼
¼
(2)
Â
Ã
Â
Ã
Â
Ã
3
3
Ln3þ
ðHLÞ
ðHLÞ
ðoÞ
ðoÞ
2
2
ðaÞ
where the distribution ratio D of the lanthanide is given by D ¼
[Ln(HL2)3](o)/[Ln3+
] .
(a)
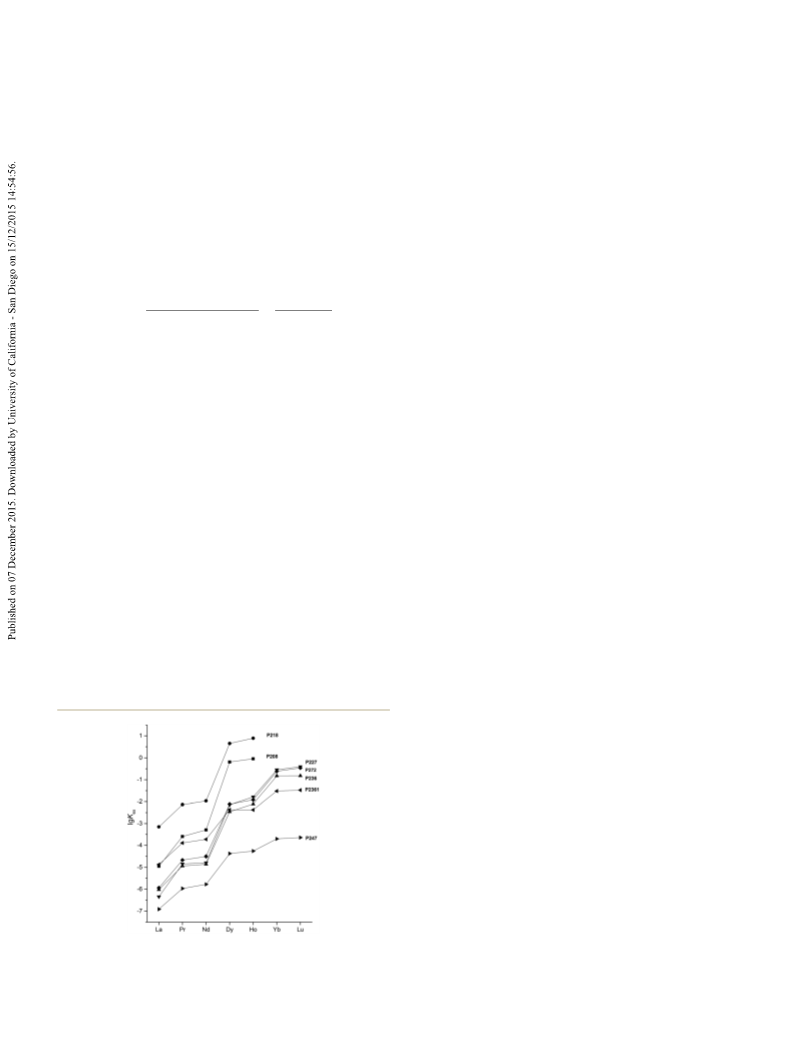
As shown in Fig. 1 and Table 3, in general, Kex for La, Pr, Nd,
Dy, Ho, Yb and Lu extraction decrease as P218 > P208 > Cya-
nex272 > P227 > P236 > P2361 > P247, except for La. Because the
solubility of P208 is poor in n-dodecane, toluene was selected as
the solvent to explore the extraction ability. Kex for rare earth
extraction with dialkylphosphinic acid in n-dodecane is greater
than that in toluene (Table S3†). Therefore, the difference in
extraction ratios could be explained for pKa values (Table 2,
entries 1–7). The pKa increased as 4.96 < 5.42 < 5.58 < 5.61 < 5.71
< 5.80 < 6.05 with increasing in steric hindrance, whereas the Kex
decreased.
´
steric effect on extraction equilibrium. As Garces et al. re-
ported,25 the difference between a stepwise and intrinsic
stability constant would be constant or vary systematically with
changes for multiple coordinate complexes. Based on this
approximation, the following equation has been proposed to
represent the extraction reaction from aqueous to organic
phase:
1/2H2L2(o) + [M(H2O)9]3+ + 3H2O(a)
(a)
+
/ [M(H2O)8L]2+ + H9O4
(3)
(o)
(a)
As shown in Table 3, the separation factor bNd/La is in same
order of magnitude where bLu/Dy differ from an order of
magnitude or more. Similar bNd/La indicate that there is not
much difference in light rare earth separation by various dia-
lkylphosphinic acids. Therefore, the substituent in the b-posi-
tion of dialkylphosphinic acids has few inuence on the light
rare earths separation. A possible reason is that light rare earths
have larger ionic radius. However, the bLu/Dy of P227 is an order
The formations of reactants and products were found from
the work by Clark, Yang and Yuan respectively.20,26,27 The
computational results are listed in Table 4.
Table 4 shows that DGLu is always more negative than DGLa
both in vacuum and in solvent for all extractants. This
computational result agrees well with experimental result that
the extraction of Lu has higher Kex than that of La. For the same
metal, the Kex is inuenced by substituents in the b-position.
For example, the order of DGLu is P208 < P272 z P227 < P236 <
P218 < P2361 < P247 with the substituent in b position changed
by the order H < metyl < ethyl < propyl < isopropyl < t-butyl,
which agrees with the experimental Kex, except for P218. Finally,
the difference between DGLu and DGLa could be used to evaluate
extractant separation activity. For instance, the absolute value of
DGLu ꢀ DGLa for P247 is smaller than that for P227, and its
separation activity is poorer. The order of separation activity for
rare earths from computational results is P236 > P227 > P272 >
P2361 > P247, which agrees with experimental results, except for
P236. Consequently, the effect of steric hindrance in the b-
position can be evaluated conveniently through proper
approximation.
In conclusion, dialkylphosphinic acid was synthesized by
optimized MW-assisted free radical addition. The reaction gives
higher yields and completed in only 2 h comparing 30 h or
more in traditional method. Moreover, this strategy has good
Fig. 1 Extraction equilibrium constants of rare earth metal extraction
by dioctylphosphinic acid.
104260 | RSC Adv., 2015, 5, 104258–104262
This journal is © The Royal Society of Chemistry 2015

 Du, Ruo-Bing
Du, Ruo-Bing
 An, Huaying
An, Huaying
 Zhang, Suhui
Zhang, Suhui
 Yu, Donghai
Yu, Donghai
 Xiao, Ji-Chang
Xiao, Ji-Chang