376
S. Kang et al. / Tetrahedron Letters 54 (2013) 373–376
1,10-binaphthyl-2,20-dicarboxylic acid 3f is known as an effective
Brønsted acid organocatalyst. In case of acenaphthylene-1,2-dione
1g and aceanthrylene-1,2-dione 1h, the corresponding diacids 3g
and 3h were obtained in high yields (92% and 83%) within 10
and 15 min, respectively (Table 2, entries 7–8).
16. Hui, Y.-Z.; Gai, Y.-H.; Chen, X.-E. Acta Chim. Sin. 1988, 6, 91–94.
17. Yang, D. T. C.; Evans, T. T.; Yamazaki, F.; Narayanna, C.; Kabalka, G. W. Synth.
Commun. 1993, 23, 1183–1187.
18. Yan, J.; Travis, B. R.; Borhan, B. J. Org. Chem. 2004, 69, 9299–9302.
19. (a) Joo, C.; Kang, S.; Kim, S. M.; Han, H.; Yang, J. W. Tetrahedron Lett. 2010, 51,
6006–6007; (b) Kang, S.; Joo, C.; Kim, S. M.; Han, H.; Yang, J. W. Tetrahedron
Lett. 2011, 52, 502–504.
20. Cyclic 1,2-diketones 1 and secondary alcohols 2 were purchased or prepared
according to the literature procedures: Aldrich, 1a, 1e, 1g, 1h, 2a, and 2c; Fluka,
2b; TCI, 2d and 2f; Ref. 25, 1b–d and 1f; Ref. 26, 2e.
While studying the diacid formation upon oxidative cleavage of
cyclic 1,2-diketones, we observed that a
-hydroxy acids 30a, 30c, and
30e were also produced in 5%, 24%, and 22% yields, respectively,
when the reactions of substrates 1a, 1c, and 1e were performed.
21. General procedure for the oxidative cleavage of cyclic 1,2-diketones
1 to
dicarboxylic acids 3: To a solution of cyclic 1,2-diketones 1 (0.5 mmol) and
4,40-dichlorobenzhydrol (1.0 mmol) in distilled THF (10 mL) was added NaH
(60% in mineral oil, 1.5 mmol) at 0 °C. While stirring, the dark-blue solution
was allowed to warm to room temperature. After stirring under O2 (1 atm),
until TLC analysis indicated a complete consumption of the starting material,
the reaction mixture was quenched by adding 0.1 N HCl to a final pH of 1–2
and extracted with CH2Cl2. The combined organic layers were dried over
MgSO4 and concentrated in vacuo. The residue was purified by flash
chromatography (preequilibrated with EtOAc/n-hexane/TFA = 1:20:0.1) to
give 3. All products 3 and 4 were characterized by 1H and 13C NMR spectra,
some of which were compared with the literature data: Aldrich, 3a and 4b; Ref.
27, 3b and 3f; Ref. 28, 4c–f.
However,
a-hydroxy acid was rarely produced when other sub-
strates were employed. Formation of diacid or
a-hydroxy acid
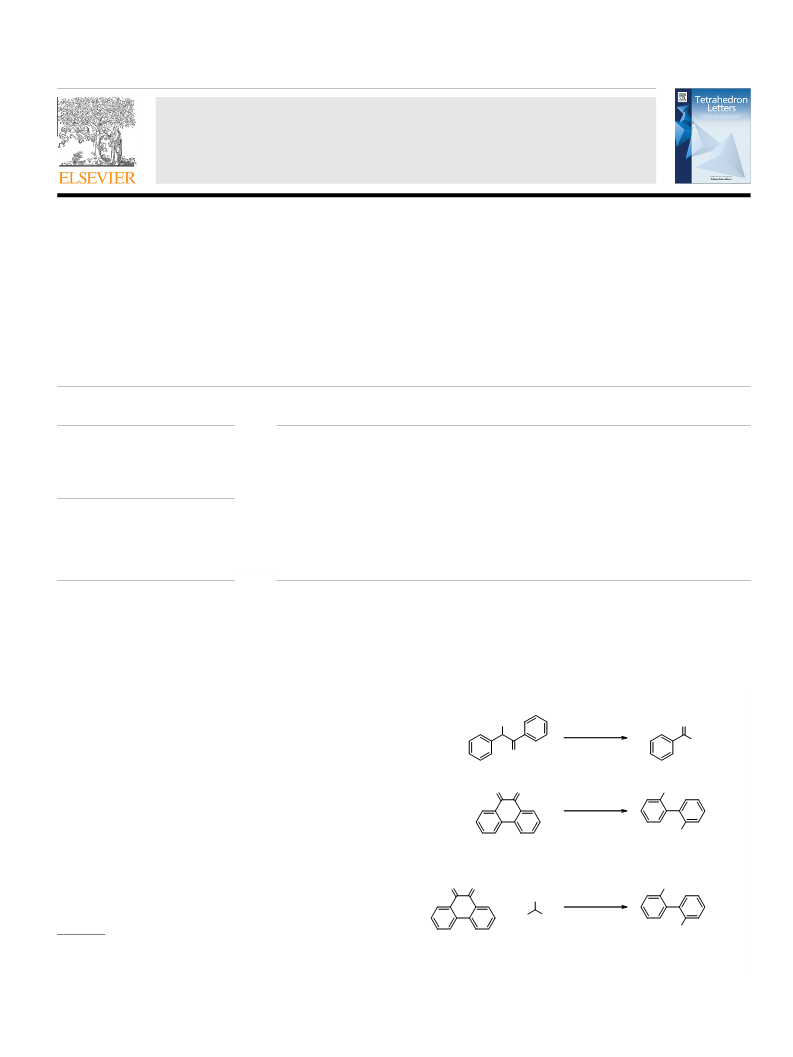
can be explained by the mechanism shown in Figure 1.22 Diacid
