November 2002
1451
(
1.0 ml, 8.7 mmol) was stirred for 2 h at room temperature (nitrogen atmo- mogenate was centrifuged at 140000ꢆg for 1 h at 4 °C in a SW 60 Ti rotor
sphere). The reaction mixture was neutralized with an aqueous sodium bi-
carbonate solution to a pH of 7 and diluted with chloroform (10 ml). The or-
(Beckman Instruments, Palo Alto, California). Cytosol protein content was
determined by Bradford’s dye binding method using bovine serum albu-
12)
ganic phase was separated, washed with water and drier over anhydrous min (BSA) as the standard.
sodium sulfate; the solvent was eliminated in vacuum. The crude product
Androgen Receptor Binding Assay Assays were performed essentially
was purified by silica gel column chromatography. Hexane–ethyl acetate as described by Cabeza et al.
13)
(
8 : 2) eluted 674 mg, 1.54 mmol (69%) of pure product 7, mp 160—165°C.
Saturation Curves and Determination of the Binding Parameters
UV (nm) 284 (eꢄ23100). IR (KBr) cm 1723, 1705, 1668, 1618. H-NMR Binding sites were saturated in the presence of increasing concentrations
ꢅ1
1
3
14)
(
(
(
CDCl ) d: 0.8 (3H, s), 1.1 (3H, s), 1.4 (3H, d, Jꢄ2 Hz), 1.98 (3H, s), 5.8
(0.5—2.5) of [ H]DHT. Results were analyzed with Scatchard plots, the
1H, s), 6.1 (2H, d, Jꢄ3 Hz). C-NMR (CDCl ) d: 16.0 (CH3 at C-16), 17.0 calculation of affinity (K ) and the total number of sites (Bmax) were deter-
mined by linear regression.
3
1
3
3
d
C-18), 19.0 (C-19), 29.0 (C-21), 122.0 (C-4), 130.0 (C-6), 179.0 (ester car-
ꢂ
bonyl), 195.0 (C-3), 206.0 (C-20). MS (m/z) 438 (M ).
Aliquots of 120 ml of cytosol (2.64 mg of protein) were incubated (in du-
plicate) for 18—20 h at 4 °C in tubes containing 0.5—2.5 nM [ H]DHT. Par-
allel sets of tubes containing identical concentrations of [ H]DHT plus 100-
3
1
7a-Cyclopentylacetoxy-16b-methylpregna-1—4,6-triene-3,20-dione
3
8
A solution of steroid 6 (1 g, 2.21 mmol) and DDQ (2.21 g, 26.44 mmol)
in dioxane (90 ml) was allowed to reflux for 2 d. Upon cooling, the precipi- fold excess of radioinert DHT were also incubated. Dextran coated charcoal
tated 2,3-dichloro-5,6-dicyanohydroquinone was filtered off. To the filtrate (800 ml) in TEDAM buffer was added and the mixture was incubated for
was added 3% aqueous sodium hydroxide solution (100 ml) and chloroform
45 min at 4 °C. The tubes were vortexed and immediately centrifuged at
(
100 ml); the mixture was stirred for 5 min. The organic phase was sepa- 800ꢆg for 10 min.
rated, washed 3 times with 3% aqueous sodium hydroxide solution and
Competition Analysis of Synthesized Steroids for ARs The competi-
water. It was dried over anhydrous sodium sulfate and the solvent was re- tion analysis of steroids 4—8 for the androgen receptors of castrated male
moved in vacuum. The crude product was purified by silica gel column chro- hamster seminal vesicles was determined on 58 animals (150—200 g), se-
matography. Hexane–ethyl acetate (6 : 4) eluted 850 mg, 1.74 mmol (70%) of lected at random and gonadectomized 30 d before the experiments. The ani-
pure product 8, mp 163—164°C. UV (nm) 222, 256, 299 (eꢄ15200, 13000,
mals were kept in a room with controlled temperature (22 °C) and light–dark
8000 respectively). IR (KBr) cm : 1724, 1710, 1668, 1649. H-NMR periods of 12 h. Food and water were provided ad libitum.
CDCl ) d: 0.8 (3H, s), 1.1 (3H, s), 1.41H, (3H, d, Jꢄ2 Hz), 2.0 (3H, s), 2.4
Androgen receptor competition studies were performed essentially as de-
ꢅ1
1
1
(
(
3
13)
2H, d, Jꢄ3 Hz), 6.1 (1H, d, Jꢄ2 Hz), 6.4 (1H, d, Jꢄ2 Hz), 6.6 (1H, d, scribed by Cabeza et al. and are briefly summarized below.
1
3
Jꢄ2 Hz), 6.8 (1H, d, Jꢄ3 Hz). C-NMR (CDCl ) d: 15.0 (C-18), 19.8 (C-
Competition Studies For competition experiments, tubes contained
1.26 nM [ H]DHT (specific activity 110 Ci/mmol) plus a range of increasing
3
3
1
9), 28.0 (C-21), 130.0 (C-6), 164.0 (C-5), 173.0 (ester carbonyl), 186.0 (C-
), 204.0 (C-20). MS (m/z) 450 (M ).
