4
,6,20–22
products such as limonene.
It should be mentioned that
temperature, the terpene removal corresponds to the b-pinene
removal due to the absence of any thermal reactions over TiO
The TiO photocatalyst—independently of the reaction tem-
◦
◦
at 60 C–80 C, the carbon balance was closed by the different
isomerization products.
2
.
2
At high temperature, thermal catalytic isomerization thus
happens together with the UV-A photocatalytic reaction, with
formation of a-pinene, camphene and limonene isomers. As a
result, the observed removal performances are further expressed
in terms of (total) terpene removal and not of b-pinene removal.
Therefore, a terpene removal efficiency of 100% means that
perature and the testing procedure—displayed firstly a period
with a total terpene removal, i.e. a 100% removal efficiency
with no terpene release, before on-stream deactivation occurred.
Therefore, the initial period during which 100% of terpene
removal was observed (characterized by the Duration of Com-
plete Terpene Removal, DCTP, and expressed in minutes) was
an important parameter to take into account. Fig. 2 shows
the DCTP as a function of the reaction temperature on the
1
00% of the terpene reactants (including the different isomers
formed) have been removed. It was important to note that
independently to the reactor temperature, the inlet total terpene
concentration was 200 ppm. At room temperature, the terpene
removal corresponded to the b-pinene removal due to the
commercial UV100 TiO photocatalyst, obtained with a first
2
dark adsorption period and with a reactor surface coverage
2
of 1.2 mg/cm . The catalyst showed a DCTP of 40 min at
◦
absence of any thermal reactions over TiO
2
.
25 C, whereas the DCTP increased to reach durations longer
◦
than 100–110 min at 60–80 C. This was in agreement with
the usual optimal operating range of photocatalysis, reported
to be 20–80 C with a slight increase in the photoactivity
3
.2. Photocatalytic oxidation/deactivation/regeneration
◦
The photocatalytic behaviour shown by the commercially avail-
able UV100 TiO
8
surface density, on the pinene removal and on the on-stream
deactivation was only investigated at a temperature of 80 C,
with the temperature within this range, due to the very small
2
was studied from room temperature up to
apparent activation energy in this medium range. Since the
23
◦
0 C. The influence of the catalyst surface coverage, i.e. the TiO
2
terpene containing tail-gas effluents released in some industrial
◦
applications are generally at temperatures close to 80 C,
◦
further investigations have been performed at this temperature.
because it appeared that increasing the reaction temperature was
beneficial to the terpene removal efficiency. In addition to that, it
must be noted that from an industrial application point of view,
for which the removal of pinenes and most generally of terpenes
from post-process tail-gas effluents is targeted (such as factories
using wood as raw material), the temperature of such tail-gas
CO was the sole reaction product detected in the outlet
stream, no gaseous intermediate by-products being observed,
thus indicating that partially oxidized by-products remained
2
adsorbed at the TiO surface, as shown later by DRIFT.
2
effluents containing those VOC pollutants is usually close to
◦
8
0 C, as measured by on-site analysis. A reaction/regeneration
cyclic procedure has also been optimized in order to maintain a
total removal efficiency over a long period.
◦
The temperature of the reactor was first increased to 80 C be-
fore two different testing procedures were applied, consisting of :
(
i) a first dark adsorption step followed by the photocatalytic step
when switching on the UV-A lamp and (ii) the direct photocat-
alytic step without waiting for any adsorption/desorption dark
equilibrium. It could be noted that within the whole reaction
temperature range, no photolysis phenomena occurred under
UV-A illumination for both photocatalytic test procedures.
3
.2.1 Using a first dark adsorption step of b-pinene followed
by photooxidation.
