THE CATALYTIC MECHANISM OF GLYAT
347
nical Industries (Pretoria, South Africa). The pColdIII vector was modified to
encode a C-terminal hexahistidine tag after the XhoI site. The recombinant
plasmid was sequenced to confirm that bovine GLYAT had been cloned
without any sequence aberrations.
major segment of biochemical genetics. One of the primary mecha-
nisms of pathogenesis in CASTOR disorders is depletion of free CoA,
which derails cellular metabolism. GLYAT converts the accumulated
acyl-CoA to acylglycines and free CoA, restoring levels of free CoA
and carnitine (Sakuma, 1991). Significant interindividual variation in
glycine conjugation capacity has been demonstrated using human
liver samples (Temellini et al., 1993). The basis for this variability is
not understood, but genetic variations in the coding sequence may be
a factor. Six nonsynonymous single-nucleotide polymorphisms have
been identified in the open reading frame of the human GLYAT gene.
However, it is not yet understood whether, or how, these variations
influence enzyme function (Yamamoto et al., 2009; Lino Cardenas et
al., 2010).
Because of the large number of compounds metabolized by
GLYAT, the human ortholog is of clinical interest. However, we have
not yet been able to express an enzymatically active recombinant
human GLYAT. Here, we report the bacterial expression and enzy-
matic investigation of a recombinant bovine GLYAT. Bovine
GLYAT is an enzyme expressed in bovine liver and kidney mito-
chondria, with a molecular mass reported to be between 33 and 36
kDa (Nandi et al., 1979; van der Westhuizen et al., 2000). Investiga-
tions of human and bovine GLYAT have shown that these enzymes
are similar in terms of molecular mass, reaction kinetics, and substrate
specificity (Bartlett and Gompertz, 1974; Kelley and Vessey, 1993;
van der Westhuizen et al., 2000). The recombinant bovine GLYAT,
combined with molecular modeling and site-directed mutagenesis,
was used to investigate the catalytic mechanism used by GLYAT. The
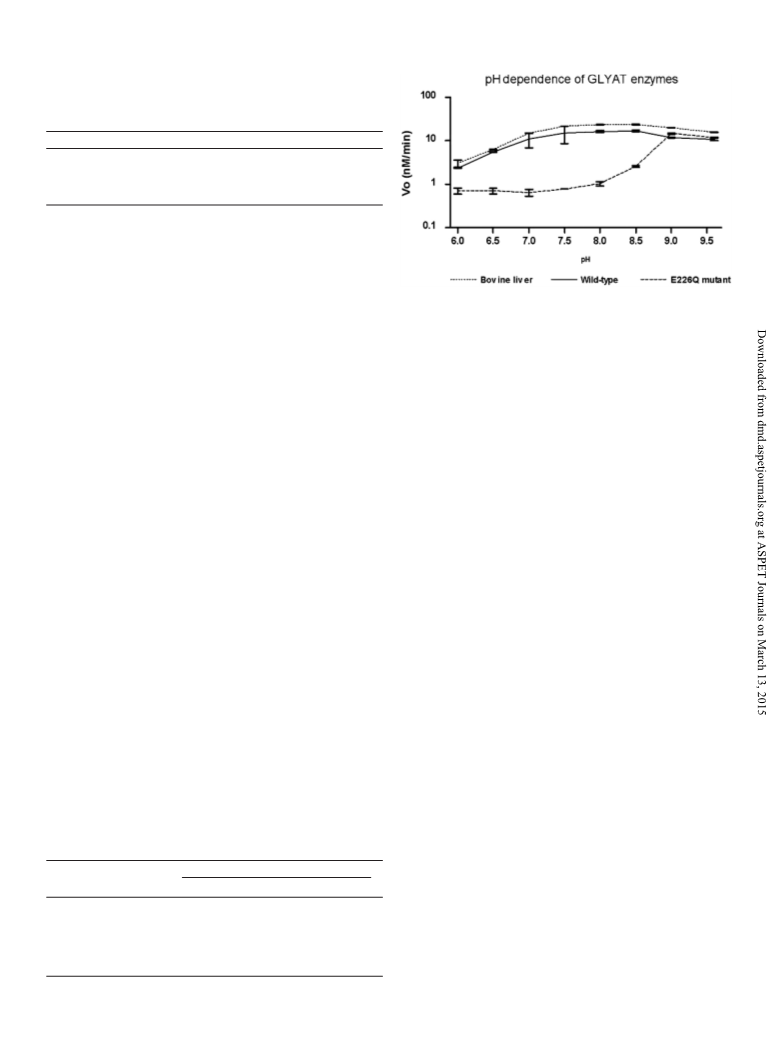
data suggest that residue Glu226 of bovine GLYAT serves as a general
base catalyst. The identification of this catalytic residue provides the
first insights into the catalytic mechanism and active site location of
the GLYAT enzymes.
