10.1002/anie.202105723
Angewandte Chemie International Edition
COMMUNICATION
which can be corroborated to 14NH4 and 15NH4 , respectively
(Figure S37-S39).[27] Ammonia quantification by indophenol
method provides 91% yield (Figures S40 and S41, Table S3).[28]
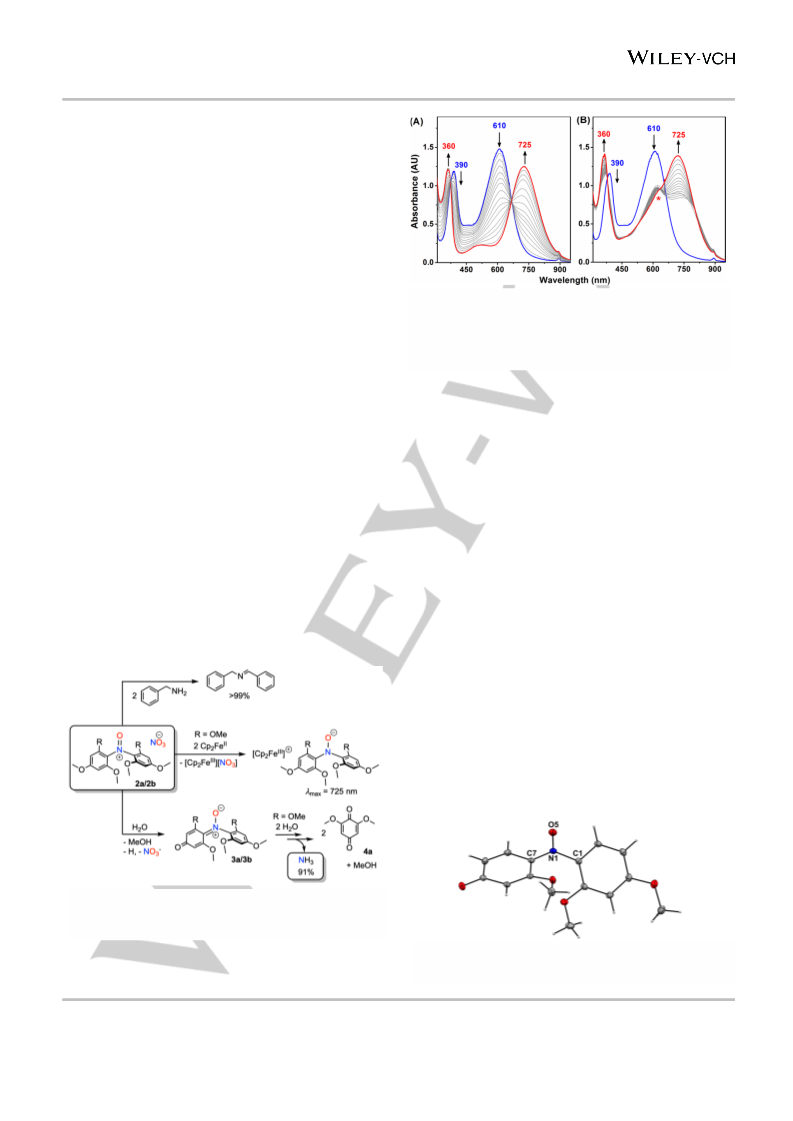
Moreover, the 1H NMR spectrum shows the coformation of
methanol (Figure S37). Surprisingly, the reaction of
[(DMB)2N+=O] (2b) with water under analogous reaction
conditions affords a stable compound 3b (Figure S42). X-ray
diffraction analysis reveals the molecular structure of 3b and
HRMS spectrum confirms the molecular composition (Figures 6,
S43-S46). We postulate that a homologous intermediate 3a may
serve as a transient species for the reaction of 2a with water prior
to the release of ammonia. Indeed, HRMS analysis on 2a in wet
acetonitrile depicts a peak at m/z = 350.1231 (calc. m/z =
350.1234 for MH+), which can be assigned to the intermediate
species 3a (Figure S47).
+
+
[3]
[4]
L. B. Maia, J. J. G. Moura, Chem. Rev. 2014, 114, 5273–5357.
J. O. Lundberg, M. T. Gladwin, A. Ahluwalia, N. Benjamin, N. S. Bryan,
A. Butler, P. Cabrales, A. Fago, M. Feelisch, P. C. Ford, et al., Nat. Chem.
Biol. 2009, 5, 865–869.
[5]
P. G. Wang, M. Xian, X. Tang, X. Wu, Z. Wen, T. Cai, A. J. Janczuk,
Chem. Rev. 2002, 102, 1091–1134.
[6]
[7]
N. Hogg, Annu. Rev. Pharmacol. Toxicol. 2002, 42, 585–600.
L. Batt, K. Christie, R. T. Milne, A. J. Summers, Int. J. Chem. Kinet. 1974,
6, 877–885.
[8]
M. R. Talipov, Q. K. Timerghazin, J. Phys. Chem. B 2013, 117, 1827–
1837.
[9]
A. Dahiya, A. K. Sahoo, T. Alam, B. K. Patel, Chem. Asian J. 2019, 14,
4454–4492.
[10]
[11]
C. L. Ford, Y. J. Park, E. M. Matson, Z. Gordon, A. R. Fout, Science
2016, 354, 741–743.
E. M. Matson, Y. J. Park, A. R. Fout, J. Am. Chem. Soc. 2014, 136,
17398–17401.
