Malonylgenistin and Malonyldaidzin Conversion Kinetics
J. Agric. Food Chem., Vol. 55, No. 9, 2007 3413
in soy foods: extraction conditions and analysis by HPLC-mass
spectrometry. J. Agric. Food Chem. 1994, 42, 2466-2474.
(5) Klump, S. P.; Allerd, M. C.; MacDonald, J. L.; Ballam, J. M.
Determinination of isoflavones in soy and selected foods
containing soy by extraction, saponification, and liquid chro-
matography: collaborative study. J. AOAC Int. 2001, 84, 1865-
1883.
(6) Murphy, P. A.; Barua, K.; Hauck, C. C. Solvent extraction
selection in the determination of isoflavones in soy foods. J.
Chromatogr., B 2002, 777, 129-138.
(7) Lin, F.; Giusti, M. M. Effects of solvent polarity and acidity on
the extraction efficiency of isoflavones from soybeans. J. Agric.
Food Chem. 2005, 53, 3795-3800.
(8) Achouri, A.; Boye, J. I.; Belanger, D. Soybean isoflavones:
efficacy of extraction conditions and effect of food type on
extractability. Food Res. Int. 2005, 38, 1199-1204.
(9) Chien, J. T.; Hsieh, H. C.; Kao, T. H.; Chen, B. H. Kinetic model
for studying the conversion and degradation of isoflavones during
heating. J. Food Chem. 2005, 91, 425-434.
(10) Mathias, K.; Ismail, B.; Corvalan, C. M.; Hayes, K. D.
Temperature and pH effects on the conjugated forms of genistin
and daidzin isoflavones. J. Agric. Food Chem. 2006, 54, 7495-
7502.
(11) Wang, H. J.; Murphy, P. A. Mass balance study of isoflavones
during soybean processing. J. Agric. Food Chem. 1996, 44,
2377-2383.
(12) Batt, H. P.; Thomas, R. L.; Rao, A. Characterization of
isoflavones in membrane processed soy protein concentrate. J.
Food Sci. 2003, 68, 401-404.
(13) Rickert, D. A.; Meyer, M. A.; Hu, J.; Murphy, P. A. Effect of
extraction pH and temperature on isoflavone and saponin
partitioning and profile during soy protein isolate production. J.
Food Sci. 2004, 69, C623-C631.
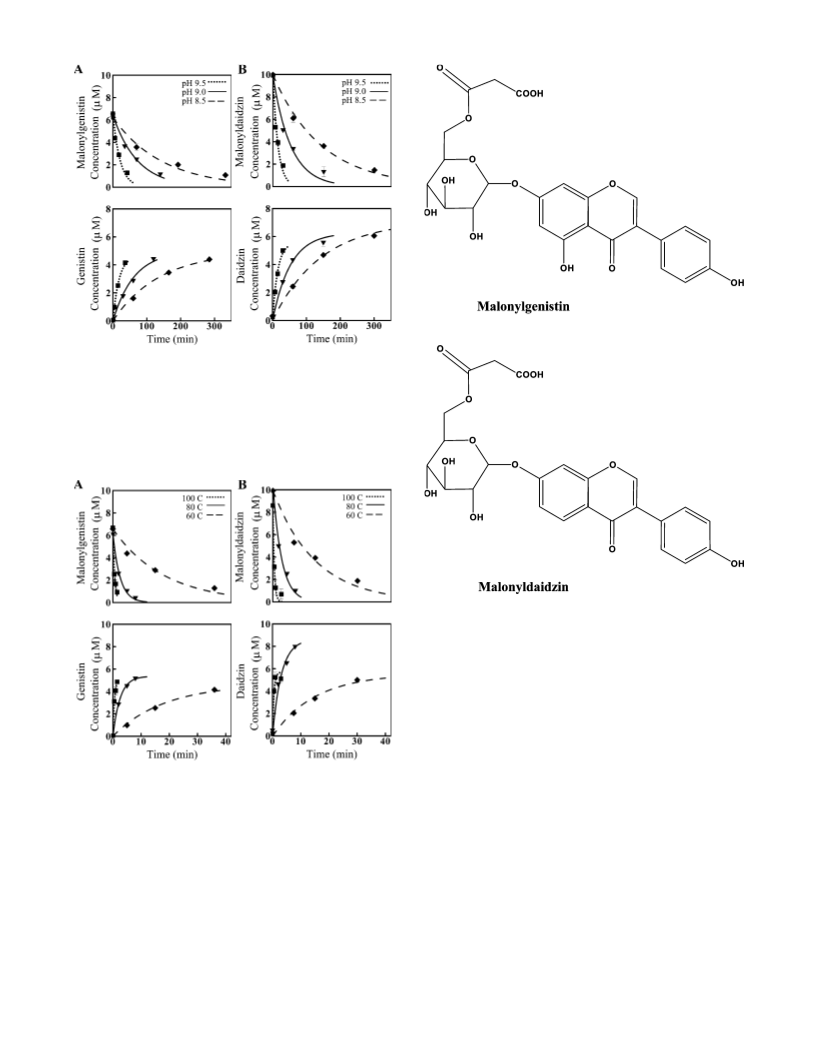
The relative simplicity of the kinetic equations, the accuracy
of the fittings, the trends in reaction rates, and the similarities
in activation energies as analyzed above suggested that the
proposed kinetics adequately described the chemical reactions
involved in the conversion of malonylglucosides, with a low
number of parameters. Results indicated that the small difference
in molecular structure of both malonylglucosides did not affect
the rate of conversion into â-glucosides. However, as pH and
heat increase, the difference in molecular structure affected
significantly the rate of degradation of each malonylglucoside
and its respective â-glucoside. This work confirmed that, along
with conversion, degradation occurs, resulting in loss. In real
heated systems, similar degradation might be occurring, leading