Construction of the E226Q Mutant Recombinant Bovine GLYAT. Site-
directed mutagenesis using a mega-primer method (Aiyar and Leis, 1993) was
used to generate the E226Q mutant coding sequence. In a first polymerase
chain reaction, the mutagenic oligonucleotide primer 5Ј-CCA GAC GGG ACA
GAT GCG GAT GG-3Ј was used with the reverse primer 5Ј-CTT CTC GAG
AGG CTC ACA GTT CCA CTG G-3Ј to generate a 240-base pair amplicon.
This amplicon was gel-purified and was used in a second polymerase chain
reaction with the forward primer 5Ј-GCC GCA TAT GAT GTT CCT GCT
GC-3Ј to generate a full-length mutated GLYAT coding sequence. The mutated
amplicon was gel-purified, digested with NdeI and XhoI, and cloned into pColdIII.
Expression and Nickel-Affinity Purification of Wild-Type and E226Q
Recombinant Bovine GLYAT. The pColdIII-bovine GLYAT plasmid was
introduced into Origami cells (Novagen, Madison, WI) by electroporation.
Expression of the recombinant GLYAT was performed as follows. The cells
from 50 ml of overnight cultures in Luria Bertani medium, containing 100
g/ml ampicillin, were harvested by centrifugation at 4000g for 5 min. The
cells were resuspended in 200 ml of Luria broth medium containing 50 g/ml
ampicillin. The cultures were gently shaken at 15°C for 1 h before isopropyl-
1-thio--D-galactopyranoside was added to a final concentration of 0.5 mM.
The cultures were incubated at 15°C for 24 h with vigorous shaking. Cells were
then harvested by centrifugation at 4000g for 20 min. The cell pellets were
resuspended in 5 ml of BugBuster protein extraction reagent (Novagen)
containing 30 U/ml lysozyme (Novagen) and 25 U/ml Benzonase nuclease
(Novagen), followed by incubation at room temperature for 5 min. Insoluble
material was removed by centrifugation at 12,000g for 25 min at 4°C. The
cleared lysates were passed through Protino Ni-TED 2000 columns (Mach-
erey-Nagel, Du¨ren, Germany) equilibrated with buffer LEW (Macherey-Na-
gel). The columns were washed with 10 ml of buffer LEW containing 20 mM
imidazole. The bound protein was eluted from the columns in 9 ml of buffer
EB (Macherey-Nagel) and was added to Vivaspin 20 ultrafiltration devices
(GE Healthcare, Chalfont St. Giles, Buckinghamshire, UK). The proteins were
concentrated to approximately 500 l by centrifugation in a fixed-angle
centrifuge at 8000g for 15 min. The proteins were then washed by adding 10
ml of 50 mM Tris-HCl, pH 8.0, and repeating the centrifugation to again
concentrate the solution to approximately 500 l. Protein expression and
purification were monitored by means of SDS-polyacrylamide gel electropho-
resis (PAGE) analyses and Coomassie Brilliant Blue staining (Laemmli, 1970).
Preparation of an Extract Containing Bovine Liver Mitochondrial
GLYAT. To prepare an extract containing bovine liver mitochondrial
GLYAT, 100 g of liver tissue was homogenized in 400 ml of 0.13 M KCl. The
homogenate was centrifuged for 10 min at 600g. The supernatant was centri-
fuged again at 9000g for 10 min to isolate mitochondria, which were then lysed
by three cycles of freezing and thawing in 10 ml of 0.13 M KCl. The lysates
were clarified by centrifugation at 35,000g at 4°C for 2 h. The GLYAT enzyme
was further enriched from these lysates by collecting the fraction soluble
between 40 and 60% ammonium sulfate. The precipitate was dissolved in 4 ml
of 50 mM Tris-HCl, pH 8.0, and was dialyzed overnight against 1000 ml of 50
mM Tris-HCl, pH 8.0 (van der Westhuizen et al., 2000). This crude mitochon-
drial GLYAT preparation is referred to as “bovine liver GLYAT” in the
remainder of this article.
