
Molecules (2018)
Update date:2022-08-17
Topics:
 Levitz, Andrew
Levitz, Andrew
 Marmarchi, Fahad
Marmarchi, Fahad
 Henary, Maged
Henary, Maged
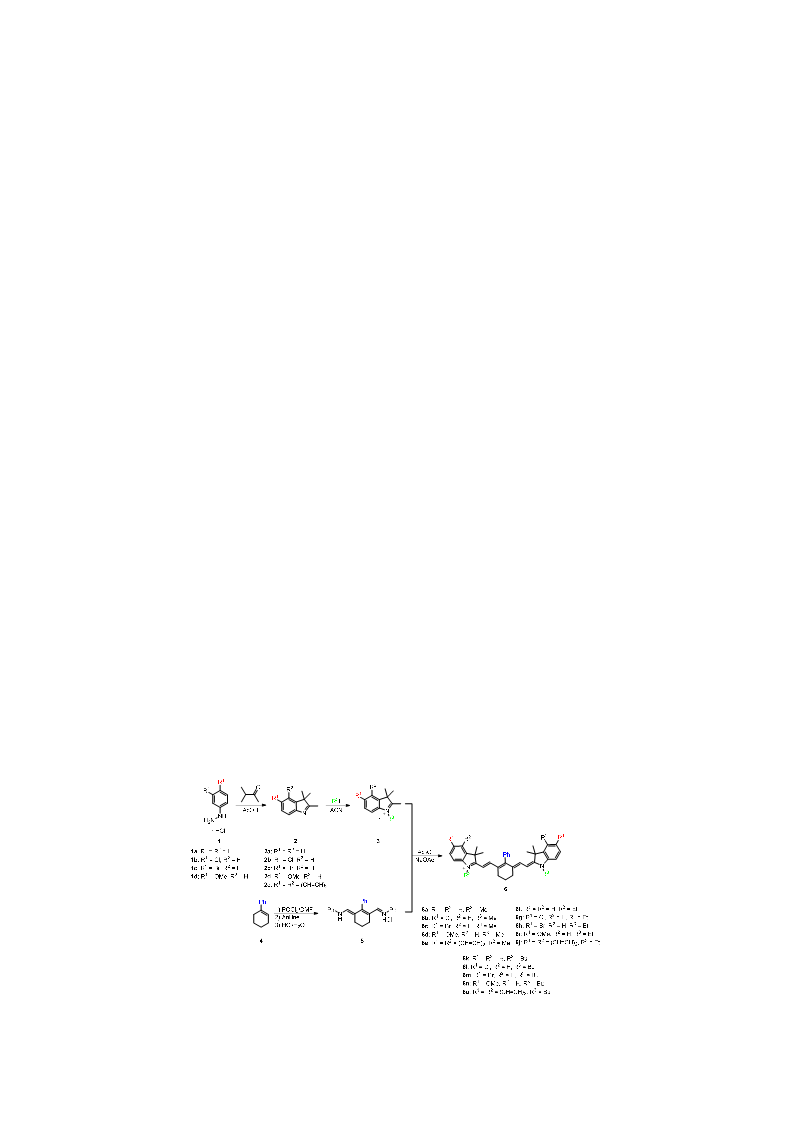
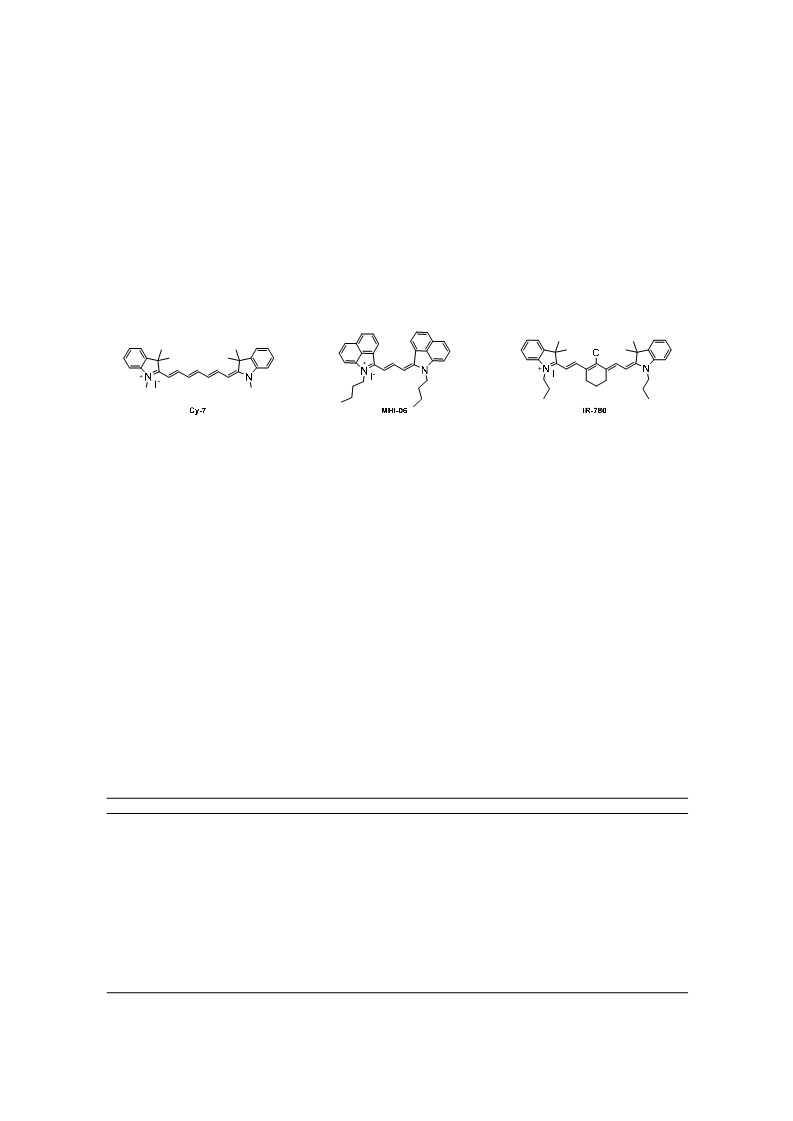
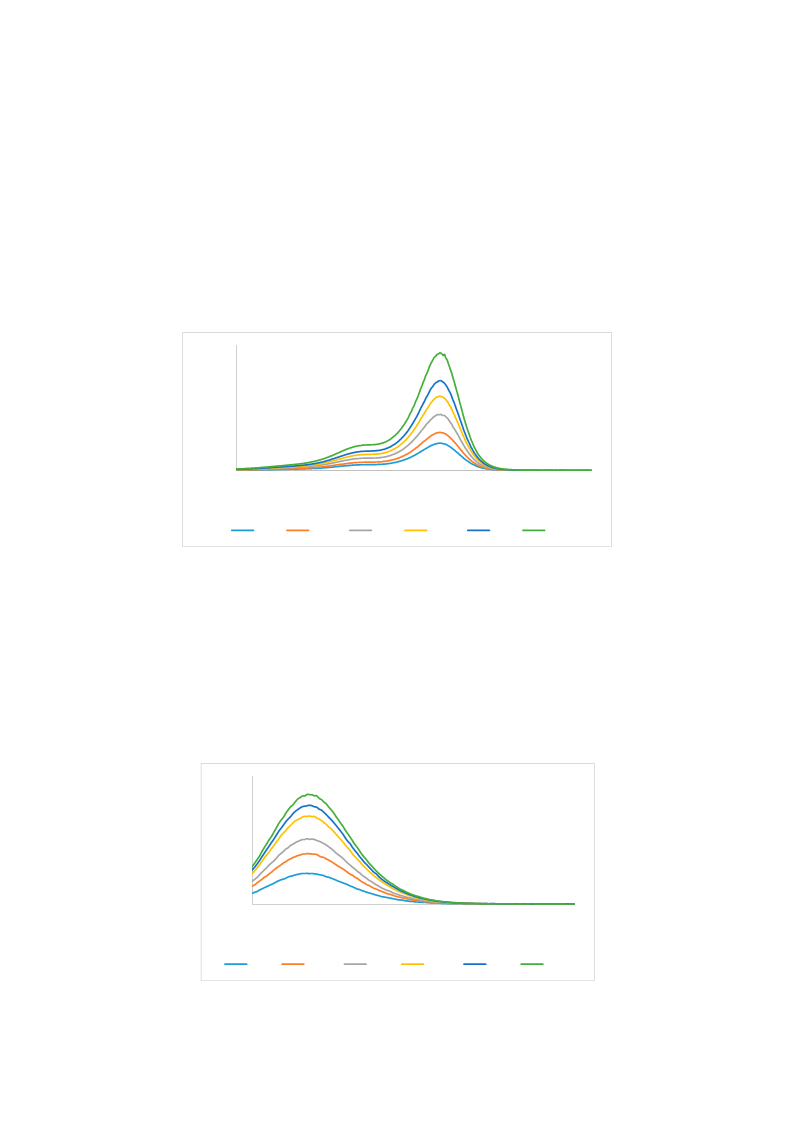
Heptamethine cyanine dyes are a class of near infrared fluorescence (NIRF) probes of great interest in bioanalytical and imaging applications due to their modifiability, allowing them to be tailored for particular applications. Generally, modifications at the meso-position of these dyes are achieved through Suzuki-Miyaura C-C coupling and SRN1 nucleophilic substitution of the chlorine atom at the meso-position of the dye. Herein, a series of 15 meso phenyl-substituted heptamethine cyanines was synthesized utilizing a modified dianil linker. Their optical properties, including molar absorptivity, fluorescence, Stokes shift, and quantum yield were measured. The HSA binding affinities of two representative compounds were measured and compared to that of a series of trimethine cyanines previously synthesized by our lab. The results indicate that the binding of these compounds to HSA is not only dependent on hydrophobicity, but may also be dependent on steric interferences in the binding site and structural dynamics of the NIRF compounds.
