
RSC Advances p. 30439 - 30447 (2019)
Update date:2022-08-11
Topics:
 Wang, Yongsheng
Wang, Yongsheng
 Zhao, Zhenzhen
Zhao, Zhenzhen
 Zhao, Yunlu
Zhao, Yunlu
 Lan, Xiaolin
Lan, Xiaolin
 Xu, Weixiang
Xu, Weixiang
 Chen, Li
Chen, Li
 Guo, Dongjie
Guo, Dongjie
 Duan, Zhengkang
Duan, Zhengkang
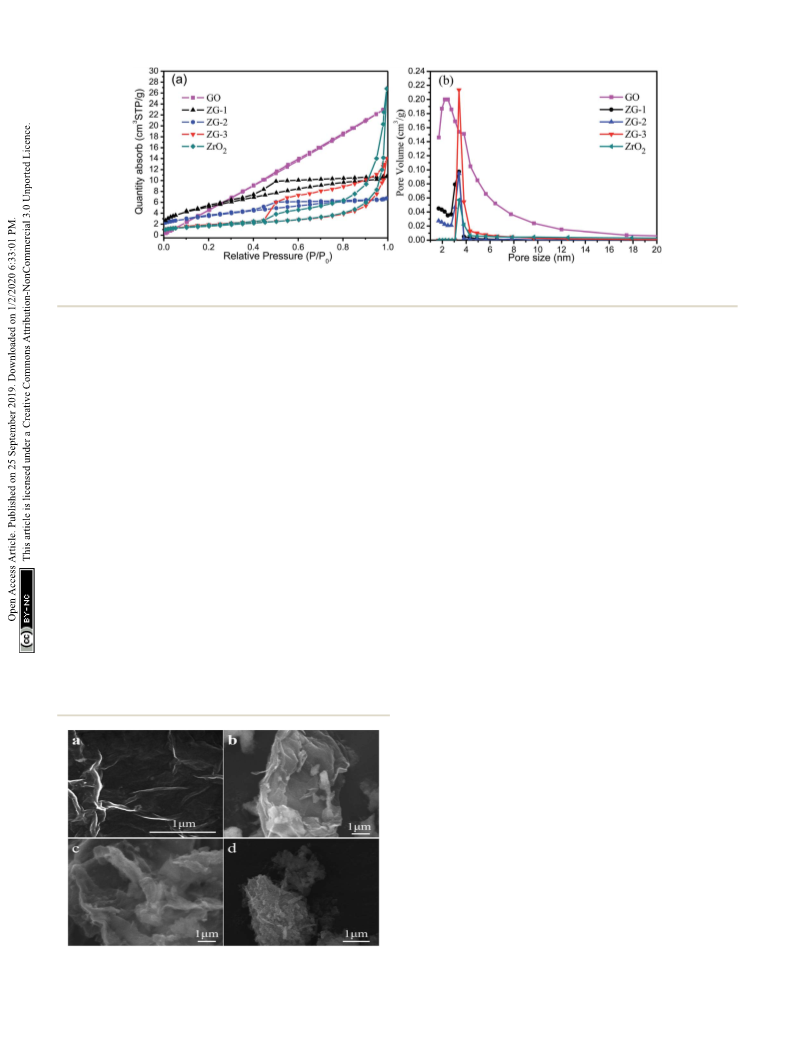
The sintering resistance of supported Cu nanoparticle (NP) catalysts is crucial to their practical application in the dehydrogenation of diethanolamine (DEA). In this paper, co-precipitation, hydrothermal synthesis, and sol-gel condensation are used to form a new support material through chemical bonding between graphene oxide and ZrO2. The composite carriers prepared by the three methods are mixed with copper nitrate and ground using a ball mill. A series of Cu/ZrO2-reduced graphene oxide (RGO) composites were prepared by calcination under nitrogen at 450 °C for 3 h and hydrogen reduction at 250 °C for 4 h. The conversion of DEA to iminodiacetic acid (IDA) reached 96% with the Cu/ZrO2-RGO catalyst prepared by hydrothermal synthesis. The conversion rate of DEA is more than 80% following the reuse of the CZG-2 catalyst for twelve cycles. The various physicochemical characterization techniques show that the Cu/ZrO2-RGO layered and wrinkled nanostructures can improve catalytic stability and suppress the sintering of the supported Cu NPs during the catalytic dehydrogenation of diethanolamine. A synergistic effect between the RGO and the Cu nanoparticles is observed. The Cu nanoparticles with RGO have a better dispersibility, and a new nano-environment is created, which is the key to improving the efficiency of diethanolamine dehydrogenation. These new Cu/ZrO2-RGO catalysts show increased durability compared to commercially produced Cu/ZrO2 catalysts and show promise for practical applications involving diethanolamine dehydrogenation.
View More







Guilin Zhenda Bio-Tech Co., Ltd.
Contact:86-773-3568977
Address:-Yangtang shangshui industry park, Lingui county,
jiangsu haian chemical co.,ltd.
Contact:86-513-15851283853
Address:No.99,Changjiang West Road,Haian County,Jiangsu Province,China
Taizhou Crene Biotechnology co.ltd
Contact:86-576-88813233 88205808
Address:Economic Developed Zone of Taizhou Zhejiang China
Contact:86-0510-85133006
Address:hubin road NO157
Xi'an Galaxy Chemicals CO., Ltd
Contact:86-29-89380370
Address:No.8, Gaoxin three road, Xi'an city.
Doi:10.1021/acs.organomet.8b00086
(2018)Doi:10.1021/acs.jafc.7b00987
(2017)Doi:10.1039/c4cy01168f
(2015)Doi:10.1002/aoc.4481
(2018)Doi:10.1002/ptr.5545
(2016)Doi:10.1021/jo950857s
(1996)