610
D. Yuan et al. / Catalysis Communications 11 (2010) 606–610
The novel magnetic catalyst is stable and shows high catalytic
100
80
60
40
20
0
activity in promoting Heck cross-coupling reaction. In addition,
the novel catalyst can be easily recovered by using a permanent
magnet and reused at least six times without significant loss of
its catalytic activity and selectivity. As a consequence, magnetic
composite microspheres prepared by soap free emulsion can be
ideal supports for immobilization of industrial homogeneous cata-
lyst. Further studies on the effects of magnetic composite micro-
spheres properties on the catalytic activity of the immobilized
catalyst as well as the application of the novel catalyst to other pal-
ladium catalyst reactions are now in progress.
1
2
3
4
5
6
Appendix A. Supplementary data
reused times
a
Supplementary data associated with this article can be found, in
Fig. 5. Recycle of the Fe3O4/P (GMA–MMA–AA)–Pd(0)1
.
References
purification. As shown in Fig. 5a, the novel catalyst can be reused at
least six times only with a slight loss of activity for the Heck reac-
tion of iodobenzene and acrylic acid.
[1] Y. Uozumi, Top. Curr. Chem. 242 (2004) 77.
[2] J.M. Thomas, R. Raja, D.W. Lewis, Angew. Chem. Int. Ed. 44 (2005) 6456.
[3] D. Astruc, F. Lu, J.R. Aranzaes, Angew. Chem. Int. Ed. 44 (2005) 7852.
[4] J. Fan, S. Chen, Y. Gao, Colloids Surf. B 28 (2003) 199.
[5] A.J. Kell, D.L.B. Stringle, M.S. Workentin, Org. Lett. 2 (2000) 3381.
[6] P.D. Stevens, J.D. Fan, H.M.R. Gardinmalla, M. Yen, Org. Lett. 7 (2005) 2085.
[7] T.J. Yoon, W. Lee, Y.S. Oh, New J. Chem. 27 (2003) 227.
[8] B. Yoon, C.M. Wai, J. Am. Chem. Soc. 127 (2005) 17174.
[9] K. Marubayashi, S. Takizawa, T. Kawakusu, Org. Lett. 5 (2003) 4409.
[10] Y.H. Zhu, C.P. Ship, A. Emi, Adv. Synth. Catal. 349 (2007) 1917.
[11] Z.F. Wang, P.F. Xiao, B. Shen, Colloids Surf. A 276 (2006) 116.
[12] J.M. Liu, X.G. Peng, W. Sun, Org. Lett. 10 (2008) 3933.
[13] R.R. Abu, D. Wang, M. Post, H. Alper, Chem. Mater. 20 (2008) 2544.
[14] D.K. Yi, S.S. Lee, J.Y. Ying, Chem. Mater. 18 (2006) 2459.
[15] I. Willner, E. Katz, Angew. Chem. Int. Ed. 42 (2003) 4576.
[16] T.J. Yoon, B.G. Kim, K.N. Yu, Angew. Chem. Int. Ed. 44 (2005) 1608.
[17] B. Baruwati, D. Guin, S.V. Manorama, Org. Lett. 26 (2007) 5377.
[18] H. Yoon, S. Ko, J. Jang, Chem. Commun. 14 (2007) 1468.
[19] S. Ko, J. Jang, Angew. Chem. Int. Ed. 45 (2006) 7564.
[20] F.G. Guo, Q.Y. Zhang, B.L. Zhang, Polymer 50 (2009) 1887.
[21] S.V. Pislaru, A. Harbuzariu, R. Gulati, T. Witt, J. Am. Coll. Cardiol. 48 (2006)
1839.
