H. Wang et al. / Applied Catalysis A: General 523 (2016) 182–192
183
have been made to obtain high surface area W/Zr catalysts through
various preparation methods. Cheng et al. [30] introduced a
mixture of W/Zr oxides on various porous silica materials by
co-impregnation and applied these catalysts in n-pentane isomer-
ization. Jiménez-López et al. [31] prepared a series of catalysts by
supporting WO3 on Zr-doped MCM-41 using impregnation and
used the catalysts for the esterification of oleic acid with methanol.
Recently, Yi and co-workers [32] synthesized Zr-WOx modified
mesoporous SiO2 catalysts using a refined hydrothermal method
for esterification of 1-butanol with acetic acid, which exhibited
WO /ZrO , which was used for comparison with WZr/SBA-15 sam-
3 2
ples prepared at the same calcination temperature and with same
◦
amount of WO loadings) and 700 C (for 20% WO /ZrO , which was
3
3
2
the optimized condition to achieve the highest amounts of Zr-WO3
in the WO /ZrO2 catalysts in reference [27]) for 3 h, respectively. A
3
reference WO3 rich catalyst (denoted as W30Zr5) with WO :ZrO2
3
ratio of 30:5 was prepared using the same method described above,
◦
followed calcination at 550 C for 3 h.
much higher activity than HZSM-5 and conventional WO /ZrO2
catalysts.
3
2.2. Characterizations
In this work, high surface area WZr/SBA-15 catalysts were pre-
pared by hydrothermal method and characterized using a variety of
techniques to understand its nature and structure. The correlation
of the structure of the catalysts with their catalytic performance for
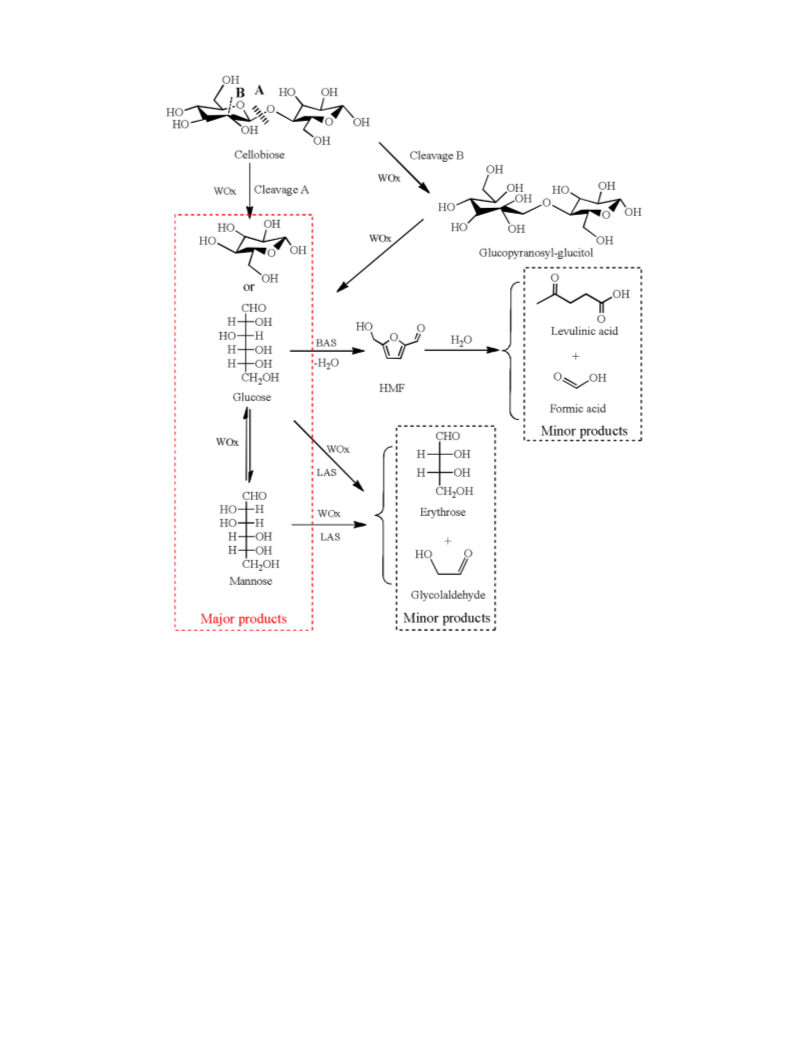
the hydrolysis of cellobiose was investigated. We found that dop-
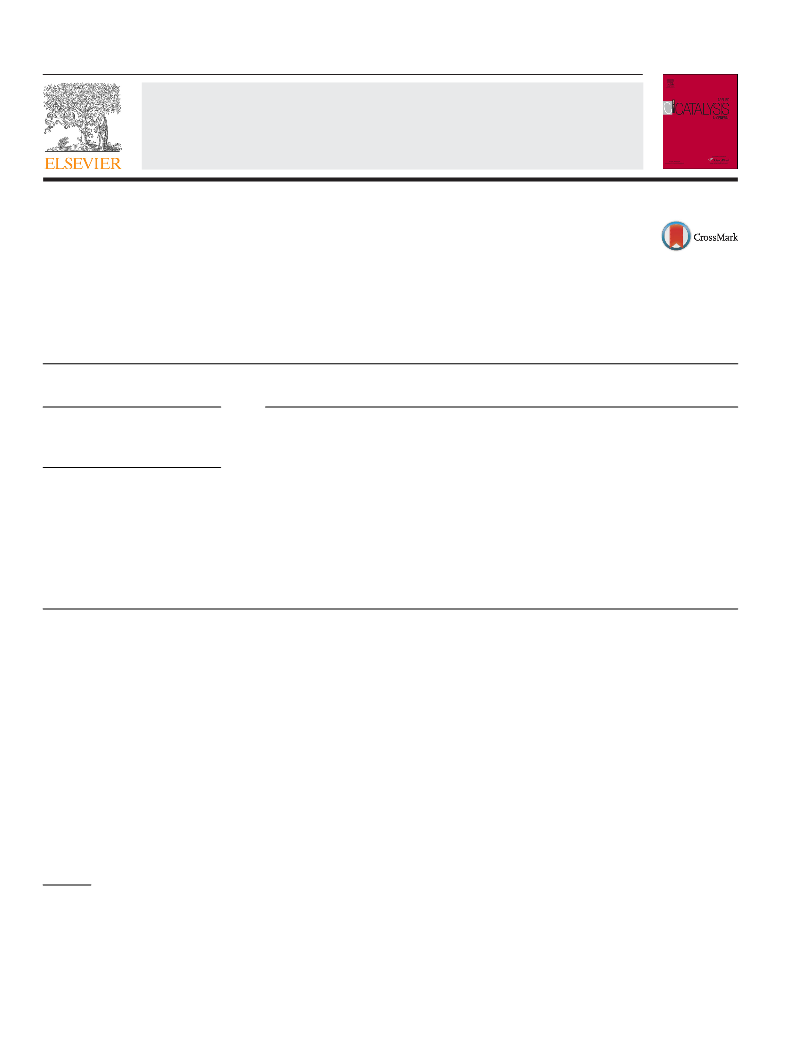
The nitrogen adsorption-desorption isotherms were obtained
on a Tristar 3000 (Micrometrics). Prior to measurements, sam-
◦
◦
ples were outgassed at 90 C for 1 h and then at 300 C for 3 h.
The specific surface area was calculated using the BET method.
The mesopore size distribution was obtained by applying the BJH
method to the adsorption branch of the isotherm.
ing proper amount of ZrO in W/SBA-15 maximized its surface area
2
and accessible strong acid sites, both contributed to a remarkably
increased activity for the hydrolysis of cellobiose to glucose and
mannose.