Materials and Methods
Sequence Analysis and Molecular Modeling. The bovine GLYAT amino
acid sequence (NP_803479) was submitted to the GenTHREADER server
(Jones, 1999) for identification of potential structural homologs. An unchar-
acterized protein from Drosophila melanogaster, with Protein Data Bank
(PDB) code 1SQH, was identified as the best homolog and was used for
molecular modeling. Although this protein is only 13% identical to bovine
GLYAT, it is structurally very similar to other GNAT enzymes and was used
as template structure because of the exceptional conservation of structure in the
GNAT superfamily of acyltransferases, despite there being virtually no se-
quence similarity between some members of the superfamily (Vetting et al.,
2005). The alignment generated by GenTHREADER was used with the struc-
ture of 1SQH as input for model generation using MODELLER 9.3 (Eswar et
al., 2008). For side-chain modeling, SCWRL 3.0 (Dunbrack Lab, Fox Chase
Cancer Center, Philadelphia, PA) was used (Wang et al., 2008). The molecular
model was superimposed with the structures of serotonin N-acetyltransferase
(PDB code 1CJW), diamine N-acetyltransferase (PDB code 2Q4V), and Esa1
(PDB code 1GHE) using the matchmaker algorithm of University of Califor-
nia, San Francisco, Chimera (UCSF Chimera) (Pettersen et al., 2004). UCSF
Chimera was used to generate images of the molecular model. The bovine
GLYAT amino acid sequence was also submitted to a Basic Local Alignment
aligned using CLUSTALX 2.0.10.
Enzyme Assays for Determination of KM Parameters. Reaction mixtures
were 400 l in volume and contained, in addition to enzyme, 25 mM Tris-HCl,
pH 8.0, 0.1 mM DTNB [5,5Ј-dithiobis(2-nitrobenzoic acid)], and varying
concentrations of substrates (Kølvraa and Gregersen, 1986). The glycine
concentration was varied from 2.5 to 20 mM, and the benzoyl-CoA concen-
tration was varied from 5 to 50 M. The assays were performed at 30°C, and
the change in absorbance at 412 nm over the first 4 min was measured using
Cloning of the Bovine GLYAT Open Reading Frame into the Bacterial
Expression Vector pColdIII. Total RNA was isolated from bovine liver tissue
using the RNeasy Mini Kit (QIAGEN, Valencia, CA). Cloned avian myelo-
blastosis virus reverse transcriptase (Invitrogen, Carlsbad, CA) was used to a Uvikon XS spectrophotometer (NorthStar Scientific, Bardsey, Leeds, UK)
generate cDNA from the bovine liver mRNA. The open reading frame of (van der Westhuizen et al., 2000). The absorbance change at 412 nm is the
bovine GLYAT was amplified from the cDNA using oligonucleotide primers
result of reduction of DTNB by the liberated thiol group of CoA, forming
(5Ј-GCC GCA TAT GAT GTT CCT GCT GC-3Ј and 5Ј-CAT CTC GAG TCA 2-nitro-5-thiobenzoate. 2-Nitro-5-thiobenzoate is a yellow species that absorbs
CAG AGG CTC AC-Ј3) that contained NdeI and XhoI restriction endonu-
clease recognition sites to facilitate cloning into pColdIII (Takara Bio USA,
at 412 nm with an extinction coefficient of 13.6 mMϪ1cmϪ1 (Ellman, 1959).
To each reaction, 1 unit of GLYAT activity was added. A unit of GLYAT
Madison, WI). Oligonucleotide primers were obtained from Inqaba Biotech- activity was defined as a change in A412 of 0.24 units, in 4 min, using cuvettes

 Badenhorst, Christoffel P. S.
Badenhorst, Christoffel P. S.
 Jooste, Maritza
Jooste, Maritza
 Van Dijk, Alberdina A.
Van Dijk, Alberdina A.