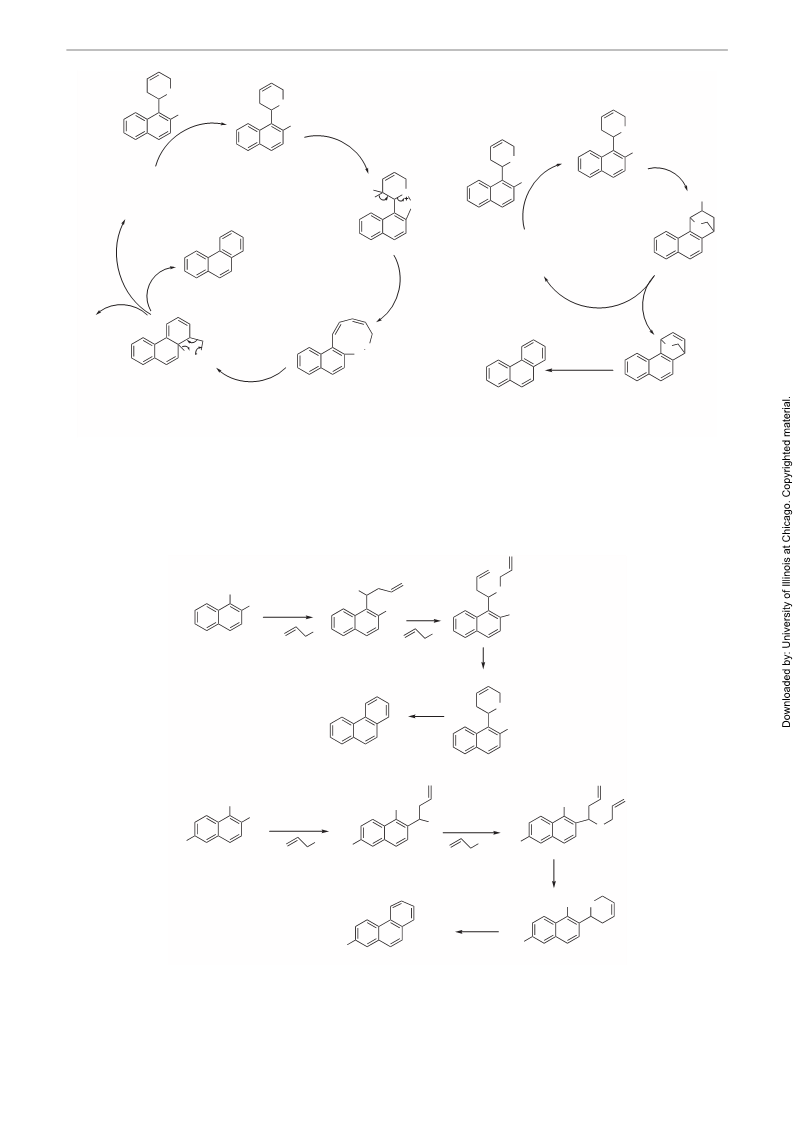
PAPER
Synthesis of Phenanthrene and Alkyl Phenanthrenes
2097
lated product 2a–f (1 mmol) was dissolved in THF (3 mL) and
slowly added into the ice-cold NaH solution. Formation of the anion
was indicated by color change. After the formation of the anion, al-
lyl bromide was added and stirring was continued at r.t.. The reac-
tion mixture was quenched with ice-water and extracted with Et2O
(2 × 20 mL). Solvent was removed under reduced pressure and the
product was purified by column chromatography.
J = 8.6 Hz, 1 H), 7.73 (d, J = 8.6 Hz, 1 H), 8.25 (d, J = 9.2 Hz, 1 H).
13C NMR (50 MHz, CDCl3): d = 19.92, 41.51, 55.65, 72.95, 80.05,
106.35, 112.51, 117.24, 120.00, 123.12, 125.54, 127.10, 127.70,
129.24, 134.87, 135.55, 137.10, 142.33, 158.33.
Anal. Calcd for C19H21BrO2: C, 63.17; H, 5.86. Found: C, 63.31; H,
5.77.
2-(1-Allyloxy-3-methylbut-3-enyl)-1-bromonaphthalene (3f)
Liquid.
1-(1-Allyloxybut-3-enyl)-2-bromonaphthalene (3a)
Liquid.