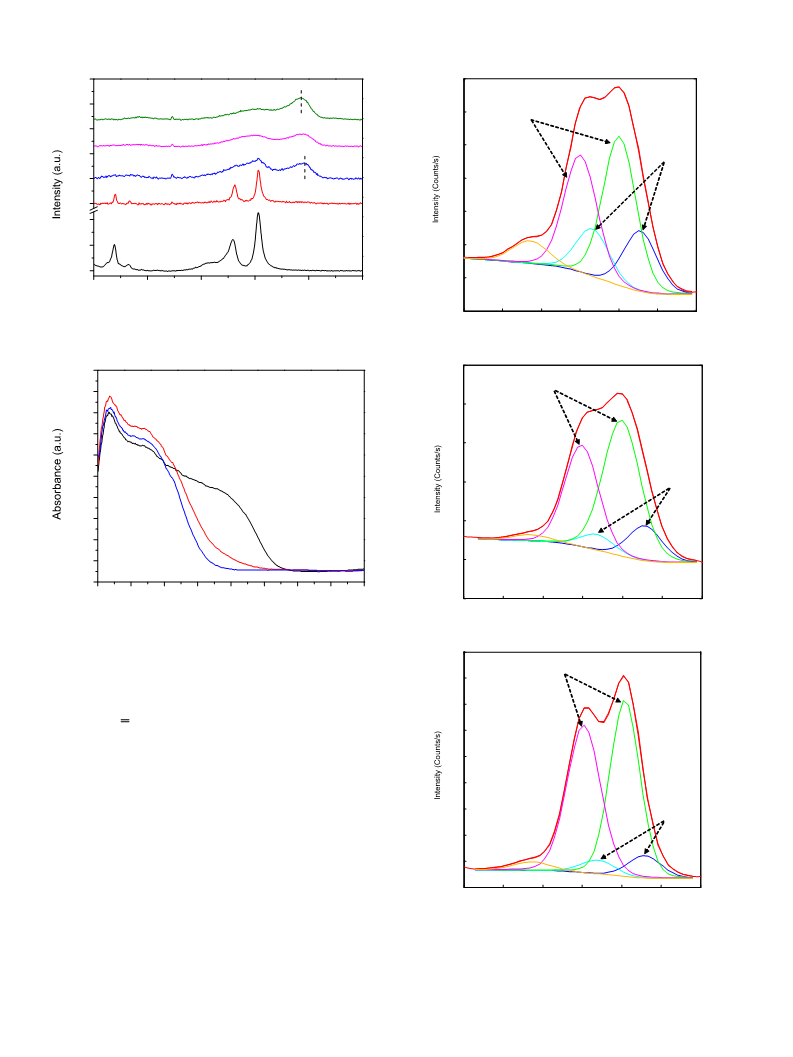
Powder X-ray diffraction (XRD) was performed on a D/MAX-
500 (Rigaku Corporation, Japan) XD-3A diffractometer using a Cu
2
K␣ radiation source, operated at 40 kV and 100 mA. The XRD pat-
◦
◦
terns were measured in the 2ꢀ range of 0.5–5 and 10–90 at a
2
2
2
. Experimental
◦
◦
scanning speed of 0.5 /min and 4 /min, respectively.
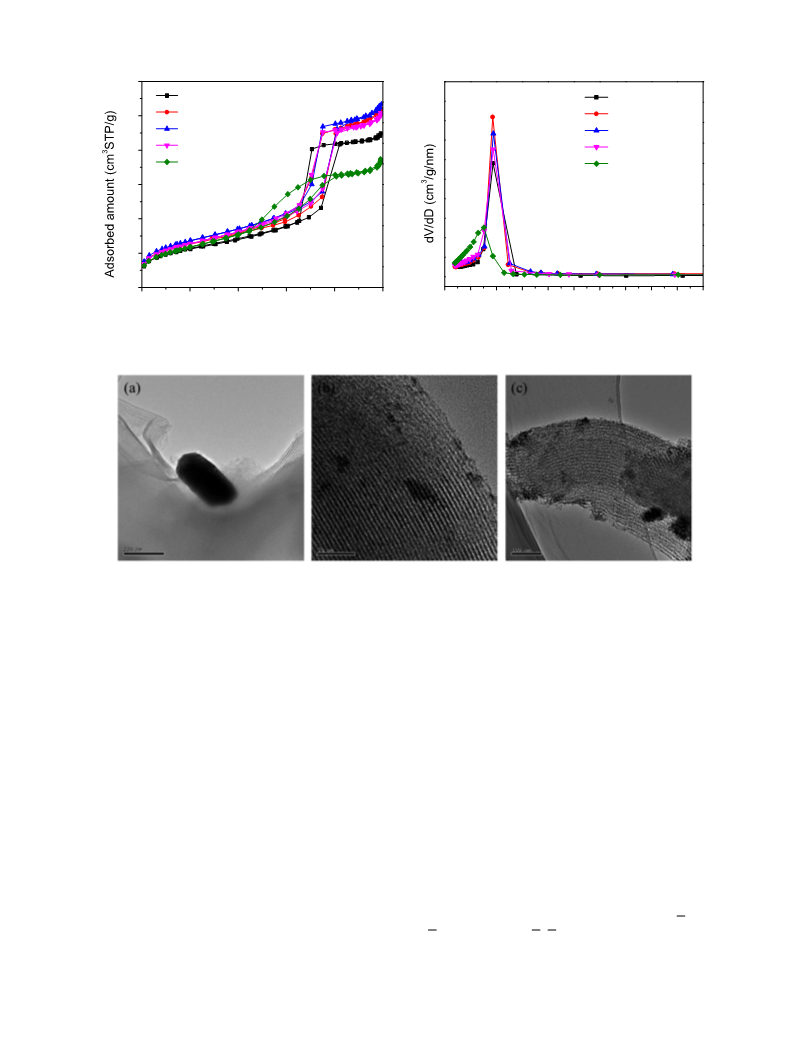
Transmission electron microscopy (TEM) images were obtained
on a JEM-1400 microscope (JOEL). Samples of fine powder were sus-
pended in ethanol with an ultrasonic treatment for 30 min and then
deposited on carbon-coated copper grids for TEM observations.