Animal and Tissues Adult male Syrian Golden hamsters (150—200 g)
were obtained from the Metropolitan University-Xochimilco of Mexico. Go-
ꢂ
3
concentrations (50—500 mM) of the following unlabeled steroid hormones:
5-a DHT, and 4—8 were prepared.
Aliquots 120 ml of cytosol (2.64 mg of protein) were added and incubated
nadectomies were performed under light ether anesthesia 30 d before treat-
(in duplicate) for 18—20 h at 4 °C in tubes mentioned above. Parallel sets of
3
ment. Animals were sacrificed by ether anesthesia. The seminal vesicles tubes containing identical concentrations of [ H]DHT plus a 100-fold excess
were immediately removed, blotted and weighed prior to their use. Tissues of radio inert DHT were also incubated; 800 ml of dextran-coated charcoal in
used in the metabolic experiment were homogenized with a tissue homoge- TEDAM buffer was added and the mixture was incubated for 45 min at 4 °C.
nizer (model 985-370; variable speed 5000—30000 rpm, Biospec Products,
Inc.).
The dextran was agitated during 30 min before the addition of charcoal to
the mixture. The tubes were vortexed and incubated for 45 min at 4 °C. After
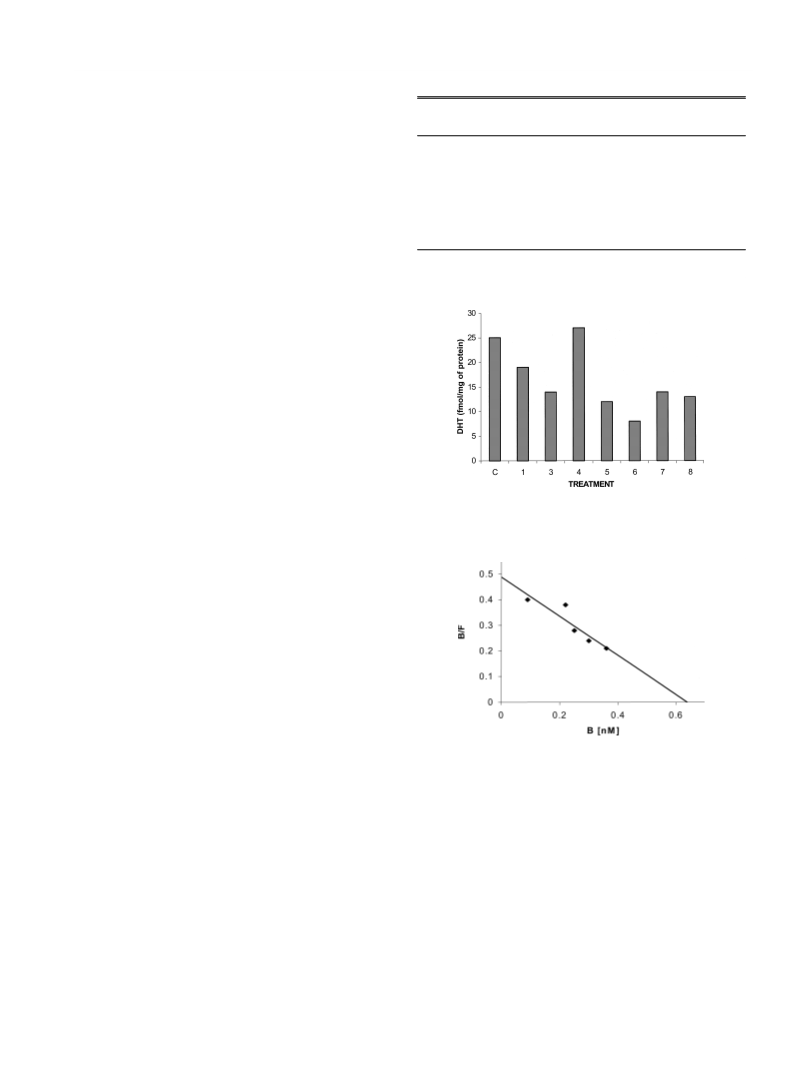
Animal Treatment The biological activity of steroids 4 to 8 (Fig. 1) the tubes were centrifuged at 800ꢆg for 10 min; aliquot (200 ml) were sub-
were determined in gonadectomized male hamsters divided in several sequently submitted to radioactive counting. Specific binding was deter-
groups. Daily subcutaneous injections of 400 mg of the steroids 4—8 (Fig. 1) mined by subtracting the mean disintegration per minute (dpm) in the pres-
dissolved in 200 ml of sesame oil were administered for 6 d together with ence of excess unlabeled steroids from the mean dpm of corresponding
3
2
00 mg of T. Three groups of animals were kept as a control, one was in- tubes containing only [ H]DHT. Results were analyzed using one-way analy-
jected with 200 ml of sesame oil, the second with 200 mg of testosterone and
sis of variance with EPISTAT software. The inhibition constant (K ) of each
i
the third with T together with finasteride for 6 d. After the treatment, the ani- compound was calculated according to the procedures described by Cheng
17)
mals were sacrificed by ether anesthesia and the seminal vesicles were dis- and Prusoff.
sected and weighed.
