ACCEPTED MANUSCRIPT
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
8
Tetrahedron
4.2.4. 2-benzyl-2-methyl-malonyl dichloride for 3 Sodium
138.9, 148.5, 170.7. HRMS (EI+): m/z calcd for C29
460.1899, found 460.1894.
24 4 2
H N O
metal (9.8 g) was added to dry ethyl alcohol (500 mL) at 0°C and
stirred for 2.5 h until sodium metal dissolved completely to form
sodium ethoxide. Methylmalonic acid diethyl ester (43.7 g) was
added dropwise over period of 2 h. The reaction solution was
refluxed for 1h. Then benzyl chloride (31.6 g) was added and
refluxed for 48 h. After concentration in vacuo for the removal of
the solvent, the residue was added by water (50 mL). The
solution was extracted with diethyl ether (100×3 mL). The
organic layer was dried over anhydrous magnesium sulfate,
filtrated and evaporated under reduced pressure. The purification
was performed by distillation under reduced pressure to give 2-
benzyl-2-methyl-malonic acid diethyl ester. Yield: 3.63 g, 4.2%;
4.2.6.
N,N’-Bis(8--quinolyl)-2-methyl-2-
(4) 2-metyl-2-
naphthalenylmethylmalonamide
naphthalenylmethymalonyl dichloride was obtained following a
published procedure. 8-aminoquinoline (2.9 g) and dry pyridine
7
(5.0 mL) were dissolved in dry benzene (150 mL). In a dark
room, the dry benzene solution (30 mL) of 2-benzyl-2-methyl
malonyl chloride (3.9 g) was added dropwise to the solution and
stirred for 72h at room temperature. 0.5 M hydrochloric acid
aqueous solution (30 mL) and chloroform (150 mL) were added
to the solution. The solution was extracted with chloroform (50 ×
5 mL) and washed with water (50 × 2 mL). The organic layer
was dried over anhydrous magnesium sulfate, filtrated and
evaporated under reduced pressure. Yield: 1.60 g, 32%; pale
1
1
1
1
1
1
1
1
1
1
2
2
2
2
2
2
2
2
2
2
3
3
3
3
3
3
3
3
3
3
4
4
4
4
4
4
4
4
4
4
5
5
5
5
5
5
5
5
5
5
6
6
6
6
6
6
1
pale yellow tough liquid; bp 77.0 °C (0.4 mmHg). H NMR
(
3
300MHz NMR, CDCl
.22 (s, 2H), 4.15 (q, 4H, J = 7.20 Hz), 7.07–7.33 (m, 5H).
NMR (75MHz NMR, CDCl ): 13.54, 19.22, 40.68, 54.34, 60.74,
26.43, 127.70, 129.78, 135.83, 171.30. Sodium hydroxide (10.3
g) was added to dry ethyl alcohol (250 mL) at room temperature
and stirred for 1.5 until sodium hydroxide dissolved
3
): 1.20 (t, 6H, J = 7.20 Hz), 1.33 (s, 3H),
13
C
-1 1
yellow crystal; mp 160.1-163.9 °C. IR(KBr): 1691.5 cm . H
3
NMR (300MHz NMR, CDCl ): 1.83 (s, 3H), 3.81 (s, 2H), 7.33-
7.46 (m, 3H), 7.42 (dd, 2H, J = 4.20 and 8.40 Hz), 7.52 (dd, 2H,
J = 1.80 and 8.40 Hz), 7.62-7.78 (m, 4H) , 7.56 (dd, 2H, J = 7.20
and 8.40 Hz), 8.13 (dd, 2H, J = 1.80 and 8.40 Hz), 8.81 (dd, 2H,
J = 1.80 and 4.20 Hz), 8.91 (dd, 2H, J = 1.80 and 7.20 Hz), 11.11
3
1
h
completely. The gained 2-benzyl-2-methyl-malonic acid diethyl
ester (13.7 g) was added dropwise over period of 1h. The
reaction solution was refluxed for 23 h. After the filtration the
pale yellow solid was obtained and washed with dry ethyl
alcohol. After filtration, the precipitate was dried under reduced
pressure. The obtained solid was dissolved in water (150 mL).