Activity. As shown in the previous sub-section, mainly ther-
mal isomerization reactions occurred in the dark, with the
main formation of a-pinene, limonene and camphene. Thus,
proceeding with a first dark adsorption of b-pinene molecules
at 60 C and 80 C (until reaching the adsorption equilibrium),
led to performing the photocatalytic removal of a mixture of
terpenes, instead of that of pure b-pinene, with an inlet flow
containing a mixture of b-pinene, a-pinene, camphene and
limonene terpenes rather than pure b-pinene. It should be noted
that removing a mixture of terpenes was in agreement with the
problematic of some industrial fields, such as those using wood
as raw material, which are concerned with polluted effluents
containing terpene mixtures and not only b-pinene. As a result,
the observed removal performances were expressed in term of
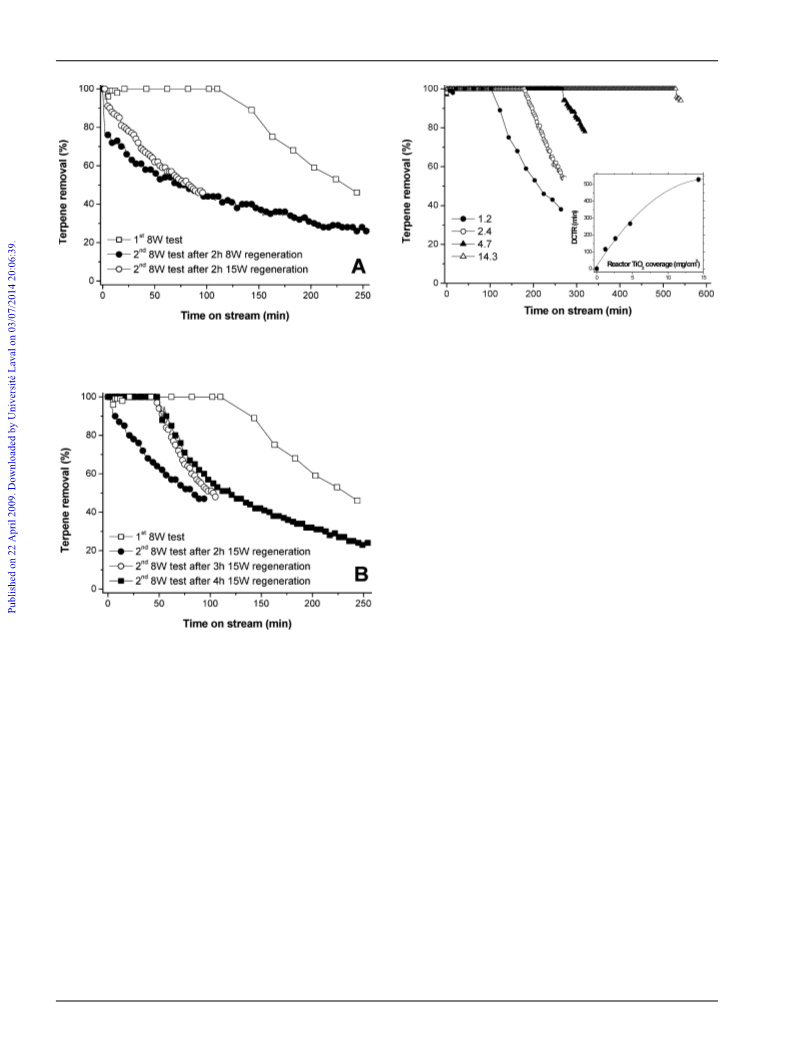
Fig. 2 DCTR (in minutes) as a function of the reaction temperature
2
on UV100 TiO
2
at 1.2 mg/cm surface coverage, with a prior dark
adsorption period, and an 8 W UV-A light.
◦
◦
The influence of the TiO surface density on the on-stream
2
terpene removal is reported in Fig. 3. It could be observed
that increasing the reactor surface coverage from 0.6 to
2
1.2 mg/cm led to extending from 50 min to 100 min, the
initial period during which complete terpene removal was
observed, whereas this duration remained at about 100 min
2
when increasing the TiO
2
loading further up to 14.3 mg/cm .
This led to estimating the VOC conversion before deactivation
occurred at 50 mgterpenes/gTiO2, 48 mgterpenes/gTiO2, 23 mgterpenes/gTiO2
14 mgterpenes/gTiO2 and 4 mgterpenes/gTiO2 for TiO
0.6 mg/cm , 1.2 mg/cm , 2.4 mg/cm , 4.7 mg/cm and
,
2
loadings of
2
2
2
2
(
total) terpene removal and not of b-pinene removal. At room
9
68 | Green Chem., 2009, 11, 966–973
This journal is © The Royal Society of Chemistry 2009

 Salem, Ibtissam
Salem, Ibtissam
 Keller, Nicolas
Keller, Nicolas
 Keller, Valerie
Keller, Valerie