[12] A. Mondal, K. P. Reddy, J. A. Bertke, S. Kundu, J. Am. Chem. Soc. 2020,
142, 1726–1730.
In summary, the present work illustrates proton-assisted
–
activations of NOx anions to NO+, which undergoes unusual
[13]
[14]
B. E. Petel, E. M. Matson, Chem. Commun. 2020, 56, 555–558.
S. E. Braley, D. C. Ashley, E. Jakubikova, J. M. Smith, Chem. Commun.
2020, 56, 603–606.
transformation to oxoammonium salts (2a-d) in the presence of
electron-rich aromatics (1a-d). Thus, this report offers
a
[15]
(a) S. Xu, D. C. Ashley, H.-Y. Kwon, G. R. Ware, C.-H. Chen, Y. Losovyj,
X. Gao, E. Jakubikova and J. Smith, Chem. Sci. 2018, 9, 4950–4958. (b)
S. Xu, H.-Y. Kwon, D. C. Ashley, C.-H. Chen, E. Jakubikova and J. M.
Smith, Inorg. Chem. 2019, 58, 9443–9451.
convenient route for the generation oxoammonium species, an
important intermediate in various oxidative transformations.[22]
Moreover, a set of complimentary analyses on the metastable
diaryl oxoammonium salts (2a-d) unfolds their spectroscopic and
structural signatures. Illustrating the oxidative reactivity of
oxoammonium species (2a), this work demonstrates 2e– redox
process in the presence of reducing agents such as benzylamine,
thiols, and ferrocene. Remarkably, the hydrolysis of 2a leads to
the generation of ammonia in near quantitative yields. Thus, this
work connects two extreme oxidation states of N, specifically N+V
[16]
[17]
[18]
[19]
[20]
S. Sankararaman, W. A. Haney, J. K. Kochi, J. Am. Chem. Soc. 1987,
109, 5235–5249.
M. A. Kamboures, J. D. Raff, Y. Miller, L. F. Phillips, B. J. Finlayson-Pitts,
R. B. Gerber, Phys. Chem. Chem. Phys. 2008, 10, 6019–6032.
S. Stefanom, F. Belaj, T. Madl, R. Pietschnig, Eur. J. Inorg. Chem. 2010,
3, 289–297.
M. Shibuya, Y. Osada, Y. Sasano, M. Tomizawa, Y. Iwabuchi, J. Am.
Chem. Soc. 2011, 133, 6497–6500.
–
in NO3 anion and N–III in NH3. Perhaps this may offer new
T. Chattopadhyay, B. S. Anju, S. Gupta, S. Ananya, J. A. Bertke, S.
Kundu, Dalt. Trans. 2019, 48, 7085–7089.
opportunities for designing electrochemical routes targeting NOx
to NH3 conversion.
[21]
[22]
[23]
[24]
See supporting information for the details of DFT calculations.
B. L. Ryland, S. S. Stahl, Angew. Chem. Int. Ed. 2014, 53, 8824–8838.
V. A. Golubev, V. D. Sen, Russ. J. Org. Chem. 2013, 49, 1143–1149.
H. Fujiwara, J. Yamabe, S. Nishimura, Chem. Phys. Lett. 2010, 498, 42–
44.
Experimental Section
[25]
[26]
R. Rathore, S. V. Lindeman, J. K. Kochi, Angew. Chem. Int. Ed. 1998,
37, 1585–1587.
Experimental details are available in the Supporting Information.
HRMS analysis on a freshly prepared crude sample obtained from the
t
reaction of 3,5-dimethoxyphenol and BuONO shows m/z at 336.1073
Acknowledgements
(calc. m/z 336.1078), which can be attributed to the formation of
corresponding oxoammonium salt. The detailed characterization of the
oxoammonium salt, however, was not possible due to the poor yield as
well as its instability.
S.K. gratefully acknowledges the Early Career Research Award
(ECR/2017/003200) from SERB. Authors are also thankful to
IISER-TVM for financial supports.
[27]
[28]
H. S. Kim, J. Choi, J. Kong, H. Kim, S. J. Yoo, H. S. Park, ACS Catal.
2021, 11, 435–445.
A. C. Nielander, J. M. McEnaney, J. A. Schwalbe, J. G. Baker, S. J. Blair,
L. Wang, J. G. Pelton, S. Z. Andersen, K. Enemark-Rasmussen, V. Čolić,
et al., ACS Catal. 2019, 9, 5797–5802.
Keywords: Nitrogen-oxides • Nitrosonium • Oxoammonium •
Ammonia • Electron rich aromatics
[29]
CCDC 2079167 (2a), CCDC 2079168 (2b), and CCDC 2079169 (3b)
contain the supplementary crystallographic data for this paper. These
data can be accessed free of charge from The Cambridge
Crystallographic Data Centre.
[1]
[2]
X. Zhang, B. B. Ward, D. M. Sigman, Chem. Rev. 2020, 120, 5308–5351.
C. Sparacino-Watkins, J. F. Stolz, P. Basu, Chem. Soc. Rev. 2014, 43,
676–706.
This article is protected by copyright. All rights reserved.

 Anju, Balakrishnan S.
Anju, Balakrishnan S.
 Kundu, Subrata
Kundu, Subrata
 Mondal, Aditesh
Mondal, Aditesh
 Sahana, Tuhin
Sahana, Tuhin