Then the solution was became pH 1 by the addition of
hydrochloric acid to form the precipitate. After filtration, the
precipitate was dissolved in diethyl ether (100 mL) and the
filtrate was washed with diethyl ether (100 × 3 mL). These
diethyl ether solution was washed with water (100 × 2 mL). The
organic layer was dried over anhydrous magnesium sulfate,
filtrated and evaporated under reduced pressure. The gained solid
was washed with hexane to give 2-benzyl-2-methyl-malonic acid.
Further purification was performed by recrystallization with
13
(
br, 2H). C NMR (75MHz NMR, CDCl
3
): 18.5, 45.1, 57.5,
1
1
1
5
16.8, 121.6, 122.0, 125.5, 125.8, 127.1, 127.4, 127.6, 127.8,
27.8, 128.3, 129.1, 132.4, 133.3, 134.0, 134.3, 136.1, 138.9,
48.6, 170.7. HRMS(FAB+): m/z calcd for
10.2056, found [M+H] 511.2142.
33 26 4 2
C H N O
4.2.7. N-Mono(8-quinolyl)acetamide (5) 8-aminoquinoline
(2.7 g) was dissolved in dry toluene (200 mL). Triethylamine
(4.0 mL) and N,N’-dimethyl-4-aminopyridine (DMAP) (0.35 g)
was added to the solution. In a dark room, the dry toluene
solution (40 mL) of acetyl chloride (2.10 g) was added dropwise
to the solution and stirred for 18h at room temperature. After
filtration for the removal of the solid, the solution was washed by
water (100 × 5 mL). The organic layer was completely
evaporated under reduced pressure. Yield: 1.94 g, 57%; pale
1
acetone. Yield: 8.67 g, 80%; pale yellow crystal. H NMR
-
1
1
(
300MHz NMR, CDCl
3
): 1.46 (s, 3H), 3.29 (s, 2H), 7.18 (dd, 2H,
yellow solid; mp 94.2-96.6 °C. IR(KBr): 1666.4 cm . H NMR
J = 1.80 and 2.40 Hz), 7.27 (d, 3H, J = 7.20 Hz). 2-Benzyl-2-
methyl-malonic acid (3.49 g) and dry pyridine (2.3 mL) were
added in dry benzene (65 mL) and stirred for 2h at room
temperature. In a dark room, oxalyl chloride (8.0 mL) was added
dropwise and stirred for 22h. Then the reaction solution was
refluxed for 24h outside a dark room. After concentration in
vacuo for the removal of the solvent, the residue was distilled
under reduced pressure to give 2-benzyl-2-methyl malonyl
dichloride. Yield: 2.43 g, 59%; pale yellow liquid; bp 76.0 °C
(300MHz NMR, CDCl ): 2.35 (s, 3H), 7.43 (dd, 1H, J = 4.20 and
8.40 Hz), 7.47 (dd, 1H, J = 1.80 and 8.40 Hz), 7.52 (dd, 1H, J =
8.10 and 8.10 Hz), 8.13 (dd, 1H, J = 1.50 and 8.40 Hz), 8.77 (dd,
1H, J = 1.80 and 8.10 Hz), 8.78 (dd, 2H, J = 1.50 and 4.20 Hz),
9.78 (br, 1H). C NMR (75MHz NMR, CDCl ): 25.3, 116.4,
121.4, 121.6, 127.4, 127.9, 134.5, 136.4, 138.2, 148.1, 168.8.
HRMS (EI+): m/z calcd for C H N O 186.0793, found
3
13
3
11
10
2
186.0797.
-
1
(0.1 mmHg). IR (KBr): 1780, 943 cm .