Raman spectroscopy was recorded on a DXR Microscope. The
He–Cd laser of 532 nm was employed as the exciting source with
an incident power of 30 mW.
Diffuse-reflectance ultraviolet–visible spectroscopy (DR
UV–vis) spectra were acquired using a UV-2550 spectrophotometer
Shimadzu Corporation, Japan).
.1. Catalyst preparation
.1.1. Chemicals
Hydrochloric acid (37%) and ethanol were purchased from Tian-
jin Kermel Chemical Reagent. Pluronic P123 was obtained from
Sigma–Aldrich. Zirconium butoxide solution (80 wt.% in n-butanol)
and ammonium metatungstate ((NH ) H W12O40·xH O) were
4
6
2
2
purchased from Tianjin Heowns Bio-Chem Technology. Zirconyl
chloride octahydrate (ZrOCl ·8H O), sodium tungstate dihydrate
2
2
(
(
Na WO ·2H O) and tetraethyl orthosilicate (TEOS) were obtained
2
4
2
X-ray photoelectron spectra (XPS) were measured on a PHI 1600
PerkinElmer). All binding energies were calibrated using the C1s
from Aladdin.
(
peak at 284.8 eV as the reference.
2
.1.2. Synthesis of the mesoporous WZr/SBA-15
The synthesis of the mesoporous WZr/SBA-15 followed the
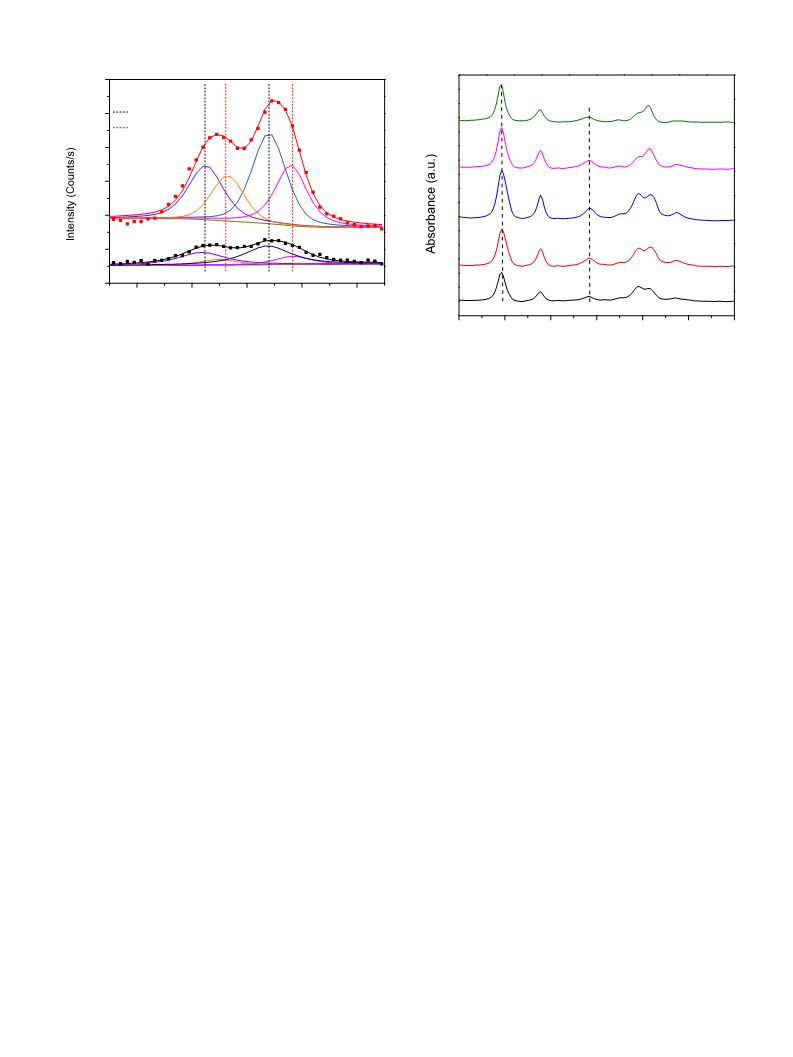
The pyridine adsorption FTIR spectra were recorded by
a
MAGNA-IR 560 spectrometer. In each measurement, self-
procedure reported by Yi [32]. Typically, pluronic 123 (2 g) was
dissolved in a mixture of de-ionized water (60 mL) and concen-
trated hydrochloric acid (6 mL). The mixture was stirred for 4 h
until a clear solution was obtained. Zirconium butoxide ethanol
solution (v/v = 1) was added drop-wise to the solution under vigor-
ous stirring. Then, an aqueous solution of sodium tungstate (3 mL)
was added drop-wise and stirred for another 1 h. TEOS of 4.17 g
supporting wafer (50 mg, 13 mm diameter) was first evacuated
◦
◦
in situ at 400 C for 1 h, followed by adsorption of pyridine at 60 C
◦
for 30 min. The spectra were recorded at 150 C after desorption.
The acid densities of Brønsted acid site and Lewis acid site were
estimated using the following equations [33]:
2
◦
CB = 1.88IA R /W
(1)
(2)
was introduced to the solution, which was then stirred at 40 C for
B
◦
2
4 h. Hydrothermal synthesis was carried out at 100 C for 24 h.
The resulting precipitate was filtered and washed with ethanol
