
Journal of the American Chemical Society p. 949 - 958 (1991)
Update date:2022-08-11
Topics:
 Newcomb, Martin
Newcomb, Martin
 Manek, M. Beata
Manek, M. Beata
 Glenn, Anne G.
Glenn, Anne G.
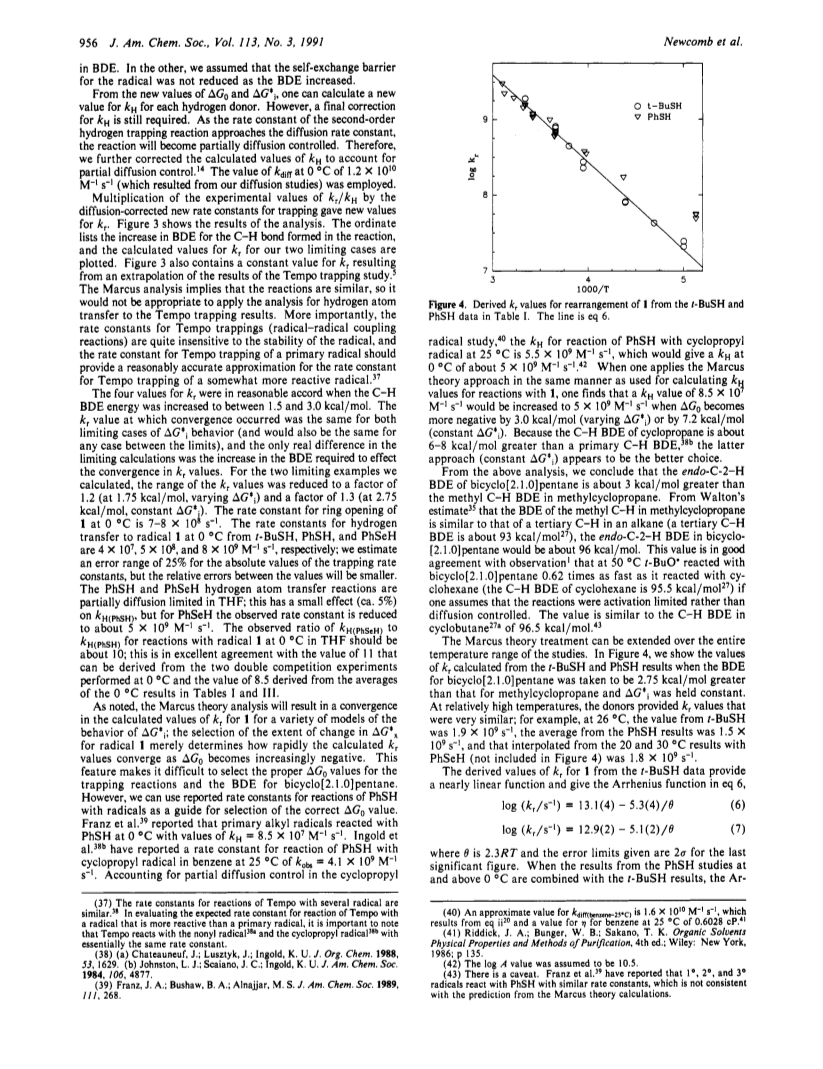
Relative rate constants (kr/kH) for ring opening of the bicyclo[2.1.0]pent-2-yl radical (1) to the cyclopent-3-enyl radical and trapping of radical 1 with t-BuSH, PhSH, and PhSeH in solvent THF were measured at temperatures between -78 and 50 °C. The hydrogen atom donors reacted more rapidly with radical 1 than with the cyclopropylcarbinyl radical (6). Rate constants for ring opening of 1 (kr) could be obtained by estimating the values of kH via Marcus theory. From initial kH values for reactions with radical 6, new kH values were calculated for increasingly exergonic reactions until the derived kr values from the three trapping agents agreed with one another and an extrapolated value of kr from Tempo trapping of 1. The results suggest that hydrogen atom transfer reactions with 1 were about 3 kcal/mol more exergonic than reactions with 6. Arrhenius functions for ring opening of 1 averaged log (ks/s-1) = 13.0 - 5.2/2.3RT; the value of kr at 25 °C is 1.5 × 109 s-1. Trapping studies of 1 and 6 with 2,6-dimethylthiophenol indicated that no special steric effects were present in hydrogen atom transfers to 1. However, highly stereoselective trapping of 1 was observed in reactions with ArSD with endo-bicyclo[2.1.0]pentane-2-d predominating, and the rate constant for decarboxylation of the endo-bicyclo[2.1.0]pentane-2-carboxy radical (endo-3) at -78 °C apparently was greater than that for decarboxylation of exo-3. The stereochemical results are ascribed to a stereoelectronic effect between the C1-C4 bond and endo-C2-X bonds of bicyclo[2.1.0]pentanes that weakens endo-C2-X bonds.
View More








Doi:10.1016/S0040-4039(01)84925-X
(1972)Doi:10.1021/acs.jmedchem.8b00460
(2018)Doi:10.1016/0031-9422(92)80083-Q
(1992)Doi:10.1021/jo00341a026
(1982)Doi:10.1002/oti.127
(1906)Doi:10.1016/j.mcat.2017.10.019
(2017)