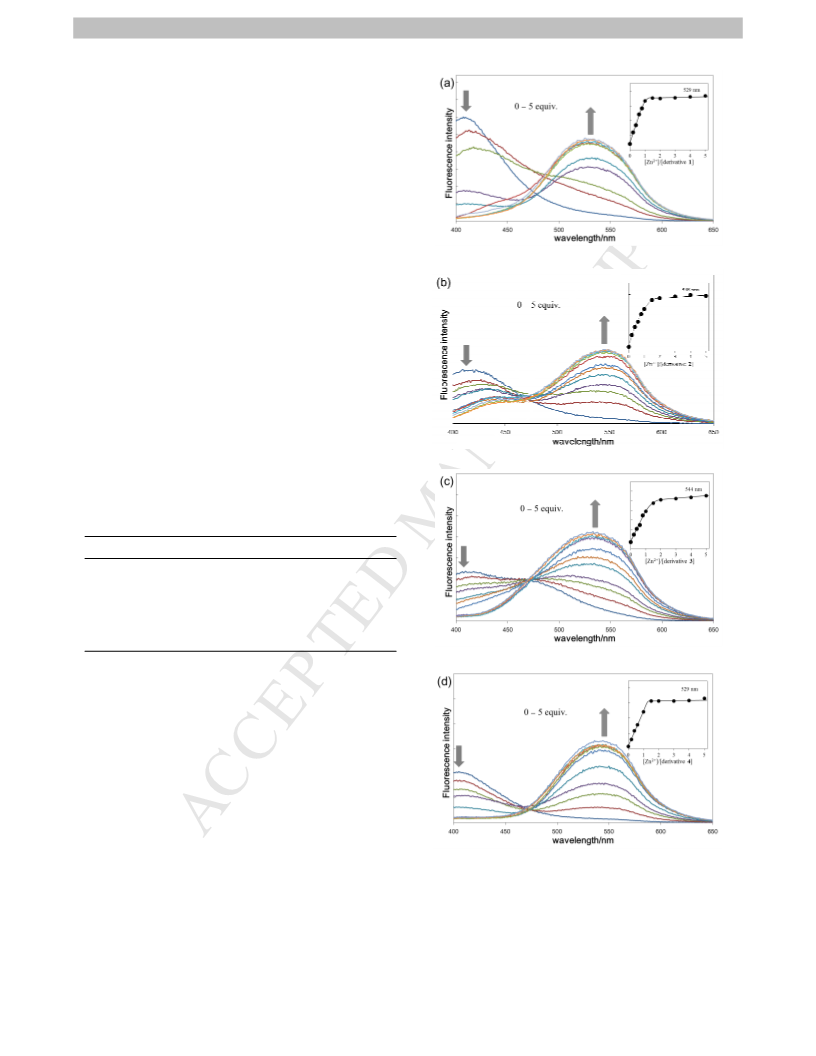
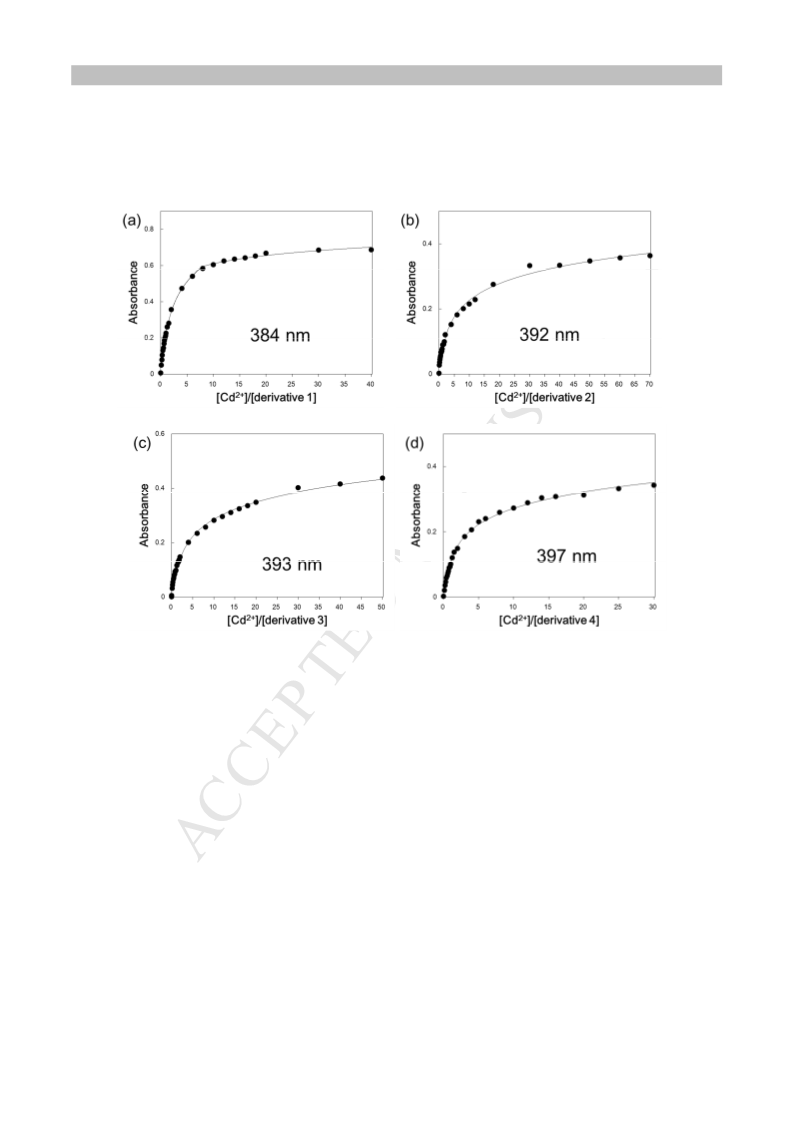
4.3. Fluorescence spectroscopy
4
.2.5. N,N’-Bis(8--quinolyl)-2-benzyl-2-methylmalonamide (3)
8-aminoquinoline (3.3 g) and dry pyridine (5.0 mL) were
Fluorescence emission spectra were recorded at room
temperature. A 1 cm × 1 cm quartz cuvette was used for the
spectroscopic analysis. Stock solutions of fluoroionophores in
DMSO (160 µM) or DMF (200 µM) were prepared for
fluorescence detections and diluted to a final concentration of 5.0
µM or 2.0 µM by mixing 500 µM stock solutions of inorganic
acetates (CH COOLi, CH COONa, CH COOK, CH COONH ,
dissolved in dry benzene (200 mL). In a dark room, the dry
benzene solution (40 mL) of 2-benzyl-2-methyl malonyl chloride
(2.3 g) was added dropwise to the solution and stirred for 18h at
room temperature. Deionized water (50 mL) and chloroform (80
mL) were added to the solution. The solution was extracted with
chloroform (100 × 3 mL) and washed with water (50 × 2 mL).
The organic layer was dried over anhydrous magnesium sulfate,
3
3
3
3
4
2
2
Mg(CH
3
COO)
2
, Ca(CH
, Ni(CH
, and CH
3
COO)
2
, Al(CH
, Cu(CH
3
COO)
3
, Mn(CH COO)
3
, Zn(CH COO)
3
,
,