2
CL = 1.42IALR /W
◦
and de-ionized water several times. Finally, it was dried at 80 C
◦
overnight and calcined at 550 C for 3 h. The catalysts are denoted
where, CB is the density of Brønsted acid site (mmol/gcat), CL is the
density of Lewis acid site (mmol/gcat), IAB and IAL are the integrated
as WxZry/SBA-15, with x and y being the WO3 and ZrO2 weight
percentage of the catalyst, respectively.
−
1
absorbance of Brønsted and Lewis band (cm ), respectively, R is
the radius of disk (0.65 cm), W is the weight of disk (mg), 1.88 and
2
.1.3. Synthesis of ZrO /SBA-15 and WO /SBA-15
2 3
1
.42 are the molar extinction coefficients (mol/cm) of pyridine
7
.1% ZrO /SBA-15 and 31.6% WO /SBA-15 were prepared using
2 3
adsorption on Brønsted acid site and Lewis acid site, respctively.
The acidic properties of the samples were also characterized by
the same procedure as the WZr/SBA-15 catalysts without the addi-
tion of sodium tungstate or zirconium butoxide, respectively.
temperature programmed desorption of ammonia (NH -TPD) in a
3
micro reactor equipped with a Cirrus 200 mass spectrometer (MKS),
as reported in previous work [34,35]. Catalyst sample of 50 mg was
2
.1.4. Synthesis of WO /ZrO
3 2
WO /ZrO was prepared using a conventional impregna-
3
2
◦
pretreated for 1 h in flowing He (30 mL/min) at 350 C to eliminate
tion method [27]. Briefly, concentrated ammonia was added
slowly to an aqueous solution of zirconyl chloride octahy-
drate until pH = 10. After stirring and aging, the precipitate was
separated by centrifugation and then dried. The as-prepared
precipitate was impregnated with an aqueous solution of ammo-
◦
any adsorbed water. After it was cooled down to 50 C, the sample
was exposed to 5% NH /He (30 mL/min) for 30 min, followed by
3
flushing with He (30 mL/min) for another 30 min to remove the
◦
physically adsorbed NH . Then the sample was heated to 800 C at
3
◦
a heating rate of 10 C/min.
◦
nium metatungstate, followed by calcination at 550 C (for 30%

 Wang, Hua
Wang, Hua
 Guo, Yingge
Guo, Yingge
 Chang, Cuirong
Chang, Cuirong
 Zhu, Xinli
Zhu, Xinli
 Liu, Xiao
Liu, Xiao
 Han, Jinyu
Han, Jinyu
 Ge, Qingfeng
Ge, Qingfeng