to loss that is independent of waste streams and protein matrices.
To confirm this assertion, complementary kinetic work ought
to be done in real systems, taking into account the protective
effect of proteins on isoflavone chemical modification, as well
as the effect of protein denaturation (due to pH and temperature)
on the rate of extraction of each type/form of isoflavone.
Overall, the findings of this work highlighted the importance
of the chemical structure of isoflavone with respect to stability
and provided useful information to manufacturers aiming to
minimize loss of isoflavones to enhance the nutritional value
of their soy products, without sacrificing the quality character-
istics. Therefore, while nutritionists investigate the biological
significance of each type/form of isoflavone, it is a necessity
to fully understand their distinct reaction to typical processing
conditions and thus minimize loss.
(14) Ismail, B.; Hayes, K. â-Glycosidase activity toward different
glycosidic forms of isoflavones. J. Agric. Food Chem. 2005, 53,
4918-4924.
LITERATURE CITED
(15) Bates, D. M.; Watts, D. G. Nonlinear Regression Analysis and
Its Applications; Wiley: New York, 1988.
(1) Setchell, K. D. R.; Zimmer-Nechemias, L.; Cai, J.; Ileubi, J.
Isoflavones content of infant formulas and the metabolic fate of
these phytoestrogens in early life. Am. J. Clin. Nutr. 1998, 68,
1453S-1461S.
(16) Wolfram, S. The Mathematica Book, 5th ed.; Wolfram Media/
Cambridge University Press: Cambridge, U.K., 2003.
(17) Van Boekel, M. A. J. S. Statistical aspects of kinetic modeling
for food science problems. J. Food Sci. 1996, 61, 477-485.
(18) Ungar, Y.; Osundahunsi, O. F.; Shimoni, E. Thermal stability
of genistin and daidzin and its effect on their antioxidant activity.
J. Agric. Food Chem. 2003, 51, 4394-4399.
(2) Hendrich, S.; Murphy, P. A. Isoflavones: source and metabolism.
In Handbook of Nutraceuticals and Functional Foods; Wildman,
R. E. C., Ed.; CRC Press: Boca Raton, FL, 2001; pp 55-75.
(3) Jackson, C. J. C.; Dini, J. P.; Lavandier, C.; Rupasinghe, H. P.
V.; Faulkner, H.; Poysa, V.; Buzzell, D.; DeGrandis, S. Effects
of processing on the content and composition of Isoflavones
during manufacturing of soy beverage and tofu. Process.
Biochem. 2002, 37, 1117-1123.
Received for review December 5, 2006. Revised manuscript received
February 22, 2007. Accepted February 22, 2007.
(4) Barnes, S.; Kirk, M.; Coward, L. Isoflavones and their conjugates
JF063511Z

 Vaidya, Nirupama A.
Vaidya, Nirupama A.
 Mathias, Kevin
Mathias, Kevin
 Ismail, Baraem
Ismail, Baraem
 Hayes, Kirby D.
Hayes, Kirby D.
 Corvalan, Carlos M.
Corvalan, Carlos M.