E.A. Zaburdaeva et al. / Journal of Organometallic Chemistry 692 (2007) 1265–1268
1267
Table 1
Oxidation of sulfides R
2
S by the system (t-BuO)
3
Al– t-BuOOH(1:1:2), 20 °C, benzene
a
N
R
Products/mole of sulfide
Volatile fraction
After hydrolysis of nonvolatile residue
t-BuOOH
t-BuOH
R
2
S
t-BuOOH
t-BuOH
R
2
S
2 2
R SO
1
2
3
4
5
6
7
Me
Et
Pr
i-Pr
Bu
Ph
0.04
0.06
0.06
0.08
0.05
–
2.26
2.30
1.73
2.16
2.40
2.24
2.29
0.04
Traces
Traces
–
–
–
–
0.03
–
1.62
–
–
–
0.94
0.79
0.96
0.95
0.96
0.80
0.96
b
b
0.15
2.22
Traces
–
0.03
0.12
–
b
b
–
–
0.14
–
0.22
1.92
2.46
b
PhCH
2
0.05
–
a
Averaged data are presented.
Not determined.
b
eluent used was a mixture benzene–hexane (9:1), developer
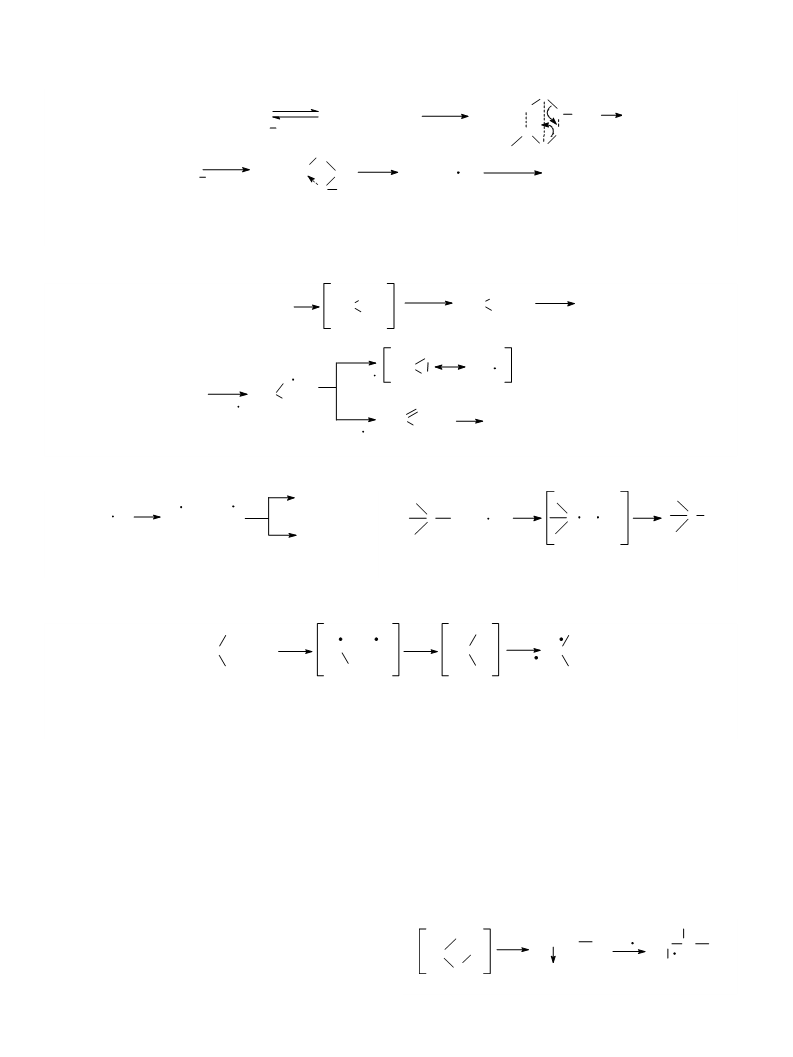
O
O
R2S + [(t-BuO)nM O2]
R2S
acetic acid and than 40% potassium iodide. The R of the
R2SO2
f
-
(t-BuO)nM
Ph;
analyzing solution and known peroxide is identical. t-
BuOOH in such conditions was not determined.
Oxidation and analyses of other hydrocarbons were car-
ried out analogously.
R = Me, Et, Pr, i-Pr, Bu, Ph, CH
M = Al, n = 3; Ti, n = 4
2
Scheme 7.
