
Tetrahedron Letters p. 1381 - 1384 (2000)
Update date:2022-08-17
Topics:
 Pascual, Alfons
Pascual, Alfons
 Ziegler, Hugo
Ziegler, Hugo
 Trah, Stephan
Trah, Stephan
 Ertl, Peter
Ertl, Peter
 Winkler, Tammo
Winkler, Tammo
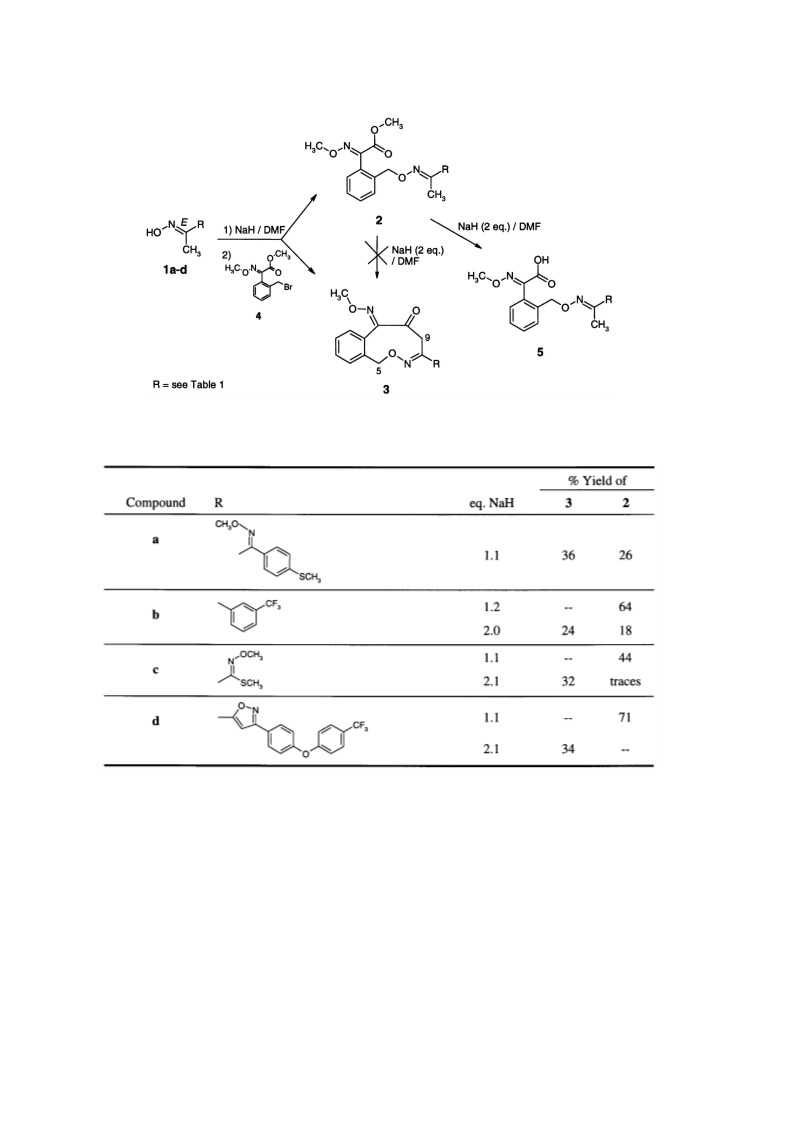
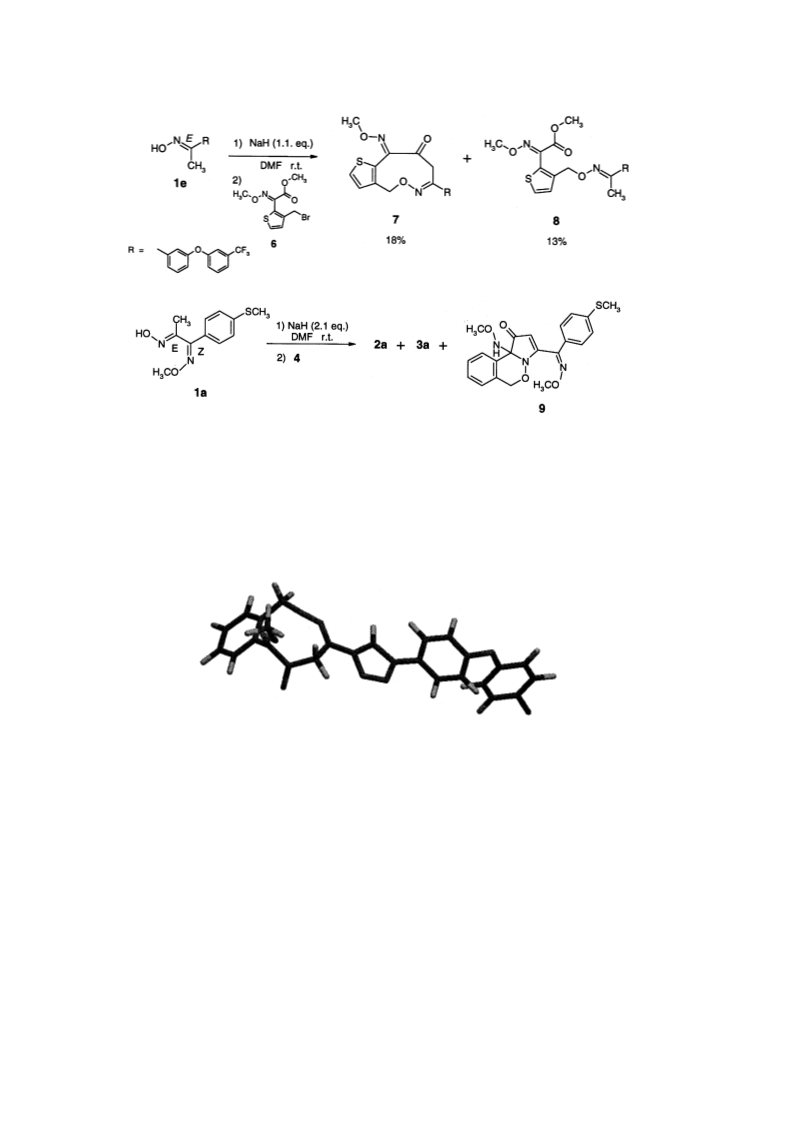
Reaction of (2-bromomethyl-phenyl)-methoxyimino-acetic acid methyl ester 4 with oximes 1 in the presence of NaH/DMF yields 8-substituted 5H,9H-6-oxa- 7-aza-benzocyclononene-10,11-dione-11-O-methyloximes 3 together with the expected open chain compounds 2. Some spectroscopic data as well as synthetic and mechanistic aspects of the formation of the novel compounds 3 are discussed. (C) 2000 Elsevier Science Ltd.
View More
Qingdao XinYongAn Chemicals Co., Ltd
Contact:+86-532-81107967
Address:Chengyang dual-port industrial park by the sea,Qingdao
NingBO Hong Xiang Biochem.Co.Ltd
website:http://www.hxbiochem.com
Contact:0574-66003444
Address:Ning Bo Bei Lun
Hangzhou Sartort Biopharma Co., Ltd
website:http://www.sartort.com
Contact:86-571-87039693
Address:No. 57, Tech Park Road, Hangzhou, Zhejiang, China
Tianjin Dongchang Fine Chemical Industry Co., Ltd.
Contact:+86-22-29894595
Address:Economic Developing Zone, Ji County, Tianjin, China
Hubei Danao Pharmaceutical Co.,Ltd.
website:http://www.danaopharm.com
Contact:+86-719-5251167
Address:Fandan Road,Danjiangkou,Hubei
Doi:10.3184/030823409X12616597939085
(2010)Doi:10.1021/jm500870u
(2014)Doi:10.1021/ma050283l
(2005)Doi:10.1002/smll.201000981
(2010)Doi:10.1002/chem.201502955
(2015)Doi:10.1016/S0039-128X(63)80131-2
(1963)