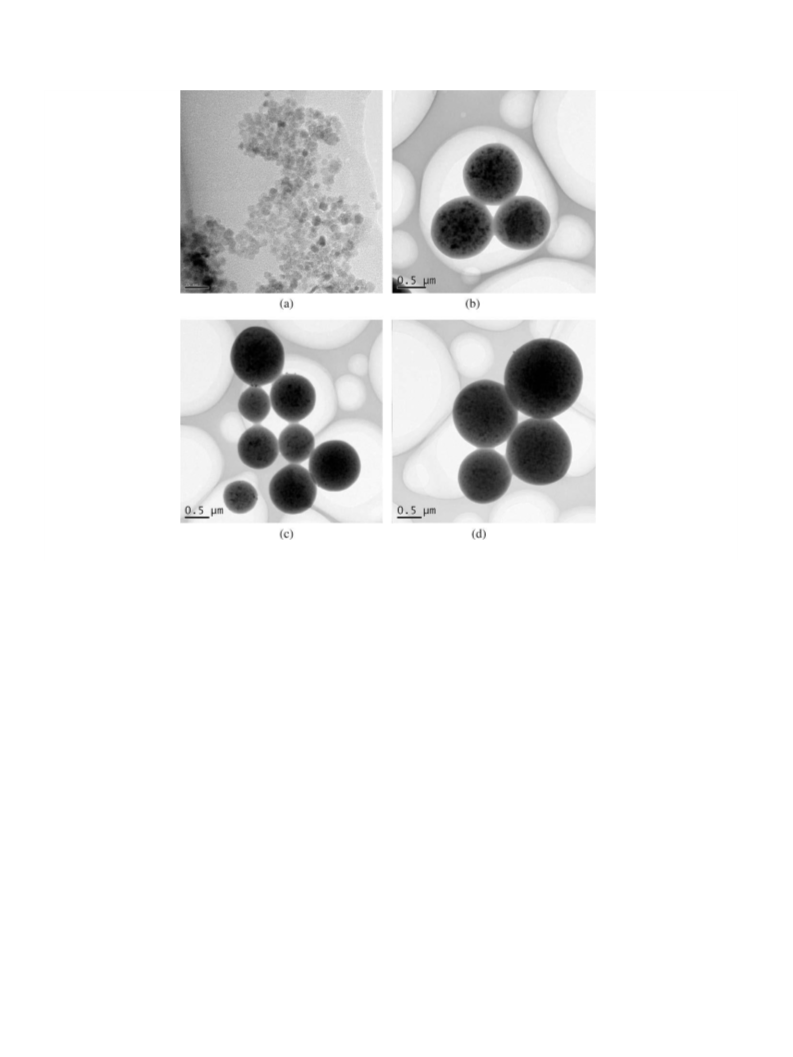
Furthermore, in order to check the stability of the catalyst, the
supported catalyst after sixth run for Heck reaction has been char-
acterized by AAS, TEM and XPS (see Supporting information). The
AAS result shows that only the leaching of 0.5 wt% palladium oc-
curs during the reaction, which indicates the coordination link of
Pd(0) with –NH2 ligand is strong, resulting that Pd(0) is strongly
bound to the surface of magnetic microspheres. Meanwhile, from
the TEM micrograph of the supported catalyst after sixth run, we
did not observe much change compared to the TEM image of the
fresh catalyst. And only few Pd nanoparticles have grown into big-
ger nanoparticles, which also indicate that the magnetic catalyst is
very stable during the reaction process. Additionally, as shown in
the XPS spectra of the catalyst reused after sixth run, only Pd(0)
particles is present in the re-catalyst, which indicates that the
Pd(II) in the fresh catalyst has been reduced to Pd(0) during the
Heck reaction, and the Pd(0) is the center of catalytic activity [29].
Overall, all these results show that the novel catalyst is an
excellent potential candidate for reusable and recoverable catalyst.
[22] K.M. Partington, E.J. Jenkinson, G. Anderson, J. Immunol. Methods 223 (1999)
195.
[23] G. Yao, D. Qi, C. Deng, J. Chromatogr., A 1215 (2008) 82.
[24] G.S. Lai, H.L. Zhang, D.Y. Han, Sensor Actuat. B 129 (2008) 497.
[25] D. Shao, A. Xia, J. Hua, C. Wang, W. Yu, Colloids Surf. A 322 (2008) 61.
[26] L.H. Fan, Y.L. Luo, Y.S. Chen, J. Nanopart. Res. 11 (2009) 449.
[27] R. Jay, Anal. Chim. 667 (1964) 36.
4. Conclusions
[28] A.G. Latha, B.K. George, K.G. Kannnan, K.N. Ninan, J. Appl. Polym. Sci. 43 (1991)
1159.
In conclusion, magnetic composite microspheres supported pal-
ladium complex catalyst has been prepared by immobilizing Pd
nanoparticles on the surface of magnetic composite microspheres.
[29] P. Liu, Y.H. Yang, Y. Liu, React. Funct. Polym. 68 (2008) 384.
1
Reactions were carried out with the magnetic catalyst (0.5 mol% Pd), iodobenzene
(5 mmol), acrylic acid (7.5 mmol), Bu3N (10 mmol), DMF (5 ml) at 95 °C in the air,
reaction time (3 h).

 Yuan, Dingzhong
Yuan, Dingzhong
 Zhang, Qiuyu
Zhang, Qiuyu
 Dou, Jinbo
Dou, Jinbo