Reaction of aniline with the tri-tert-butoxyaluminium–
tert-butylhydroperoxide (1:1:2) system. To t-BuOOH
kinetically independent molecule but oxidizes the substrate
to sulfones directly in the coordination sphere of the
metal with the intermediate formation of thiadioxirane
(0.72 g) was added a mixture of (t-BuO) Al (0.98 g) and
3
PhNH (0.37 g) in benzene (20 mL) and left for 1 day at
2
ꢀ20 °C. Then the solvent and the volatile components
(
Scheme 7).
were condensed off a trap cooled with liquid nitrogen.
It should be noted that the action of the Ph Bi–t-
The volatile fraction contained PhNO
BuOH (0.91 g). The residue, dry yellow powder, was
hydrolyzed with 10% H SO , extracted with diethyl ether,
and dried with Na SO . GLC analysis of the ether hydro-
lyzate detected PhNO2 (0.04 g), t-BuOH (0.33 g), t-
BuOOH (0.09 g), PhN@NPh (0.01 g) and PhN(O)@NPh
(0.29 g).
EPR spectra were detected on a Bruker ER200D-SRC
spectrometer equipped with an ER 4105 DR double reso-
nator (working frequency ꢀ9.5 GHz) and an ER 4111
VT temperature-controlling unit. Diphenylpicrylhydrazyl
(DPPH) was used as a standard in the determination of
the g-factor.
2
(0.04 g) and t-
3
BuOOH system on the sulfides differs from that described
above. The main direction is not the oxidation of the S
atom but the cleavage of the S–C bond of the substrate.
For example in the case of dibenzylsulfide ꢀ50% benzalde-
hyde has been found. At the same time interaction of diiso-
propylsulfide with (5)–(2) system leads to acetone (26%).
We offer a radical mechanism for this transformation.
2
4
2
4
2
. Experimental
Reaction of 1,1-diphenylethane with tri-tert-butoxyalu-
minium– tert-butylhydroperoxide (1:1:2) system. The mix-
ture of (t-BuO) Al (2.38 g), t-BuOOH (1.74 g) and
The reactions with other amines were carried out
analogously.
3
Ph CHCH (1.77 g) in 25 mL of benzene was left for 4 days
2
3
at room temperature. Then the solvent and volatile compo-
nents from the red-brown solution were condensed off a
trap cooled with liquid nitrogen. The volatile fraction con-
Oxidation of dipropyl sulfide by tri-tert-butoxyalumin-
ium–tert-butylhydroperoxide (1:1:2) system. t-BuOOH
(1.44 g) was added to a solution of (t-BuO)
and Pr S (0.94 g) in benzene (20 mL). After 1–2 min, the
reaction mixture was self-heated. The formation of Pr SO ,
2
Al (1.96 g)
3
tained t-BuOH (1.85 g) and PhC(O)CH (0.02 g). The res-
2
3
idue, red oil, was hydrolyzed with 10% H SO , extracted
2
2
4
with diethyl ether, and dried with Na SO . In the ether
whose amount obtained was 0.78 g (65%) in 4.5 h, was
monitored by GLC. Then the solvent and volatile compo-
nents of the reaction mixture were condensed in a trap
cooled with liquid nitrogen. The volatile fraction contained
t-BuOH (0.84 g) and t-BuOOH (0.02 g). Sulphuric acid
(10%) was added to a colorless powder-like residue, the
2
4
solution of t-BuOH (1.23 g), PhC(O)CH (0.07 g), PhOH
3
(
0.07 g), Ph CO (0.03 g), Ph C(OH)CH
(0.27 g),
2
2
3
Ph C@CH (0.10 g), Ph C(OOH)CH (0.27 g), PhCOOH
2
2
2
3
(
0.04 g) and non-reacted Ph CHCH (0.85 g) were
2 3
detected. In the aqua-acid part 0.27 g of aluminum were
found.
mixture was extracted with Et
with Na SO and analyzed by GLC. Pr
O, and the extract was dried
2
Ph C(OOH)CH was also identified by paper chroma-
2
4
2
SO (1.12 g,
2
2
3
tography. The ether from the part of ether solution was
condensed and the residue was dissolved in benzene. The
94%), t-BuOH (1.53 g) and t-BuOOH (0.11 g) were
found.

 Zaburdaeva, Elena A.
Zaburdaeva, Elena A.
 Dodonov, Viktor A.
Dodonov, Viktor A.
 Stepovik, Larisa P.
Stepovik, Larisa P.