View More


Shanghai Sunwise Chemical Co., Ltd
website:http://www.sunwisechem.com
Contact:86-021-33883180
Address:Room 10E, Building G, Westlink Center, No. 2337 Gudai Road, Minhang District, Shanghai, China PC: 201100
SHAANXI TOP PHARM CHEMICAL CO.LTD
Contact:+86-029-85733403
Address:No.108 ,west sector,south er huan,xi'an,china
Shanghai Hanhong Scientific Co.,Ltd.
website:http://www.chemvia.com
Contact:+86-21-64541543,54280654,13918533501
Address:Jiachuan Road 245
Shenzhen Hongyuan Import & Export Co., Ltd.
Contact:0755-26407171
Address:Shenzhen Hongyuan Chemical New Materials Technology Co., Ltd.
Beijing Mesochem Technology Co.,LTD
website:http://www.mesochem.com
Contact:0086-10-57862036
Address:2301, Floor 23, Building 9 Lippo Plaza, Yard 8 Ronghua Middle Road, ETDZ, Beijing, China
Doi:10.1021/ja953548s
(1996)Doi:10.1016/j.jfluchem.2005.07.012
(2005)Doi:10.1002/poc.1460
(2009)Doi:10.1016/j.carres.2016.01.012
(2016)Doi:10.1063/1.442344
(1981)Doi:10.1246/bcsj.43.959
(1970)