3 is formed via the Baeyer–Villiger oxidation mechanism, whereas
a
-hydroxy acid 30 is formed via the benzilic acid rearrangement
mechanism.23 Nucleophilic attack of hydroperoxide 5 and hydrox-
ide 50 on cyclic 1,2-diketone 1 leads to the formation of diacid 3
and a
-hydroxy acid 30, respectively. Hydroperoxide 5 and hydrox-
ide 50 involved are released from the intermediates 20 and 6,
respectively. Regardless of cyclic 1,2-diketone 1 employed, diacid
3 was almost exclusively formed under our reaction conditions.
These results indicate that the nucleophilic attack of hydroperox-
ide 5 on cyclic 1,2-diketone 1 proceeds faster and more efficiently.
This may be due to the higher reactivity of hydroperoxide gener-
ated in situ and in organic solvent.24
Characterization of compound 3c: TLC (MeOH/CH2Cl2 = 1:5) Rf = 0.21; 1H NMR
(400 MHz, DMSO-d6)
d 12.77 (brs, 2H), 7.84 (d, J = 8.4 Hz, 2H), 7.68 (d,
J = 8.4 Hz, 2H), 7.43 (s, 2H); 13C NMR (100 MHz, DMSO-d6) d 166.75, 143.71,
132.52, 131.57, 130.40, 129.30, 124.85; HRMS (EI+) for C14H8Br81BrO4 (M+),
calcd 399.8768, found 399.8764.
Characterization of compound 3d: TLC (MeOH/CH2Cl2 = 1:5) Rf = 0.16; 1H NMR
(600 MHz, DMSO-d6) d 12.47 (brs, 2H), 7.33 (s, 2H), 7.09 (dd, J = 8.4, 2.4 Hz, 2H),
7.05 (d, J = 8.4 Hz, 2H), 3.81 (s, 6H); 13C NMR (150 MHz, DMSO-d6) d 167.96,
157.70, 134.75, 131.93 (2C), 116.57, 114.21, 55.29; HRMS (EI+) for C16H14O6
(M+), calcd 302.0790, found 302.0789.
In summary, we have developed a facile method for the oxida-
tive cleavage of cyclic 1,2-diketones to dicarboxylic acids with
hydroperoxide generated in situ. In situ generation of hydroperox-
ide was effected by the oxidation of 4,40-dichlorobenzhydrol to
4,40-dichlorobenzophenone using NaH under O2 atmosphere. Using
hydroperoxide thus generated in organic solvent enabled such oxi-
dative cleavage to proceed fast and efficiently.
Characterization of compound 3e: TLC (MeOH/CH2Cl2 = 1:6) Rf = 0.28; 1H NMR
(600 MHz, DMSO-d6) d 12.86 (brs, 2H), 8.01 (d, J = 8.4 Hz, 1H), 7.96 (d, J = 8.4
Hz, 1H), 7.94 (d, J = 8.4 Hz, 1H), 7.92 (d, J = 6.0 Hz, 1H), 7.63–7.57 (m, 3H), 7.51
(td, J = 7.8, 1.2 Hz, 1H), 7.34 (d, J = 8.4 Hz, 1H), 7.31 (d, J = 7.2 Hz, 1H); 13C NMR
(100 MHz, DMSO-d6) d 169.55, 167.65, 141.13, 137.30, 131.79, 131.13, 131.06,
130.85, 130.47, 130.04, 128.82, 128.07, 127.95, 127.83, 127.74, 127.09, 126.12,
125.03; HRMS (EI+) for C18H12O4 (M+), calcd 292.0735, found 292.0732.