1H NMR (200 MHz, CDCl3): d = 1.89 (s, 3 H), 2.47 (d, J = 7 Hz,
2 H), 3.80–3.85 (m, 1 H), 3.89–3.93 (m, 1 H), 4.82 (m, 2 H), 5.15–
5.30 (m, 3 H), 5.84–6.30 (m, 1 H), 7.52–7.67 (m, 3 H), 7.82 (m,
2 H), 8.37 (dd, J = 8, 8.2 Hz, 1 H).
1H NMR (200 MHz, CDCl3): d = 2.65–2.75 (m, 1 H), 2.91–3.02 (m,
1 H), 3.82–3.87 (m, 2 H), 5.00–5.27 (m, 4 H), 5.52 (dd, J = 6.2,
8.4 Hz, 1 H), 5.80–5.98 (m, 2 H), 7.47–7.59 (m, 4 H), 7.81 (m,
1 H), 8.88 (m, 1 H).
13C NMR (50 MHz, CDCl3): d = 22.86, 45.12, 70.03, 79.23, 112.90,
117.15, 122.91, 124.72, 126.51, 127.36, 127.41, 128.17 (2C),
132.19, 134.64, 139.86, 142.36.
13C NMR (50 MHz, CDCl3): d = 40.35, 69.91, 82.19, 117.05,
117.14, 123.03, 125.72, 126.13, 126.42, 128.75, 129.72, 130.25,
132.46, 133.63, 134.70, 134.79, 135.05.
Anal. Calcd for C18H19BrO: C, 65.27; H, 5.78. Found: C, 65.17; H,
5.88.
Anal. Calcd for C17H17BrO: C, 64.37; H, 5.40. Found: C, 64.49; H,
5.28.
1-(1-Allyloxy-3-methylbut-3-enyl)-2-bromonaphthalene (3g)
Liquid.
2-(1-Allyloxybut-3-enyl)-1-bromo-6-methoxynaphthalene (3b)
Liquid.
1H NMR (200 MHz, CDCl3): d = 1.91 (s, 3 H), 2.58 (m, 1 H), 2.99
(m, 1 H), 3.85–3.99 (m, 2 H), 4.82 (s, 1 H), 4.88 (s, 1 H), 5.15–5.31
(m, 1 H), 5.69 (dd, J = 4.8, 9.2 Hz, 1 H), 5.82 (m, 1 H), 7.53–7.60
(m, 2 H), 7.65 (s, 2 H), 7.83–7.83 (m, 1 H), 8.99 (m, 1 H).
1H NMR (200 MHz, CDCl3): d = 2.51–2.58 (m, 2 H), 3.78–3.93 (m,
2 H), 3.94 (s, 3 H), 5.01–5.30 (m, 5 H), 5.84 (m, 2 H), 7.13 (m,
1 H), 7.23 (dd, J = 2.6, 9.2 Hz, 1 H), 7.57 (m, 1 H), 7.72 (m, 1 H),
8.23 (d, J = 9.2 Hz, 1 H).
13C NMR (50 MHz, CDCl3): d = 22.80, 43.96, 69.88, 81.08, 112.96,
117.20, 122.83, 125.79, 125.92, 126.09, 128.69, 129.50, 130.18,
132.47, 133.62, 134.72, 135.58, 142.54.
13C NMR (50 MHz, CDCl3): d = 41.23, 55.47, 69.88, 80.03, 106.23,
116.95, 117.00, 119.80, 122.95, 125.36, 126.90, 127.54, 129.07,
134.56, 134.75, 135.39, 136.89, 158.19.
Anal. Calcd for C18H19BrO: C, 65.27; H, 5.78. Found: C, 65.43; H,
5.70.
Anal. Calcd for C18H19BrO2: C, 62.26; H, 5.52. Found: C, 62.20; H,
5.65.
2-(1-Allyloxy-3-methylbut-3-enyl)-1-bromo-6-methoxynaph-
thalene (3h)
Liquid.
1-Bromo-2-[1-(2-methylallyloxy)but-3-enyl]naphthalene (3c)
Liquid.
1H NMR (200 MHz, CDCl3): d = 1.83 (s, 3 H), 2.57 (t, J = 6.4 Hz,
2 H), 3.73 (d, J = 12.6 Hz, 1 H), 3.84 (d, J = 12.6 Hz, 1 H), 4.91–
5.18 (m, 5 H), 5.85–6.02 (m, 1 H), 7.49–7.65 (m, 3 H), 7.84 (d,
J = 8 Hz, 2 H), 8.34 (d, J = 8 Hz, 1 H).
13C NMR (50 MHz, CDCl3): d = 19.76, 41.23, 72.89, 79.98, 112.44,
117.18, 123.02, 124.81, 126.54, 127.47, 128.09, 128.17, 132.19,
134.20, 134.61, 139.40, 142.09.