filtrated and evaporated under reduced pressure. Yield: 1.99 g,
Co(CH
Cd(CH
3
COO)
COO)
2
3
COO)
2
3
COO)
2
-
4
.
6%; pale yellow solid; mp 150.1-155.7 °C. IR(KBr): 1668.3 cm
3
2
3
COOAg). The excitation wavelengths
1
1
3
H NMR (300MHz NMR, CDCl ): 1.79 (s, 3H), 3.65 (s, 2H),
were 345 nm (for DMSO) and 320 nm (for DMF) and the
emission spectra from ca. 320 to 700 nm were collected (every 2
nm). Excitation and emission slits width were 5 nm.
7
2
8
.16-7.30 (m, 5H), 7.44 (dd, 2H, J = 4.20 and 8.40 Hz), 7.53 (dd,
H, J = 2.10 and 8.40 Hz), 7.54 (dd, 2H, J = 6.75 and 8.40 Hz),
.14 (dd, 2H, J = 1.80 and 8.40 Hz), 8.85 (dd, 2H, J = 2.10 and
13
6.75 Hz), 8.85 (dd, 2H, J = 1.80 and 4.20 Hz), 10.94 (br, 2H).
NMR (75MHz NMR, CDCl ): 18.5, 44.9, 57.4, 116.8, 121.6,
121.9, 126.9, 127.1, 127.8, 128.2, 130.2, 134.3, 136.1, 136.4,
C
Likewise, for the investigation of solvent effects, 100 µM
stock solutions of fluoroionophores and inorganic perchlorates in
3
3 3
CH CN/CHCl (9:1, v/v) were prepared. inorganic perchlorates

 Moriuchi-Kawakami, Takayo
Moriuchi-Kawakami, Takayo
 Kawata, Keita
Kawata, Keita
 Nakamura, Sho
Nakamura, Sho
 Koyama, Yoshiaki
Koyama, Yoshiaki
 Shibutani, Yasuhiko
Shibutani, Yasuhiko