Characterization of compound 3g: TLC (MeOH/CH2Cl2 = 1:7) Rf = 0.31; 1H NMR
(400 MHz, DMSO-d6) d 8.53 (t, J = 7.8 Hz, 4H), 7.91 (t, J = 7.6 Hz, 2H); 13C NMR
(100 MHz, DMSO-d6) d 160.79, 135.47, 132.56, 131.43, 129.78, 127.64, 119.07.
Characterization of compound 3h: TLC (EtOAc/n-hexane = 1:3) Rf = 0.33; 1H NMR
(600 MHz, DMSO-d6) d 9.58 (d, J = 9.0 Hz, 1H), 9.39 (s, 1H), 8.73 (d, J = 9.0 Hz,
1H), 8.70 (d, J = 6.6 Hz, 1H), 8.40 (d, J = 8.4 Hz, 1H), 8.00 (t, J = 7.5 Hz, 1H), 7.93
(t, J = 7.8 Hz, 1H), 7.80 (t, J = 7.5 Hz, 1H); 13C NMR (150 MHz, DMSO-d6) d
160.93, 160.35, 138.50, 136.81, 135.27, 132.90, 132.31, 131.90, 130.68, 130.55,
128.64, 126.94, 126.17, 125.11, 118.89, 111.44.
Acknowledgments
This work was supported by the NRF Grant (No. 2010-0022070)
to H.H. and the NRF WCU program (R31-2008-10029) and the KE-
TEP Human Resources Development Program (20124010203270)
to J.W.Y.
22. Proposed mechanism for the conversion of 2 to 4, see: (a) Lewis, G. E. J. Org.
Chem. 1965, 30, 2433–2436; (b) Wang, X.; Wang, D. Z. Tetrahedron 2011, 67,
3406–3411.
References and notes
23. Kürti, L.; Czakó, B. Strategic Applications of Named Reactions in Organic Synthesis;
Elsevier: New York, 2005. pp. 28–29 and 52–53.
1. Biradar, A. V.; Sathe, B. R.; Umbarkar, S. B.; Dongare, M. K. J. Mol. Catal. A: Chem.
2008, 285, 111–119.
2. Sato, K.; Aoki, M.; Noyori, R. Science 1998, 281, 1646–1647.
24. Following the reviewer0s suggestion, we performed the oxidative cleavage of
cyclic 1,2-diketone 1a (0.5 mmol) using H2O2 (30%, 102 lL, 1.0 mmol) and
NaOH (0.4 N, 2.5 mL, 1.0 mmol) in THF (7.5 mL) at room temperature, which
gave the diacid 3a in 91% yield after 1 h. Note that our new method also gave
the comparable yield within a shorter reaction time (see Table 2, entry 1).
25. For the synthesis and characterization of cyclic 1,2-diketone 1b, see: (a) Park,
T.-H.; Koh, K.; Wong-Foy, A. G.; Matzger, A. J. Cryst. Growth Des. 2011, 11, 2059–
2063; Compound 1c: (b) Kim, H.-J.; Lee, E.; Park, H.-S.; Lee, M. J. Am. Chem. Soc.
2007, 129, 10994–10995; (c) Zhang, J.; Wang, X.; Su, Q.; Zhi, L.; Thomas, A.;
Feng, X.; Su, D. S.; Schlögl, R.; Müllen, K. J. Am. Chem. Soc. 2009, 131, 11296–
11297; Compound 1d: (d) Trosien, S.; Waldvogel, S. R. Org. Lett. 2012, 14,
2976–2979; Compound 1f: (e) Page, P. C. B.; Buckley, B. R.; Farah, M. M.;
Blacker, A. J. Eur. J. Org. Chem. 2009, 3413–3426; (f) Shen, H.-C.; Tang, J.-M.;
Chang, H.-K.; Yang, C.-W.; Liu, R.-S. J. Org. Chem. 2005, 70, 10113–10116; (g)
Furutani, T.; Hatsuda, M.; Imashiro, R.; Seki, M. Tetrahedron: Asymmetry 1999,
10, 4763–4768; (h) Brunner, H.; Goldbrunner, J. Chem. Ber. 1989, 122, 2005–
2009; (i) Modler-Spreitzer, A.; Fritsch, R.; Mannschreck, A. Collect. Czech. Chem.
Commun. 2000, 65, 555–560.
26. For the synthesis and characterization of secondary alcohol 2e, see: Pavia, M.
R.; Lobbestael, S. J.; Nugiel, D.; Mayhugh, D. R.; Gregor, V. E.; Taylor, C. P.;
Schwarz, R. D.; Brahce, L.; Vartanian, M. G. J. Med. Chem. 1992, 35, 4238–4248.