1H NMR (200 MHz, CDCl3): d = 1.86 (s, 3 H), 2.46 (m, 2 H), 3.73–
3.91 (m, 2 H), 3.94 (s, 3 H), 4.79 (m, 1 H), 5.13–5.29 (m, 3 H),
5.81–5.95 (m, 1 H), 7.12 (d, J = 2.4 Hz, 1 H), 7.25 (dd, J = 2.4,
9 Hz, 1 H), 7.58 (dd, J = 2.2, 8.4 Hz, 1 H), 7.72 (d, J = 8.4 Hz, 1 H),
8.24 (d, J = 9 Hz, 1 H).
13C NMR (50 MHz, CDCl3): d = 23.01, 45.36, 55.56, 70.04, 79.24,
106.30, 112.98, 117.19, 119.97, 123.00, 125.39, 127.15, 127.65,
129.15, 134.86, 135.50, 137.49, 142.54, 158.28.
Anal. Calcd for C18H19BrO: C, 65.27; H, 5.78. Found: C, 65.36; H,
5.65.
Anal. Calcd for C19H21BrO2: C, 63.17; H, 5.86. Found: C, 63.30; H,
5.71.
2-Bromo-1-[1-(2-methylallyloxy)but-3-enyl]naphthalene (3d)
Liquid.
1-Bromo-2-[3-methyl-1-(2-methylallyloxy)but-3-enyl]naphtha-
lene (3i)
Liquid.
1H NMR (200 MHz, CDCl3): d = 1.69 (s, 3 H), 2.61–2.74 (m, 1 H),
2.93–3.07 (m, 1 H), 3.74 (m, 2 H), 4.82 (s, 1 H), 4.87 (s, 1 H), 5.06
(m, 2 H), 5.5 (m, 1 H), 5.83–6.04 (m, 1 H), 7.40–7.55 (m, 4 H), 7.80
(m, 1 H), 8.88 (d, J = 8.2 Hz, 1 H).
13C NMR (50 MHz, CDCl3): d = 19.81, 29.78, 40.36, 72.85, 82.12,
112.54, 117.04, 125.79, 125.94, 126.11, 126.38, 128.71, 129.67,
125.94, 126.11, 126.38, 128.71, 129.67, 130.23, 132.40, 133.61,
134.89, 135.09, 142.15.
1H NMR (200 MHz, CDCl3): d = 1.75 (s, 3 H), 1.90 (s, 3 H), 2.49
(m, 2 H), 3.68 (d, J = 12.4 Hz, 1 H), 3.83 (d, J = 12.4 Hz, 1 H),
4.83–4.99 (m, 4 H), 5.25 (dd, J = 5.4, 7.6 Hz, 1 H), 7.49–7.68 (m,
3 H), 7.84 (dd, J = 2.4, 8.6 Hz, 2 H), 8.36 (dd, J = 8.2, 0.8 Hz, 1 H).
13C NMR (100 MHz, CDCl3): d = 19.88, 23.07, 45.41, 73.22, 79.57,
112.50, 113.21, 123.07, 124.93, 126.66, 127.55 (2C), 128.32 (2C),
132.41, 134.39, 140.14, 142.36, 142.58.
Anal. Calcd for C18H19BrO: C, 65.27; H, 5.78. Found: C, 65.40; H,
5.60.
Anal. Calcd for C19H21BrO: C, 66.09; H, 6.13. Found: C, 65.95; H,
6.25.
1-Bromo-6-methoxy-2-[1-(2-methylallyloxy)but-3-enyl]naph-
thalene (3e)
Liquid.
2-Bromo-1-[3-methyl-1-(2-methylallyloxy)but-3-enyl]naphtha-
lene (3j)
Liquid.
1H NMR (200 MHz, CDCl3): d = 1.75 (s, 3 H), 2.52–2.59 (m, 2 H),
3.64–3.86 (m, 2 H), 3.93 (s, 3 H), 4.9–5.14 (m, 5 H), 5.86–6.01 (m,
1 H), 7.12 (d, J = 2.4 Hz, 1 H), 7.22–7.28 (m, 1 H), 7.59 (d,
Synthesis 2010, No. 12, 2092–2100 © Thieme Stuttgart · New York

 Jana, Rathin
Jana, Rathin
 Biswas, Anup
Biswas, Anup
 Samanta, Shubhankar
Samanta, Shubhankar
 Ray, Jayanta K.
Ray, Jayanta K.