In Vitro Metabolic Studies with Seminal Vesicles Homogenates from
Acknowledgements We gratefully acknowledge the financial support of
male hamster seminal vesicles (35.34 mg of protein) were prepared from in- DGAPA and CONACYT for the projects IN 200301 and G33450-M.
tact adult male animals, using Krebs–Ringer phosphate buffer solution at pH
1
1)
of 6. Tissue preparations were incubated in duplicate with 3.4 mCi of
References
3
ꢂ
[
H]T (specific activity 95 Ci/mmol) in the presence of 0.5 mM NADPH
and 12.5 mM glucose-6-phosphate plus 0.8ꢆ10 g of glucose-6-phosphate
1) Bruchovski N., Wilson J. D., J. Biol. Chem., 243, 2012—2021 (1968).
2) Metcalf W. B., Levy M. A., Holt D. A., Trends Pharmacol Sciences,
10, 491—495 (1989).
3) Imperato-McGinley J., Guerrero L., Gautier T., Peterson R. E.,
Science, 27, 1213—1215 (1974).
4) Imperato-McGinley J., Peterson R. E., Leshin M., Griffin J. E., Cooper
G., Draghi S., Berenyi M., Wilson J. D., J. Clin. Endocrinol. Metab.,
50, 15—22 (1980).
ꢅ6
1)
dehydrogenase and 8.7 mM of finasteride or steroids 4—8 in a Dubnoff
metabolic incubator at 37 °C for 60 min in the presence of air. The final in-
cubation volume was 3 ml. The same experiment was performed with each
one of the new steroidal compounds. Incubation without tissues was used as
a control. The incubation was terminated by addition of dichloromethane
3
and the [ H] steroid was extracted (4ꢆ) using 10 ml of dichloromethane.
The protein content of the homogenates was determined by Bradford’s dye-
5) Wilbert D. M., Griffin J. E., Wilson J. D., J. Clin. Endocrinol. Metab.,
56, 113—120 (1983).
6) Weisser H., Tunn S., Debus M., Krieg M., Steroids, 59, 616—620
(1994).
7) Brooks J. R., Berman C., Primka R. L., Reynolds G. F., Rasmusson G.
H., Steroids, 47, 1—19 (1986).
12)
binding method using bovine serum albumin (BSA) as the standard. Isola-
tion and purity assessment of radioactive DHT was carried out. The isolated
compound was purified with steroid carriers (T and DHT) by a thin layer
chromatographic system (chloroform : acetoneꢄ9 : 1). The steroid carriers
were detected using phosphomolibdic acid reagent and an ultraviolet lamp
(
254 nm). Radioactivity was determined in a Packard Tri carb 2100 TR liq-
8) McConnel J., Wilson J. D., George F. J., J. Clin. Endocrinol. Metab.,
25, 505—509 (1992).
9) Cabeza M., Gutierrez E., Miranda R., Heuze I., Bratoeff E., Flores G.,
Ramirez E., Steroids, 64, 413—421 (1999).
uid scintillation Analyzer, using Ultima Gold (Packard Downers Grove, IL)
as counting vehicle. The counting efficiency of [ H] was 67%. The losses of
radioactivity during the procedure were calculated in agreement with the re-
3
sults obtained from the control experiment without tissue. The formation of 10) Cabeza M., Quiroz A., Bratoeff E., Murillo E., Ramirez E., Flores G.,
DHT was calculated and expressed as fmol of DHT/mg protein/h.
Chem. Pharm. Bull., 47, 1232—1236 (1999).
Cytosol Preparation for Androgen Receptor Competition Analysis 11) Ramírez E., Cabeza M., Heuze I., Gutiérrez E., Bratoeff E., Membrillo
12)
Tissue homogenization was performed as described above using TEDAM
:3 20 mM Tris–HCl, pH 7.4 at 4 °C, 1.5 mM EDTA, 0.25 mM dithiothreitol,
and 10 mM sodium molibdate containing 10% glycerol (v/v). The ho-
M., Lira A., Chem. Pharm. Bull., 50, 15—20 (2002).
12) Bradford M. M., Anal. Biochem., 72, 248—254 (1986).
13) Cabeza M., Vilchis F., Lemus A. E., Díaz de León L., Pérez-Palacios
1

 Cabeza, Marisa
Cabeza, Marisa
 Bratoeff, Eugene
Bratoeff, Eugene
 Flores, Eugenio
Flores, Eugenio
 Ramirez, Elena
Ramirez, Elena
 Calleros, Jorge
Calleros, Jorge
 Montes, Diana
Montes, Diana
 Quiroz, Alexandra
Quiroz, Alexandra
 Heuze, Ivonne
Heuze, Ivonne