27. For the characterization of dicarboxylic acid 3b, see: (a) Vonlanthen, D.;
Rotzler, J.; Neuburger, M.; Mayor, M. Eur. J. Org. Chem. 2010, 120–133;
Compound 3f: (b) Schlosser, M.; Bailly, F. J. Am. Chem. Soc. 2006, 128, 16042–
16043.
3. de Boer, J. W.; Brinksma, J.; Browne, W. R.; Meetsma, A.; Alsters, P. L.; Hage, R.;
Feringa, B. L. J. Am. Chem. Soc. 2005, 127, 7990–7991.
4. (a) Herrmann, W. A.; Fischer, R. W.; Marz, D. W. Angew. Chem., Int. Ed. Engl.
1991, 30, 1638–1641; (b) Herrmann, W. A.; Fischer, R. W.; Scherer, W.; Rauch,
M. U. Angew. Chem., Int. Ed. Engl. 1993, 32, 1157–1160; (c) Rudolph, J.; Reddy, K.
L.; Chiang, J. P.; Sharpless, K. B. J. Am. Chem. Soc. 1997, 119, 6189–6190; (d)
Yudin, A. K.; Sharpless, K. B. J. Am. Chem. Soc. 1997, 119, 11536–11537.
5. Fujita, M.; Costas, M.; Que, L., Jr. J. Am. Chem. Soc. 2003, 125, 9912–9913.
6. (a) Shing, T. K. M.; Tam, E. K. W.; Tai, V. W.-F.; Chung, I. H. F.; Jiang, Q. Chem.-
Eur. J. 1996, 2, 50–57; (b) Neisius, N. M.; Plietker, B. J. Org. Chem. 2008, 73,
3218–3227; (c) Griffith, W. P.; Shoair, A. G.; Suriaatmaja, M. Synth. Commun.
2000, 30, 3091–3095; (d) Kogan, V.; Quintal, M. M.; Neumann, R. Org. Lett.
2005, 7, 5039–5042; (e) Plietker, B. J. Org. Chem. 2003, 68, 7123–7125.
7. Travis, B. R.; Narayan, R. S.; Borhan, B. J. Am. Chem. Soc. 2002, 124, 3824–3825.
8. Wang, A.; Jiang, H. J. Org. Chem. 2010, 75, 2321–2326.
9. Xing, D.; Guan, B.; Cai, G.; Fang, Z.; Yang, L.; Shi, Z. Org. Lett. 2006, 8, 693–696.
10. (a) Bailey, P. S. Chem. Rev. 1958, 58, 925–1010; (b) Criegee, R. Angew. Chem., Int.
Ed. Engl. 1975, 14, 745–752; (c) Larock, R. C. Comprehensive Organic
Transformations, 2nd ed.; Wiley-VCH: New York, 1999.
p 1213; For the
explosion of gaseous ozone, see: (d) Koike, K.; Inoue, G.; Fukuda, T. J. Chem. Eng.
Jpn. 1999, 32, 295–299.
11. Franz, J. E.; Herber, J. F.; Knowles, W. S. J. Org. Chem. 1965, 30, 1488–1491.
12. (a) Miyamoto, K.; Tada, N.; Ochiai, M. J. Am. Chem. Soc. 2007, 129, 2772–2773;
(b) Miyamoto, K.; Sei, Y.; Yamaguchi, K.; Ochiai, M. J. Am. Chem. Soc. 2009, 131,
1382–1383; (c) Thottumkara, P. P.; Vinod, T. K. Org. Lett. 2010, 12, 5640–5643.
13. Kojima, H.; Takahashi, S.; Hagihara, N. Tetrahedron Lett. 1973, 14, 1991–1994.
14. Nwaukwa, S. O.; Keehn, P. M. Tetrahedron Lett. 1982, 23, 3135–3138.
15. Sawaki, Y.; Foote, C. S. J. Am. Chem. Soc. 1983, 105, 5035–5040.
28. For the characterization of ketones 4c and 4d, see: (a) Bottalico, D.; Fiandanese,
V.; Marchese, G.; Punzi, A. Synthesis 2009, 2316–2318; Compound 4d: (b) Liao,
Y.-X.; Hu, Q.-S. J. Org. Chem. 2010, 75, 6986–6989; Compound 4e: (c) Head, N.
J.; Olah, G. A.; Prakash, G. K. S. J. Am. Chem. Soc. 1995, 117, 11205–11210;
Compounds 4c and 4f: (d) Su, W.; Jin, C. Synth. Commun. 2004, 34, 4249–4256.

 Kang, Sunhae
Kang, Sunhae
 Lee, Soyoung
Lee, Soyoung
 Jeon, Minju
Jeon, Minju
 Kim, Sun Min
Kim, Sun Min
 Kim, Young Sug
Kim, Young Sug
 Han, Hogyu
Han, Hogyu
 Yang, Jung Woon
Yang, Jung Woon